Here, we seek to explore some controversial, poorly known, or irrelevant drugs in medicine in a menagerie of drug cocktails.
1. What drugs best treat SVT?
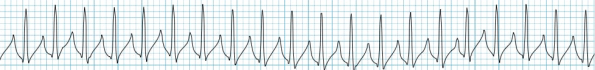
A patient presents with the following rhythm strip, they’re totally stable, on no medications and with no significant medical problems. Take a minute, think about how you are going to treat it.
Thought about it? Great. There appears to be significant practice variation when it comes to the treatment of stable SVT, but hopefully, you’re thinking of using either Diltiazem or Adenosine.
Adenosine is what most people are perhaps the most comfortable with given its historical use – but, it makes people feel awful. Fortunately, Diltiazem is a much better-tolerated alternative. A Cochrane review encompassing 7 RCT’s and over 600 patients found that diltiazem was effective at terminating SVT 93% of the time, compared to 90% with adenosine. The adverse events recorded with diltiazem were rare (0.6%), but much more frequent with adenosine (10.8%). A concern related to diltiazem is the potential for causing hypotension, however, in this review of over 600 patients, only 0.6% (1/622) patients had documented hypotension. (1) For a more in-depth discussion on this literature, check out this great post from First10EM.
This one is pretty easy – diltiazem is better tolerated and as effective (if not more effective) than adenosine, so this is my first-line therapy for the treatment of uncomplicated, stable SVT.
But, let’s say you have to give adenosine – because the QRS is wide, or you have diagnostic uncertainly of the underlying rhythm, there are a few ways to enhance your efficacy and patient tolerance.
Pre-medication
The feeling of ‘death’ caused by adenosine is a result of bronchospasm and vasospasm, pre-medicating patients with nitroglycerine and Ventolin (2 sprays/2 puffs) helps to mitigate the bronchspasm/vasospasm, and the patient will often tolerate the adenosine better. There is no direct evidence looking at this, but the anecdotal experience of many is that it appears it is better tolerated in this manner.
One syringe to rule them all
We often have to tinker with a stopcock or using two syringes to quickly administer adenosine given its short half-life. However, there is evidence to support giving adenosine mixed into a 20 cc syringe of normal saline. (2,3) Admittedly the studies are small, however, there is a signal that this technique does not dilute the adenosine and increases the potential efficacy due to quicker delivery to the heart. A study by McDowell et al demonstrated first dose (6 mg) success of 73.1% compared to 40.7% with dual-syringe technique. Second dose success (12 mg) of 100% versus 70.4% with dual-syringe technique.
So what do I do? If I have a stable patient with uncomplicated SVT, I’ll always reach for diltiazem first. If I have diagnostic uncertainly, or concern for an underlying accessory pathway or the QRS is wide, I’ll give adenosine in a 20 cc syringe of NS after pre-medicating with nitro and Ventolin.
2. Can you load Dilantin orally?
Short answer? Yes, totally.
The problem with IV Dilantin in patients who are sub-therapeutic is that it may cause hypotension and decreased LOC (on a post-ictal patient, this is quite the double whammy), and result in prolonged ED length of stay. In the patient who is at baseline now (and able to tolerate po) there is evidence that a single oral dose of Dilantin (1000 mg) can achieve therapeutic serum concentrations between 3-14 hours. (4-7) The main downside associated with oral Dilantin is GI intolerance, so there is also evidence supporting two separate doses of Dilantin (500 mg) two hours apart. This also results in therapeutic serum concentrations within 18-24 hours after administration.
The one benefit to IV Dilantin is that it’ll result in therapeutic serum concentrations sooner, and so in your patient with multiple seizures, or who still remains altered, this would be the preferred method of administration.
In the patient who is otherwise well, it is reasonable to administer 500 mg po Dilantin at assessment, and another 500 mg two hours later (could be given after discharge, if the patient is reliable). This will help facilitate ED flow and decrease the length of stay for these patients.
