Traumatic brain injury (TBI) is a common yet challenging presentation in emergency medicine. As CT imaging becomes increasingly sensitive, clinicians are identifying more subtle intracranial injuries. The Brain Injury Guidelines (BIG) and Modified Brain Injury Guidelines (mBIG) were developed to help risk-stratify patients with traumatic intracranial hemorrhage and guide standardized, evidence-based decisions around observation, imaging, and disposition.
This post reviews how and why these guidelines were developed, the clinical criteria they incorporate, and the validation of both BIG and mBIG. It also explores their relevance in the Canadian emergency medicine context, their potential to improve resource use, and the practical challenges that may limit their broader adoption.
Watch the entire Grand Rounds here
Background
Traumatic brain injury (TBI) is a major driver of emergency department visits and hospital admissions across North America. In Canada alone, we see an average of 137,000 TBI-related emergency visits each year. Additionally, there are approximately 21,000 TBI-related hospitalizations reported annually. [1]
While the majority of these presentations are classified as mild TBIs, about 10% will have intracranial hemorrhage on CT. However, only 3.5% of these patients ultimately require neurosurgical intervention, a decision that frequently triggers interhospital transfer, even in otherwise stable patients. [20]
This is a scenario emergency physicians encounter daily, whether working in a tertiary care center, rural ED, or community hospital. Beyond the clinical impact, these cases carry significant system-wide implications: bed occupancy, patient flow, interfacility transfers, and variability in care.
As access to CT imaging has expanded and technology has improved, we are detecting increasingly subtle traumatic brain injuries, such as small contusions, isolated subdural hematomas, and trivial subarachnoid hemorrhages.
However, this diagnostic progress has introduced a new challenge: what do we do with these injuries?
Historically, the answer was to err on the side of caution. Any visible blood was considered high risk. Patients with even minor findings on CT scans were routinely admitted to monitored settings (ICU/NACU), referred to neurosurgery, and scheduled for repeat imaging.
At the time, this conservative approach made sense. Our understanding of TBI pathophysiology was limited, and we lacked evidence-based tools to safely identify patients at low risk of deterioration.
However, this approach is not without consequences. It strains hospital resources by occupying monitored beds with clinically stable patients, increases radiation exposure through repeat imaging, prolongs hospital stays, and introduces potential harms associated with hospitalization itself. In rural and community settings, it often results in the transfer of stable patients over long distances to trauma centers, separating them from their support systems and placing additional pressure on tertiary care facilities. Perhaps most concerning, it drives practice variation: two patients with identical CT findings may receive entirely different care depending on the hospital, the setting, or the provider.
It was within this context that the Brain Injury Guidelines (BIG), and more recently, the Modified Brain Injury Guidelines (mBIG), were developed.
The Brain Injury Guidelines (BIG)
The Brain Injury Guidelines (BIG) were first published in 2014 by Dr. Bellal Joseph, Dr. Peter Rhee, and colleagues at the University of Arizona in response to rising practice variation in the management of traumatic brain injury. Their goal was to create a simple, evidence-informed framework to help clinicians identify which patients with TBI could be safely managed without routine neurosurgical consultation, repeat imaging, or hospital admission. [2][7]
The guidelines were derived from a retrospective review of 3,803 patients with blunt TBI treated at a Level I trauma center. Among these, 1,232 patients had positive findings on their initial CT head. [2][7]
The researchers reviewed multiple factors and proposed the following components as part of the guidelines:
- Patient history, including intoxication, loss of consciousness, and use of antiplatelets or warfarin
- Neurologic examination
- Initial CT scan findings, specifically the type and size of intracranial hemorrhage and the presence of skull fractures

Based on these variables, patients were categorized into BIG 1, BIG 2, or BIG 3, each with corresponding management recommendations regarding observation, repeat CT imaging, and the need for neurosurgical consultation. [2]
BIG 1 – Low Risk Group

BIG, brain injury guidelines; antiplatelets, aspirin, Plavix; EDH, epidural hemorrhage; IVH, intraventricular hemorrhage; IPH, intraparenchymal hemorrhage; LOC, loss of consciousness; SAH, subarachnoid hemorrhage; SDH, subdural hemorrhage; skull #, skull fracture.
