The COVID-19 pandemic has been the most globally disruptive event of a generation. But is COVID a once in a lifetime event, or just another in a long line of infectious threats? Human history has been marked by new and re-emerging illnesses often on a global scale. How likely are we to see further global pandemics in the near future? How long until the next one? And more importantly, what do we need to do to get ready?
Consider this case
You’re on a Sunday evening shift in your Emergency Department (ED). Wait times are 6 hours, beds are blocked, and the waiting room is full – a typical picture for Canadian tertiary care centres. A 25 year old patient presents to triage with a cough, bloody stool, and fever. Consider what will happen, starting from the time they walk through the front door.
- How will they be triaged? Is it likely this will be recognized as a high risk presentation?
- Will the triager know about a recent outbreak in, say Hong Kong or West Africa?
- Once triaged, where will this patient be put to wait? Will they go back to the main waiting room with everyone else?
- Once they are in the ED, will everyone immediately know what PPE to wear?
- Will this patient pose a threat to other patients? To staff? To you? To your family when you go home?
It’s a scary thought, but it’s one we’ve been living this for years – and well before COVID.
A History Lesson
“Those who cannot remember the past are condemned to repeat it.” – George Santayana
We have a lot to learn from history, but it is amazing how quickly events fade from collective memory. To many, COVID seems like an anomaly in history, but it is just the newest in a long line of pandemics in human history. COVID emerged just one year after the 100 year anniversary of the H1N1 influenza pandemic of 1918, also known as the Spanish flu. The Spanish flu was one of the deadliest pandemics in human history, and among the most disruptive to society. Looking back, there are so many parallels to that pandemic and now. Influenza had opened the 20th century in much the same way that COVID opened the 21stcentury. But there would be many more infections of global concern that would follow. Just 40 years ago we saw the advent of HIV, one of the longest continuous pandemics in human history and also among the deadliest. We’ve also faced highly profiled outbreaks of Ebola, Avian flu, and SARS; not to mention several re-emergences of influenza H1N1, most recently in 2009 as “Swine flu”.
The following table provides some highlights (not comprehensive) that help to frame some major points in history:
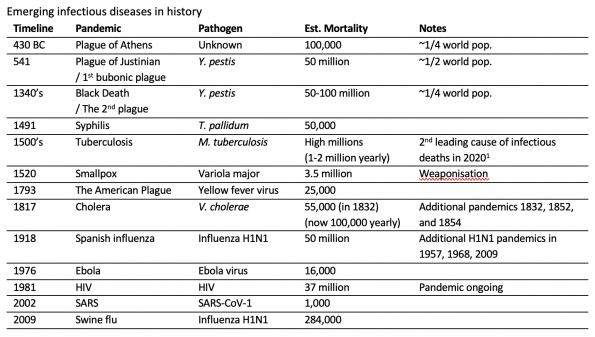
Several other outbreaks of significance in the 20th century have included (in no particular order): Malaria, Lassa virus, Marburg virus, Rift Valley Fever, West Nile virus, Chikungunya, Zika, Plague, Nipah virus, Polio, Salmonella, and Diphtheria2,3. This is not an exhaustive list. Adapted from Morens and Fauci (2020)3.
Are We Entering and Era of the Pandemic?
It is becoming clear that outbreaks are occurring with increasing frequency, and will likely continue to do so. In fact, just one week after giving this talk, reports of re-emergence of monkeypox in North American and European populations began making the news4. Is it that we are simply becoming better at detecting infections? Or is there evidence that supports an increase in outbreak frequency?