3. Let’s talk about antibiotics
Cross-reactivity
Many practitioners and educators continue to believe and propagate the myth that penicillin and cephalosporin cross-reactivity approaches 10%. Unfortunately, this is a myth based on bad science. To understand where this comes from, you need to know that in the 1950s, penicillins and cephalosporins were manufactured in labs using the same fungus, and so you had physical cross-contamination of powders, resulting in the phenomena that is reported today.
Further evidence has shown that the actual rate of cross-reactivity between cephalosporins and penicillins approaches 0%. (8-10) In fact, patients with a penicillin allergy are 3-4 times more likely to have an allergic reaction to a structurally unrelated-antibiotic, suggesting that a penicillin allergy is a general risk factor for antibiotic-associated allergies, and not specific to cephalosporins.
So don’t be afraid to give your patient a cephalosporin in the setting of a penicillin allergy, if it is the appropriate antibiotic choice! Severe allergic reactions to cephalosporins occur in about 0.02% of patients who take them, so the probability of a patient having an adverse event is quite low.
Most Penicillin allergies aren’t real…
Since we’re talking about penicillin, it is important to note that only 10% of reported penicillin allergies are real. This can be a significant problem because we often end up treating patients with second or third-line therapies, or using unnecessarily broad-spectrum antibiotics, which can have significant resistance implications. (11)
This can have significant harms, especially on a population level. To give you an example, let’s say you get bit by this super vicious dog:

My dog, super vicious, obviously.
First-line therapy would ideally be Amox-Clav, however, given a ‘documented’ penicillin allergy, you have a few other options:
- Monotherapy with a fluoroquinolone
- Doxy or Septra + Clinda or Flagyl
- Something worse
Ugh, that is a nasty combination of antibiotics, for what is probably not a real allergy.
So what do I do? I ask patients what their penicillin allergy represents, if it is anaphylaxis, ok sure – stay away from the PCN. But if the answer is almost anything else (I had a rash, I was a kid etc), trial a dose of Amox-Clav in the ED! If no reaction occurs, the patient can leave with their prescription in 30-60 minutes (important to document on the script that you are aware of the allergy and you tested the patient in the ED, to ensure the pharmacy doesn’t send it back). Many people who state that they’re allergic to penicillin, actually got treated for ‘strep throat’ (when it was actually mono) and developed the classic Amoxicillin/EBV rash.
IV Antibiotics
I know for sure that you have heard the following:
“Wow, that cellulitis looks really bad, we need to use some strong IV Antibiotics” or “my doctor said I need IV antibiotics for this infection I have”. Amongst patients and practitioners, it is falsely assumed that IV antibiotics are superior to oral antibiotics. A great quote from First10EM to demonstrate the fallacy in this thinking: “the bacterium living in your tissues remains ignorant of the antibiotic’s port of entry.”
The truth is, there is a plethora of research looking at comparing IV and oral antibiotics, and excluding all sepsis-based research, the studies all suggest that oral antibiotics are non-inferior to IV antibiotics, and in some studies, oral antibiotics performed better!
There is a Cochrane review looking at 25 RCTs for the treatment of cellulitis, and found that; “Surprisingly, oral antibiotics appeared to be more effective than antibiotics given into a vein for moderate and severe cellulitis.” (12)
Similarly, in studies on pneumonia in adults and children, pediatric and adult UTIs, pyelonephritis, chronic osteomyelitis, and febrile neutropenia, no difference was found between IV and oral antibiotics, often with a trend favouring oral antibiotics. For a much more in-depth look at the literature, check out another great summary from First10EM.
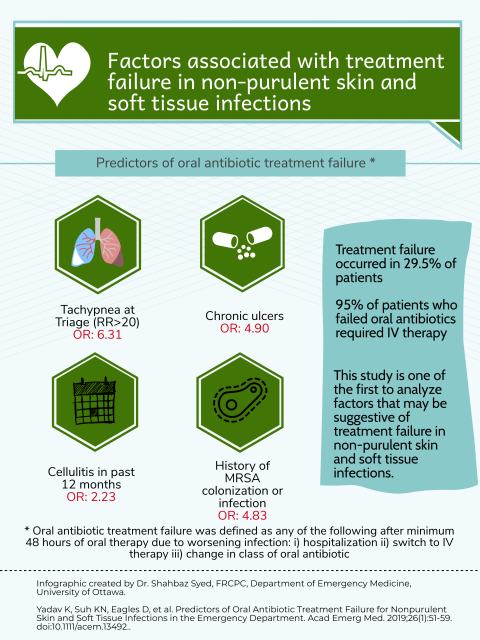
An IDSA guideline supports these findings by suggesting that in patients with non-purulent cellulitis, IV antibiotics should be reserved for patients who are immunocompromised or have systemic signs of infection, hemodynamic instability or altered mental status. It has also been found that adherence to these guidelines results in decreased treatment failure rates amongst ED patients. (13)
What about a single dose of IV antibiotics?