Patients in the BIG 1 group had mild injuries, normal neurologic exams, no anticoagulant or antiplatelet use, and were not intoxicated. CT findings were minimal and included:
- Subdural or epidural hematoma (≤4 mm)
- A single intraparenchymal hemorrhage (≤4 mm)
- Trace subarachnoid hemorrhage
- No intraventricular hemorrhage or skull fracture
Management:
Recommended 6 hours observation in the Emergency Department, No repeat head CT and no neurosurgery consult. If they remained neurologically stable and were reliable, they could be discharged directly from the ED.
Outcomes:
In the original study, they found that none of the 121 BIG 1 patient deteriorated or required neurosurgical intervention. So BIG 1 gave a safe framework to de-escalate care and reduce unnecessary admissions, consults, and radiation. [2]
BIG 2 – Moderate Risk Group
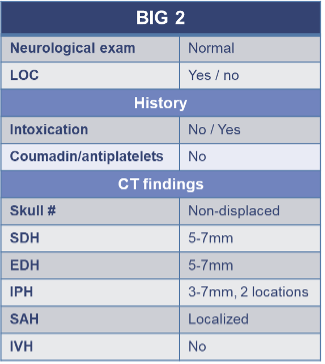
BIG, brain injury guidelines; antiplatelets, aspirin, Plavix; EDH, epidural hemorrhage; IVH, intraventricular hemorrhage; IPH, intraparenchymal hemorrhage; LOC, loss of consciousness; SAH, subarachnoid hemorrhage; SDH, subdural hemorrhage; skull #, skull fracture.
Patients in the BIG 2 category had a normal neurologic exam and could have mild risk factors such as intoxication, but were not on warfarin or antiplatelet agents. Imaging findings were slightly more significant but still localized and low risk overall, including:
- Subdural or epidural hematoma (5–7 mm)
- A single intraparenchymal hemorrhage up to 7 mm, or up to two small IPHs
- Localized subarachnoid hemorrhage
- Non-displaced skull fracture
No intraventricular hemorrhage, midline shift, or herniation was present.
Management:
Recommended hospital admission for observation (the duration not specified but usually less than 24 hours), with no requirement for routine repeat CT or neurosurgical consultation unless clinical deterioration occurs.
Outcomes:
In the original study, they found that 9 out of 313 patients (2.9%) in this group experienced deterioration (7 with CT progression, 2 with clinical decline), but importantly, none required neurosurgical intervention. This supports the idea that while BIG 2 patients need close monitoring, they could still be safely managed without routine consults or repeat imaging. [2]
BIG 3 – High Risk Group

BIG, brain injury guidelines; antiplatelets, aspirin, Plavix; EDH, epidural hemorrhage; IVH, intraventricular hemorrhage; IPH, intraparenchymal hemorrhage; LOC, loss of consciousness; SAH, subarachnoid hemorrhage; SDH, subdural hemorrhage; skull #, skull fracture.
BIG 3 included patients with severe injury or high-risk features such as abnormal neurologic exams, inability to assess neurologic status, anticoagulation or antiplatelet use, intoxication, or high-risk CT findings, including:
- Displaced skull fractures
- Intracranial hemorrhage ≥8 mm
- Intraventricular hemorrhage
- Scattered subarachnoid hemorrhage
- Multiple concurrent injuries
Management:
Patients in BIG 3 were considered to have severe traumatic brain injuries that required close monitoring. The management proposed, largely aligned with current standards of care for high-risk TBI, included admission (often to a higher level of care such as the ICU or NACU), mandatory neurosurgical consultation and repeat head CT imaging to monitor for progression.
Outcomes:
In the original study, 100% of the patients who required neurosurgical intervention, whether that was a craniectomy or EVD placement, came from the BIG 3 group. [2]
This early work laid the foundation for a practical risk stratification tool that could help safely reduce unnecessary hospital admissions, consultations, and imaging particularly in low-risk patients.