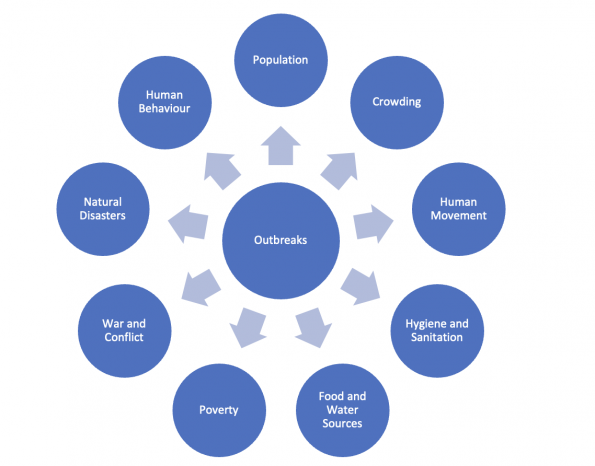
An increase in the rate of emerging infection events has indeed been shown5. The rate has increased nearly four-fold between 1940 to 2004, controlled for reporting bias. Most new infections are zoonoses (60.3%), meaning they originate as animal illnesses. The rate of emergence was most impacted by human population density and wildlife richness (a measure of diversity of species).
An increase in outbreak events was similarly seen from 1980 to 20136. There was an increase in total number of outbreaks, defined as first-occurrence events, but also in the number of unique endemic diseases within a given region at any single time point. In contrast, there was a small decrease in per capita cases. At first a decrease in per capita cases is a good thing, but this isn’t as good as it sounds. With a higher absolute number of outbreaks there is a greater chance that any individual one of them will undergo a high-risk mutation or otherwise breakthrough to be the next “big one”.
From this data, it is also possible to map “hot spots” where emerging infections may be more likely to emerge5. Unfortunately, there is a mismatch between geographical areas where there are greater efforts for data collection (namely, North America and western Europe) and areas where infection emergence events are more likely. From the perspective of global health and disease surveillance, this is a target for improvement.
How do New Infections Emerge?
We are seeing an increase in rate of new outbreaks. But how do infections suddenly become highly infectious pandemic-makers? Emerging infections arise from a complex interplay and balance between agent, host, and environment3. But in basic terms, there are essentially two ways in which an infection can emerge or re-emerge:
- Changes in geographical ranges or adaptations (e.g., an old infection in a new area);
- Changes in genetics allowing invasion of a new niche, often jumping to a new host species entirely (e.g., migration of SARS to human population)
Many emerging infections originate or propagate in non-human populations. A zoonosis is a human infection caused by an animal pathogen3. Of those diseases considered to be emerging, approximately 75% are zoonotic7. Most zoonoses lead to dead-end infections, meaning it doesn’t spread further2. Zoonotic spillover or host-switching is the process by which a pathogen adapted to one host species becomes adapted to another host species3. When zoonotic spillover occurs, there is much greater potential for human-to-human spread. Some prominent examples of infections emerging from zoonotic spillover are COVID, SARS, Nipah Virus, Ebola, and HIV,
There are a multitude of human and environmental contributing factors to increasing outbreaks.
Pandemic Recognition
One particularly important role of public health is data surveillance and identification of infection trends. The International Health Regulations are a very important international agreement when it comes to pandemic recognition8. This is international law that came into effect in 2007, agreed by most countries of the world including Canada. It details commitments for reporting and sharing of data, and for coordination of efforts. All nations are required to have capacity for surveillance and reporting, and this is shared globally through the World Health Organization (WHO). In Canada, the Public Health Agency of Canada (or PHAC) collates national data and shares it with the WHO9. Reporting is done through what is labelled the Canadian Notifiable Disease Surveillance System10. Provinces and territories each individually report data to the federal government through PHAC.
When enough data is gathered internationally on an outbreak of concern, there is a system in place through the WHO for deciding on what constitutes a Public Health Emergency of International Concern8. In 2009, influenza H1N1, or “Swine flu”, was the first such case declared under the International Health Regulations11. This method of recognition of disease is vey important to Canadians because it is far more likely that any major emerging infection will come from outside of Canada, having been first recognized somewhere else.
The WHO also creates reports on diseases that are high priority for research and development.
The WHO R&D Blueprint initiative was created in 201412. It is meant to help fast-track research and development in real time through data sharing and collaboration. They keep an updated list of high priority infections. These are diseases that are of high concern due to some combination of transmissibility, potential for spread, lack of effective vaccinations, or lack of therapies. The list always includes “Disease X”. Disease X is the explicit acknowledgement of diseases with pandemic potential that are not yet known to us.