We often see in the ED, a patient is given a dose of IV antibiotics and then sent home on an oral antibiotic, or patients are switched to IV antibiotics after less than 48 hours of oral therapy under the guise of ‘treatment failure’ despite the fact that there is no evidence to support this practice. With no evidence to support this practice, there are significant potential documented harms. When a patient is given a dose of IV antibiotics and sent home on oral antibiotics it increases the risk of diarrheal illness and C. Diff (OR 2.73, 95% (1.38-5.43)) compared to those who were just sent home on oral antibiotics. (14) I think every patient deserves a 48-72 hour trial of oral antibiotics before deciding to change antibiotics. It should also be noted that often, changing classes of antibiotics is sufficient, rather than starting IV antibiotics in most cases.
Treatment failure
Now, if you remain reluctant or hesitant to change your practice, perhaps an easier middle ground is to only consider IV antibiotics in patients with risk factors for treatment failure, research by Yadav et al (15) helps to provide some insights:
So what?
Ok, I have one last harm for you to consider that occurs as a result of inappropriate antibiotic use. One dose, of any antibiotic, can change an individual’s gut flora for 6-12 months, and in some cases, their native bacteria may never recover. (15-16)
While this doesn’t seem like a big deal, there has been evidence that has demonstrated the gut flora of patients with fibromyalgia, IBS, and IBD is altered in comparison to healthy controls. (15-16) Now, obviously, gut microbiology is complicated with multiple influencing factors, but it is very plausible that antibiotics are playing a role in these illnesses, and I think it would presumptuous of us to suggest that we are not having significant downstream impacts on patients as a result of our antibiotic usage.
4. What drugs treat Cannabis Hyperemesis Syndrome?
Usually, you can identify the patients suffering from this because you can hear the retching down the hallway of your ED. This can be a challenging disease entity to manage, as the symptoms can be difficult to manage. The evidence looking at this rather novel disease entity is weak, but there are some treatment suggestions available.
I do think an approach to these patients is important, because they often have frequent ED presentations, and practice variation is significant. If we’re able to treat them early in their ED course, we can often decrease their length of stay and minimize a lot of unnecessary investigations – I’m specifically thinking about imaging. Their abdominal discomfort often pushes people towards imaging, but aggressive anti-emetic therapy may lead you down a more conservative route if they improve.
Zofran
Often a mainstay of treatment, conventional dosing is 4 mg –> 8 mg, however, in the cyclical vomiting literature, doses up to 16 mg are reported (I’d get an ECG to check the QTc before giving this kind of dose). (17)
Haloperidol
2 mg IV Haloperidol is an excellent option for controlling symptoms in these patients. (18) The literature does extrapolate using 5 mg IV from the gastroparesis world, however, anecdotally I have seen quite a bit of EPS with this large a dose, and so 2 mg seems to be a nice sweet spot as an initial dose. Unfortunately, there are no trials looking at the appropriate dosing and effectiveness of Haldol in these patients, so most of our knowledge around this is experiential thus far.
Capsaicin cream
0.075% applied topically over the abdomen. Don’t know why it works, but it certainly appears to be effective, and the patient can be discharged with a prescription. (19-20)
5. Does Tamiflu work?
It doesn’t. Trying to prove that it ‘does’ work has become a huge mess.
Historically, Roche has been reluctant to reveal the final trial data on tamiflu, and the BMJ had been involved in a lengthy legal battle with Roche to attempt to access the data. Check here for some further insights into the messy history of this drug.