Validation of the Brain Injury Guidelines (BIG)

Validating the Brain Injury Guidelines: results of an American Association for the Surgery of Trauma prospective multi-institutional trial Joseph B, Obaid O, Dultz L, et al. J Trauma Acute Care Surg. 2022;93(2):157-165. [3]
In 2022, the first large prospective, multi-center validation of the Brain Injury Guidelines (BIG) was published in The Journal of Trauma and Acute Care Surgery. The study enrolled 2,033 patients aged 16 years or older with traumatic brain injury visible on initial head CT between 2018 and 2020 across nine Level I and II trauma centers in the United States. Patients were classified into BIG 1, 2, or 3 based on neurologic exam, use of anticoagulants or antiplatelets, intoxication status, and CT findings.[3] [4]
The primary outcome was the need for neurosurgical intervention, while secondary outcomes included clinical/neurological deterioration, radiographic progression on repeat CT, post-discharge ED visits, and 30-day readmissions.[3]
Key Findings [3]:
- In the BIG 1 group (301 patients):
- No patient required neurosurgical intervention
- Only 1.3% showed progression on CT, none of whom had clinical deterioration
- No 30-day readmissions or TBI-related ED revisits were reported
- In the BIG 2 group (295 patients):
- No patient required neurosurgical intervention
- 1% had radiographic progression, but only 0.7% worsened clinically
- Again, no post-discharge ED visits or readmissions occurred
- All neurosurgical interventions (280 cases, 19.5%) occurred in the BIG 3 group (1,437 patients)
Importantly, the study estimated that strict adherence to the BIG protocol could have safely avoided [3]:
- 425 repeat head CT scans
- 401 prolonged hospitalizations
- 511 neurosurgical consultations
However, the study did have several limitations that affect its generalizability:[3][4]
- 399 cases were excluded due to missing data
- All sites were high-resource trauma centers with neurosurgical access
- Long-term patient outcomes were not assessed
- No explanation was provided for specific admission decisions or care variation
Despite the limitations, these findings strongly support BIG as a safe and effective tool for identifying low-risk TBI patients who can be managed conservatively without the need for repeat imaging, neurosurgical consultation, or hospital admission. In particular, the BIG 1 and 2 categories accurately captured patients who did not require surgical intervention or escalation in care. While further validation is still needed in community and resource-limited settings, this study suggests that BIG could help emergency departments manage TBI more safely and efficiently, especially in well-appearing patients with minor intracranial hemorrhage. [3]
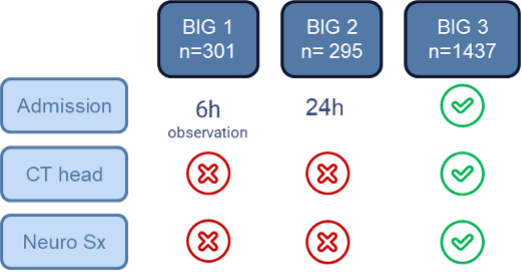
FIGURE 1- Validating the Brain Injury Guidelines: Results of an American Association for the Surgery of Trauma Prospective Multi-Institutional Trial.

Diagnostic utility of Brain Injury Guidelines (BIG): Systematic review and meta-analysis for prediction of neurosurgical intervention in traumatic brain injury. Kannan S, Gillespie CS, Lee KS, Phang I, McMahon CJ. Brain Inj. 2024;38(13):1093–1100 [12]
While the prospective validation offers strong support for the Brain Injury Guidelines, its overall diagnostic performance hadn’t been evaluated in a pooled analysis until recently.
In July 2024, a systematic review and meta-analysis by Kannan et al. synthesized data from 13 studies involving 9,032 patients, making it the largest and most comprehensive assessment of BIG to date.[12] This meta analysis included 1,433 patients in the BIG 1 category, 2,136 patients in the BIG 2 category, and 3,100 patients in the BIG 3 category.[12]
Its goal was to predict two key outcomes:
- The need for neurosurgical intervention
- Radiologic deterioration
Key Findings [12]:
- BIG 1 demonstrated:
- Pooled sensitivity of 1.00 (100%) for predicting neurosurgical intervention
- Pooled sensitivity of 0.98 for predicting radiologic deterioration
- Very low specificity, reflecting a high number of false positives
- BIG 2 and 3 showed low sensitivity, reinforcing that the BIG framework is most reliable as a rule-out tool for low-risk patients rather than a rule-in tool for high-risk cases
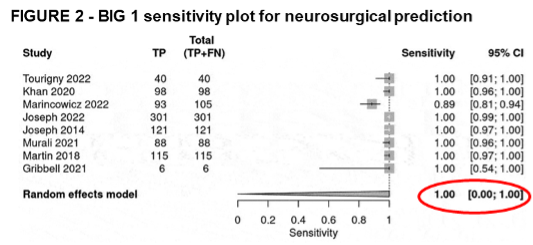
[12] S. Kannan, C. S. Gillespie, K. S. Lee, I. Phang, and C. J. McMahon, “Diagnostic utility of Brain Injury Guidelines (BIG): systematic review and meta-analysis for prediction of neurosurgical intervention in traumatic brain injury,” Brain Inj., vol. 38, no. 13, pp. 1093–1100, Nov. 2024, doi: 10.1080/02699052.2024.2375593.