What Should I Do? – ED issues in Pandemic Response
Think again about the case at the beginning of this post. As you think about the case, realize that many, if not most, emergency departments will have extremely limited pandemic preparedness plans. Most EDs are overcrowded and stretched to the limit already. Most will lack the resources to continually evaluate and develop plans if they do have them. And only a very select few departments will have leaders in emergency preparedness to rely on. When I gave this talk in May 2022, over of half of ER physicians I surveyed thought their ER would not be ready for the next pandemic. Yet, perhaps with either some cynicism or deference, over half also thought ER physicians would not be the ones to determine how the ED responds.
This may be a scary thought: The rate of infectious outbreaks is accelerating. It probably won’t be another 50 or 100 years to the next big pandemic. The threat of a pandemic could realistically become a norm in the way we work in the next generation in the ED. Even if not, at least some part of pandemic preparedness is going to affect us heavily in the coming years. What does working in a pandemic mean for ED practice? How can we best protect ourselves?
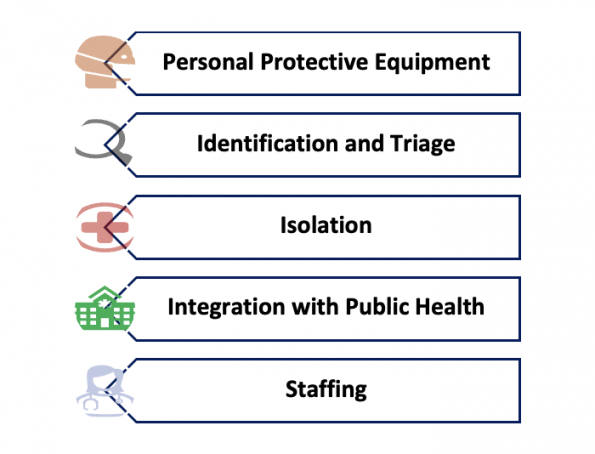
This is clearly a complicated question with many layers. The public health response crosses spectrums of health care, politics, economics, and culture. On an institution-wide level, disaster response and surge planning are crucial factors to response to emergencies. These factors are obviously extremely important to our practice, but there are other issues that are seen and felt directly by an ER physician on duty – the aspects that you are likely to run across on a day-to-day basis. Some of the major issues seen in the modern era (based on reviews and post-event analyses of recent major outbreak responses13,14), include personal protective equipment (PPE), identification and triage, isolation practices, integration with public health, andstaffing. These are issues commonly faced in any modern pandemic and they are likely to continue to be ongoing issues for us in the coming decades. I’ve selected just a few things to highlight with each, but these are complex topics that could each have their own post.
- Personal Protective Equipment – We all know how important PPE is. It’s one of those things we know, but time and time again we get it wrong. A few years pass, we forget about it, and we have to learn it all over again. The two central issues in PPE are around training and supply. A major challenge in PPE training is keeping everyone up to date. This is less of an issue now that we are two years into COVID, but think back to the beginning of the pandemic how many people really had no idea how to don and doff. In surveys of EDs worldwide early in the course of the COVID pandemic, 100% of respondents felt the need to overhaul or intensify their PPE training methods15,16 Despite the recognized importance of PPE, a scoping review of SARS and early COVID identified PPE shortages and training as gaps in the response to multiple prior pandemics14. PPE supply chain issues were identified by 50-60% of EDs in the early waves of the COVID pandemic15,16. Many methods for re-using PPE were developed out of necessity, like UV sterilizing, prolonged wearing, recycling, and 3D-printing.
- Identification and Triage – Triage history is extremely important for early identification, and almost all other processes depend on it. Case definitions are particularly important to making decisions on triage, isolation, contact tracing, and risk management. Definitions that are too narrow risk missing cases, whereas too broad and they over-triage creating burden and alert fatigue. When there is rapidly evolving and changing information, a challenge for the future is how to make this information as accurate as possible, while still being readily accessible in real time. Some potential solutions are improved methods for data sharing between hospitals and systems; involving a public health liaison at triage17; or perhaps leveraging artificial intelligence and machine learning18.