Ultimately, amongst multiple studies, it appears that Tamiflu has had no impact on complications, was not helpful in prophylaxis but has multiple known harms.
- NNT: None, does not decrease hospitalizations or complications
- NNH:
- Vomiting: 22
- Nausea: 28
- Headaches: 32
- Neuropsych: 94
- Adverse renal events: 150
A Cochrane review summarized the data on Tamiflu, and the only benefit was a decrease in symptoms by 17 hours, but these studies have significant biases and author conflicts. Additionally, many of the associated harms were not reported in the original studies. (21)
In 2020, the Lancet published the largest study on Tamiflu and suggested that patients treated with the medication had symptom resolution on average one day sooner. (22) A bold claim, and one worth diving into:
This was a pragmatic, open-label, multi-center randomized trial (non-blinded)
- Population: Patients with a flu-like illness within 72 hours of symptom onset
- Intervention: Tamiflu
- Control: No treatment
- Outcome: Patient reported symptom recovery
Results:
- 3266 patients
- Duration of illness decreased in the Tamiflu group (5.7 vs 6.7 days)
- No difference in any other outcomes.
Now, ultimately, this is an unblinded trial that is measuring a subjective outcome, so any conclusions drawn from this trial are merely just demonstrating the placebo effect. Patients know which therapy they’re getting, and so the subjective reporting of their symptoms is ultimately meaningless.
Interestingly, taking a deeper dive into the study, Tamiflu was equally effective whether or not the patient actually had the flu, and there is no reason that Tamiflu should make people without the flu feel any better. Again, ultimately, demonstrating the placebo effect.
6. What are the best drugs to treat Hyperkalemia?
As a medical student and resident, I was conventionally taught to treat Hyperkalemia acutely with:
- Calcium Gluconate 3 g IV (or calcium chloride)
- Insulin 10 u IV Regular
- D50W 1 amp
- +/- Ventolin Neb
- +/- Bicarbonate amp if they are acidotic
Using this ‘usual’ therapy, 17% of dialysis patients being treated for hyperkalemia develop hypoglycemia. This is often a result of the fact that insulin is going to last around 4 hours, while the dextrose lasts about one hour. (23)
Instead, some evidence suggests that using 5 units of Regular Insulin to shift the potassium is just as efficacious but results in less hypoglycemia. Additionally, the recommendation is to provide 2 amps of D50W either immediately, or a second dose in one to two hours after the first. (24)
References
- Alabed S, Sabouni A, Providencia R, Atallah E, Qintar M, Chico TJA. Adenosine versus intravenous calcium channel antagonists for supraventricular tachycardia. Cochrane Database of Systematic Reviews 2017, Issue 10. Art. No.: CD005154. DOI: 10.1002/14651858.CD005154.pub4
- McDowell M et al. Single-Syringe Administration of Diluted Adenosine. Acad Emerg Med 2019. PMID: 31665806
- Choi SC et al. A Convenient Method of Adenosine Administration for Paroxysmal Supraventricular Tachycardia. J Korean Soc Emerg Med 2003
- Osborn HH, et al. Single-dose oral phenytoin loading. Ann Emerg Med 1987;16(4):407-12. [PMID 3826809]
- Evens RP, et al. Phenytoin toxicity and blood levels after a large oral dose. Am J Hosp Pharm 1980;37(2):232-5. [PMID 7361796]
- Ratanakorn D, et al. Single oral loading dose of phenytoin: a pharmacokinetic study. J Neurol Sci 1997;147(1):89-92. [PMID 9094065]
- Record KE, et al. Oral phenytoin loading in adults: rapid achievement of therapeutic plasma levels. Ann Neurol 1979;5(3):268-70. [PMID 443759]