These results confirm that BIG 1 is highly effective for identifying patients unlikely to need neurosurgical intervention, supporting its use in ED observation and discharge decisions in the right patient population. Conversely, the low specificity across all groups highlights the limitations of using BIG to predict who will deteriorate, especially in BIG 2 and 3 patients.[12]
The Modified Brain Injury Guidelines (mBIG)
The Modified Brain Injury Guidelines (mBIG), introduced in 2020, refine the original BIG criteria to enhance safety, reproducibility, and real-world applicability across centers.[5][6] Like BIG, mBIG uses three core elements for risk stratification: neurologic exam, anticoagulation/antiplatelet status, and CT head findings but it incorporates several important updates to improve consistency and clarity.[5][6]
Key Changes Introduced by mBIG: [5][6][13]
- Neurologic Exam: mBIG explicitly defines an abnormal exam as a GCS <13, the presence of focal neurologic deficits, or pupillary abnormalities. Unlike the original BIG, it also incorporates initial GCS into risk stratification.
- Intoxication: Defined as blood alcohol level >80 mg/dL, providing a clear and objective threshold that was absent from the original BIG criteria.
- Anticoagulation: Unlike the original BIG, mBIG includes DOACs, heparin, and enoxaparin. Aspirin alone is no longer considered high risk; therefore, patients taking only aspirin are not automatically classified as mBIG 3.
- Epidural Hematomas: All epidural hemorrhages are now classified as high risk (mBIG 3) given their unacceptably high risk of clinical deterioration.
- Radiologic Definitions: mBIG introduces clearer, measurable definitions for subarachnoid hemorrhage (e.g., based on sulci/hemisphere involvement) rather than subjective descriptors like “trace.”
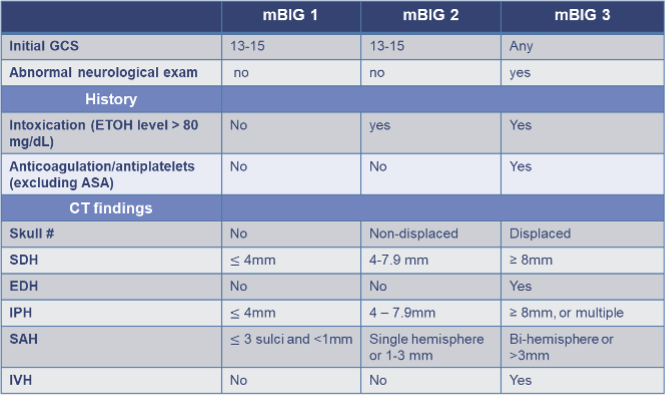
mBIG, modified brain injury guidelines; anticoagulation, DOACs, warfarin, heparin, enoxaparin; antiplatelets, Plavix; EDH, epidural hemorrhage; IVH, intraventricular hemorrhage; IPH, intraparenchymal hemorrhage; LOC, loss of consciousness; SAH, subarachnoid hemorrhage; SDH, subdural hemorrhage; skull #, skull fracture.
mBIG 1 – Low Risk
Criteria:
- GCS 13-15 or at baseline
- Normal neurologic exam
- No anticoagulation/antiplatelets (DOACs, warfarin, clopidogrel, etc.)
- No intoxication
- CT shows isolated and minimal injuries, such as:
- Subdural (SDH) or intraparenchymal hemorrhage (IPH) ≤4 mm
- Localized subarachnoid hemorrhage (SAH) involving ≤3 sulci and <1mm
- No skull fracture, intraventricular (IVH) or epidural hemorrhage (EDH)
Management:
- 6-hour ED observation with neurological vital checks q2h
- No repeat head CT
- No neurosurgical consultation
If stable, discharge home from ED. If the injury occurred more than 24 hours prior to presentation, observation is not required.
mBIG 2 – Moderate Risk
Criteria:
- GCS 13-15 or at baseline
- Normal neurologic exam
- May be intoxicated (EtOH level > 80 mg/dL)
- No anticoagulation/antiplatelets (DOACs, warfarin, clopidogrel, etc.)