- Isolation – Similarly to what we saw PPE training and supply, isolation practice is typically an area of significant performance gap in pandemic responses. Nosocomial spread is all too common. Proper isolation process is crucial to protecting staff and patient’s alike. There are two main methods: Deflecting and Segregating. Deflecting involves methods to direct patients away from the ED. This could include things like community screening, testing, and triage. Many systems also have made use of “fever clinics” for well patients19,20. Segregating patients within the ED involves cohorting patients away from the rest of the ED population. But all attempts at good isolation require space. Many departments are simply not built in a way that promotes good infection prevention control and patient flow. Some EDs may need to be rebuilt with more side rooms. Others may need to optimize their design and process21.
- Integration with Public Health – Communication and integration of public health in clinical work is a constant challenge. This was a particularly troublesome gap identified in responses to SARS, H1N1, and early in COVID13. Crisis Standards of Care, have been applied to pandemics as an adaptation of an emergency response framework22. It outlines roles, administrative structures, and communication methods that improve operations under crisis conditions. Other frameworks also attempt to optimize communication between public health at the ED. For example, an article by Khan et al. reviews key components of effective communication between public health and emergency departments23.
- Staffing – A pandemic causes a prolonged stress to the health care system, and EDs in particular. It becomes very important to have a human resource plan with surge capacity to accommodate these shifts, and to build redundancy in the system. In the early COVID pandemic, almost half of EDs internationally did not have any backup staffing plan at all, and many found theirs inadequate16. Chronic understaffing at baseline only exacerbated the problem. Staff get exposed and need to isolate, or get infected and need to quarantine. This can be as much as 10-20% of staff at a given time. As the pandemic lingers, more and more staff are leaving health care altogether. Burnout is a major issue in health care, only worsened by the pandemic. Any ED pandemic readiness plan will have to consider staffing, and staff wellness as a top priority.
Closing thoughts
Here’s that scary thought again: Pandemics aren’t going away. They have been with us throughout history. And the rate is accelerating. It’s probably not going to be another 100 or even 50 years to the next big one. It is highly possible that we will be working under threat of pandemic more often than not in the coming decades.
We all know we need to be better prepared next time. But the same phrases have been said at every stage in history. A major challenge with preparedness is collective memory loss. The fear of COVID is already beginning to fade in our collective consciousness. As inflation kills our budgets, hard decisions are going to need to be made – but hopefully we can make sure compromising our safety is not one of them. If I can leave you with anything, it’s to realize that the core parts of ED pandemic preparedness that will protect us most are factors entirely in our control. As staff and leaders in our EDs, we must pay attention to things like PPE training and supply, triages processes, isolation practices, integration of ED care with public health, and managing our staffing. These are core principles of managing risk in the midst of a pandemic. Pandemics are becoming more common. In an era of pandemics, our EDs must be pandemic ready.
References
- World Health Organization. Tuberculosis. Published 2021. https://www.who.int/news-room/fact-sheets/detail/tuberculosis
- Morens DM, Folkers GK, Fauci AS. The challenge of emerging and re-emerging infectious diseases. Nature. 2004;430(6996):242-249. doi:10.1038/nature02759
- Morens DM, Fauci AS. Emerging Pandemic Diseases: How We Got to COVID-19. Cell. 2020;183(3):837. doi:10.1016/j.cell.2020.10.022
- Public Health Agency of Canada. Public Health Agency of Canada Confirms 2 Cases of Monkeypox.; 2022. https://www.canada.ca/en/public-health/news/2022/05/public-health-agency-of-canada-confirms-2-cases-of-monkeypox.html
- Jones KE, Patel NG, Levy MA, et al. Global trends in emerging infectious diseases. Nature. 2008;451(7181):990-993. doi:10.1038/nature06536
- Smith KF, Goldberg M, Rosenthal S, et al. Global rise in human infectious disease outbreaks. J R Soc Interface. 2014;11(101):20140950. doi:10.1098/rsif.2014.0950
- World Health Organization. A Brief Guide to Emerging Infectious Diseases and Zoonoses. (World Health Organization, ed.). WHO-SEARO; 2014.