- Bhattacharya S. The facts about penicillin allergy: a review. J Adv Pharm Technol Res. 2010;1(1):11‐
- Apter AJ, Kinman JL, Bilker WB, et al. Is there cross-reactivity between penicillins and cephalosporins?. Am J Med. 2006;119(4):354.e11‐e3.54E19. doi:10.1016/j.amjmed.2005.10.052
- Herbert ME, Brewster GS, Lanctot-Herbert M. Medical myth: Ten percent of patients who are allergic to penicillin will have serious reactions if exposed to cephalosporins. West J Med. 2000;172(5):341. doi:10.1136/ewjm.172.5.341
- Patterson RA, Stankewicz HA. Penicillin Allergy. [Updated 2019 Apr 23]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2020 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK459320/
- Kilburn SA, Featherstone P, Higgins B, Brindle R. Interventions for cellulitis and erysipelas. Cochrane Database of Systematic Reviews 2010, Issue 6. Art. No.: CD004299. DOI: 10.1002/14651858.CD004299.pub2
- Dennis L. Stevens, Alan L. Bisno, Henry F. Chambers, E. Patchen Dellinger, Ellie J. C. Goldstein, Sherwood L. Gorbach, Jan V. Hirschmann, Sheldon L. Kaplan, Jose G. Montoya, James C. Wade, Practice Guidelines for the Diagnosis and Management of Skin and Soft Tissue Infections: 2014 Update by the Infectious Diseases Society of America, Clinical Infectious Diseases, Volume 59, Issue 2, 15 July 2014, Pages e10–e52, https://doi.org/10.1093/cid/ciu296
- Haran JP, Hayward G, Skinner S, et al. Factors influencing the development of antibiotic associated diarrhea in ED patients discharged home: risk of administering IV antibiotics. Am J Emerg Med. 2014;32(10):1195‐ doi:10.1016/j.ajem.2014.07.015
- Minerbi, A., Gonzalez, E., Brereton, N. J. B., Anjarkouchian, A., Dewar, K., Fitzcharles, M.-A., … Shir, Y. (2019). Altered microbiome composition in individuals with fibromyalgia. PAIN, Articles in Press. https://doi.org/10.1097/j.pain.0000000000001640
- Menees S, Chey W. The gut microbiome and irritable bowel syndrome. F1000Res. 2018;7:F1000 Faculty Rev-1029. Published 2018 Jul 9. doi:10.12688/f1000research.14592.1
- Witsil JC, Mycyk MB. Haloperidol, a Novel Treatment for Cannabinoid Hyperemesis Syndrome. Am J Ther. 2017;24(1):e64‐ doi:10.1097/MJT.0000000000000157
- Hickey J., Witsil J., Mycyk M. Haloperidol for treatment of cannabinoid hyperemesis syndrome. AJEM. June 2013; 31(6)p1003.e5-1003.e6
- Moon AM, Buckley SA, Mark NM. Successful Treatment of Cannabinoid Hyperemesis Syndrome with Topical Capsaicin. ACG Case Rep J. 2018;5:e3. Published 2018 Jan 3. doi:10.14309/crj.2018.3
- McConachie SM, Caputo RA, Wilhelm SM, Kale-Pradhan PB. Efficacy of Capsaicin for the Treatment of Cannabinoid Hyperemesis Syndrome: A Systematic Review. Ann Pharmacother. 2019;53(11):1145‐ doi:10.1177/1060028019852601
- Jefferson T et al. Oseltamivir for Influenza in Adults and Children: Systematic Review of Clinical Study Reports and Summary of Regulatory Comments. BMJ 2014. PMID: 24811411
- Butler CC, van der Velden AW, Bongard E, et al. Oseltamivir plus usual care versus usual care for influenza-like illness in primary care: an open-label, pragmatic, randomised controlled trial. Lancet. 2020; 395(10217):42-52. PMID: 31839279ISRCTN27908921
- Moussavi K, Fitter S, Gabrielson SW, Koyfman A, Long B. Management of Hyperkalemia With Insulin and Glucose: Pearls for the Emergency Clinician. J Emerg Med. 2019;57(1):36‐ doi:10.1016/j.jemermed.2019.03.043
- LaRue, H.A., Peksa, G.D. and Shah, S.C. (2017), A Comparison of Insulin Doses for the Treatment of Hyperkalemia in Patients with Renal Insufficiency. Pharmacotherapy, 37: 1516-1522. doi:1002/phar.2038