- CT shows:
- Subdural (SDH) or intraparenchymal (IPH) 5–7.9 mm
- Subarachnoid hemorrhage (SAH) involving one hemisphere or 1-3mm
- Non-displaced skull fracture
- No intraventricular (IVH) or epidural hemorrhage (EDH), midline shift or herniation.
Management:
- Admit for 24–48 hours to a trauma service with neurological vital checks q2h ×6 hours, then q4h
- No routine repeat CT head
No neurosurgical consultation
mBIG 3 – High Risk
Includes any of the following:
- Abnormal neurologic exam (GCS <13, focal findings, abnormal pupils)
- intoxication (EtOH level > 80 mg/dL)
- Use of anticoagulants/antiplatelets (warfarin, DOACs, LMWH, clopidogrel)
- CT shows:
- Subdural (SDH) or intraparenchymal hemorrhage (IPH) >=8 mm
- Any intraventricular hemorrhage (IVH)
- Bi-hemisphere subarachnoid hemorrhage (SAH) of >3mm
- Any epidural hemorrhage
- Multiple concurrent hemorrhages, midline shift.
- Displaced skull fracture
Management:
- Admit to monitored setting (ICU/NACU)
- Neurosurgical consultation
- Repeat head CT
Built-in Safety Net
mBIG reinforces that all patients must have a GCS of 15 or be at their neurologic baseline prior to discharge. This ensures that only stable and reliable patients are discharged from hospital. [5][6][13]
If a patient experiences any neurologic deterioration, they are immediately escalated to the mBIG 3 pathway, which triggers:[13]
- a repeat head CT
- immediate neurosurgical consultation
- Formal hospital admission
This built-in escalation mechanism prioritizes patient safety and ensures timely intervention for those at risk of progression.[13]
Validation of the Modified Brain Injury Guidelines (mBIG)

Following the introduction of mBIG in 2020, researchers set out to determine whether the modified guidelines were not only theoretically sound but also safe and effective in real-world clinical practice. The most significant validation to date comes from a 2022 multicenter study by Khan et al., which conducted a retrospective review of prospectively collected data across three Level I trauma centers to assess the safety and impact of mBIG implementation. [6]
The study included 764 patients with mild traumatic brain injury (TBI) and intracranial hemorrhage (ICH), focusing on those classified as mBIG 1 and 2 patients, to compare outcomes before and after the implementation of the modified guidelines. [6]
Key Findings [6]:
- Resource use decreased significantly:
- Fewer repeat head CT scans
- Fewer neurosurgical consultations
- Shorter hospital and ICU stays
- Safety was maintained:
- No significant differences in clinical or radiographic progression, neurosurgical intervention, or mortality.
- Only 8 (1.6%) patients clinically worsened during observation (2 from mBIG 1, 6 from mBIG 2). Of these, only one mBIG 2 patient required surgery (craniotomy), and all others fully recovered.
Importantly, the study found no neurosurgical interventions were required in any mBIG 1 patients, reinforcing that this group can be safely observed and discharged without repeat imaging or neurosurgical input.[6]
These results support mBIG as a safe and effective tool for managing well-appearing patients with small intracranial bleeds. In particular, mBIG 1 and 2 patients can be safely managed using this protocol, leading to improvements in resource utilization without compromising patient outcomes or safety.
Limitations of BIG and mBIG
While the Brain Injury Guidelines (BIG) and Modified Brain Injury Guidelines (mBIG) represent important steps toward more standardized and efficient care for mild TBI, several limitations remain.