- World Health Organization. International Health Regulations. (World Health Organization, ed.). WHO Press; 2005.
- Public Health Agency of Canada. Canada and the International Health Regulations (IHR): Overview. Published 2016. Accessed June 19, 2022. https://www.canada.ca/en/public-health/services/emergency-preparedness-response/international-health-regulations-2005.html
- Totten S, Medaglia A, McDermott S. Updates to the Canadian Notifiable Disease Surveillance System and its interactive website. Canada Commun Dis Rep. 2019;45(10):4257-4261. doi:10.14745/ccdr.v45i10a02
- Wilder-Smith A, Osman S. Public health emergencies of international concern: A historic overview. J Travel Med. 2020;27(8). doi:10.1093/jtm/taaa227
- Mehand MS, Al-Shorbaji F, Millett P, Murgue B. The WHO R&D Blueprint: 2018 review of emerging infectious diseases requiring urgent research and development efforts. Antiviral Res. 2018;159:63-67. doi:10.1016/j.antiviral.2018.09.009
- Cable J, Heymann DL, Uzicanin A, et al. Pandemic diseases preparedness and response in the age of COVID-19-a symposium report. Ann N Y Acad Sci. 2021;1489(1):17-29. doi:10.1111/nyas.14534
- Nadarajan GD, Omar E, Abella BS, et al. A conceptual framework for emergency department design in a pandemic. Scand J Trauma Resusc Emerg Med. 2020;28(1):118. doi:10.1186/s13049-020-00809-7
- Auerbach MA, Abulebda K, Bona AM, et al. A National US Survey of Pediatric Emergency Department Coronavirus Pandemic Preparedness. Pediatr Emerg Care. 2021;37(1):48-53. doi:10.1097/PEC.0000000000002307
- Mahajan P, Shu-Ling C, Gutierrez C, et al. A Global Survey of Emergency Department Responses to the COVID-19 Pandemic. West J Emerg Med. 2021;22(5):1037-1044. doi:10.5811/westjem.2021.3.50358
- Glasser JW, Hupert N, McCauley MM, Hatchett R. Modeling and public health emergency responses: lessons from SARS. Epidemics. 2011;3(1):32-37. doi:10.1016/j.epidem.2011.01.001
- Anderson RM, Fraser C, Ghani AC, et al. Epidemiology, transmission dynamics and control of SARS: The 2002-2003 epidemic. Philos Trans R Soc Lond B Biol Sci. 2004;359(1447):1091-1105. doi:10.1098/rstb.2004.1490
- Hall GG, Perry AG, VanDijk A, Moore KM. Influenza assessment centres: A case study of pandemic preparedness to alleviate excess emergency department volume. CJEM. 2013;15(4):198-205. doi:10.2310/8000.2012.120808
- Shih H-I, Ho T-S, Chang C-M, et al. Impacts of rapid flu clinic services at an emergency department during the pandemic flu season. Am J Infect Control. 2012;40(2):165-169. doi:10.1016/j.ajic.2011.03.006
- Douillet D, Saloux T, Ravon P, et al. Adaptation of ED design layout during the COVID-19 pandemic: a national cross-sectional survey. Emerg Med J. 2021;38(10):789-793. doi:10.1136/emermed-2020-211012
- Farcas A, Ko J, Chan J, Malik S, Nono L, Chiampas G. Use of Incident Command System for Disaster Preparedness: A Model for an Emergency Department COVID-19 Response. Disaster Med Public Health Prep. 2021;15(3):e31-e36. doi:10.1017/dmp.2020.210
- Khan Y, Sanford S, Sider D, et al. Effective communication of public health guidance to emergency department clinicians in the setting of emerging incidents: A qualitative study and framework. BMC Health Serv Res. 2017;17(1):312. doi:10.1186/s12913-017-2220-5