-
- Methodology limitation: Retrospective study design
Most studies evaluating BIG and mBIG are retrospective in design, which introduces the potential for selection bias. While early prospective validation studies have shown promising results, particularly for BIG, larger multicenter prospective trials are still needed, especially for mBIG. A full systematic review of mBIG is also pending, making it difficult to draw broad conclusions about its generalizability across diverse patient populations and practice settings. - Applicability to high-resource settings
Both guidelines were developed and primarily validated in high-resource, Level I trauma centers with consistent and robust neurosurgical support. Their safety and effectiveness in rural or community settings, where access to advanced imaging, continuous monitoring, and specialist consultation may be limited, remain less well studied. - Underrepresentation of geriatric and cognitively impaired patients
Older adults and patients with baseline cognitive impairment (e.g., dementia, delirium) are underrepresented in the validation studies. These populations may present atypically and are at higher risk of complications, limiting the generalizability of the guidelines. - Lack of longitudinal outcome tracking
The guidelines focus on acute disposition decisions but do not account for long-term outcomes and recovery trajectories, such as cognitive recovery, post-concussion symptoms and management, or functional decline. This limits their utility in comprehensive care planning and follow-up. - Omission of some high-risk clinical variables
Neither BIG nor mBIG account for comorbidities such as frailty or dementia. The guidelines assume clinicians can interpret GCS and neurologic findings relative to baseline but do not provide structured guidance on managing patients with chronic cognitive impairment. This could raise the risk of premature discharge in vulnerable patient groups.
- Methodology limitation: Retrospective study design
Implications of the Brain Injury Guidelines:
Despite some limitations, the implementation of standardized protocols like mBIG in emergency departments has the potential to deliver meaningful, system-wide improvements in the management of traumatic brain injuries.
As demonstrated in the growing body of evidence, mBIG has been associated with fewer repeat CT scans, fewer neurosurgical consultations, and reduced hospital admissions. For emergency departments, this translates to shorter lengths of stay, improved patient flow, and potentially significant cost savings.
Importantly, both BIG and mBIG consistently identify low-risk TBI patients who can be safely managed without escalation of care, repeat imaging, or neurosurgical consultation. For mBIG 1 and 2 patients, the risk of clinical deterioration is exceedingly low. This provides clinicians with a validated framework to support discharge decisions, particularly in patients who are well-appearing, neurologically stable, and meet clear criteria.
The adoption of mBIG also offers broader system-level benefits. By standardizing care, the guidelines help reduce practice variation across providers, support better interdisciplinary alignment, and promote more efficient use of monitored beds and imaging resources.
Finally, in rural or community settings, mBIG may help reduce unnecessary transfers for low-risk patients, keeping them closer to home and preserving tertiary care resources for higher-acuity cases.
Implementation Challenges of mBIG
While mBIG offers clear clinical promise, real-world adoption presents several challenges that must be addressed for successful implementation.
- Need for coordinated protocols
Without a standardized, institution-wide protocol and shared understanding across services, mBIG risks creating variability, confusion, and even conflict rather than clarity. Effective use would require alignment between Emergency Medicine, Radiology, Neurosurgery, Trauma, and inpatient teams. Furthermore, the success of mBIG depends on consistent and precise radiology reporting. Variability in describing small bleeds or skull fractures can lead to inconsistent classification and management. - Cultural barriers
Discharging patients with visible intracranial hemorrhage on CT, without neurosurgical input, may still feel uncomfortable for many clinicians, even when the patient meets low-risk criteria. Shifting this mindset may require time, strong interdisciplinary collaboration, and active support from Neurosurgery. - Operational constraints
Limited staffing, monitoring capacity, and ED space can make it difficult to reliably carry out the observation periods and neurological vital checks required by mBIG. In smaller or busier departments, these logistics may pose significant barriers. - Follow-up limitations
Safe discharge of low-risk patients from the emergency department is supported by reliable follow-up and clear return instructions. In settings where outpatient follow-up is difficult to arrange, clinicians may be hesitant to apply mBIG recommendations, which can ultimately hinder its adoption in practice.
Conclusions:
While no clinical guideline is perfect, the Modified Brain Injury Guidelines (mBIG), particularly mBIG 1, offer a promising tool for managing a common and often resource-intensive emergency department presentation. The available evidence demonstrates that mBIG 1 has high sensitivity for predicting the need for neurosurgical intervention and supports the safe discharge of well-appearing patients with small bleeds, without requiring repeat CT imaging or neurosurgical consultation.
This represents a practical and evidence-informed shift in care that could significantly reduce emergency department length of stay, lower the number of unnecessary CT scans, alleviate pressure on monitored beds, decrease the consult burden on neurosurgery, and ultimately improve both patient experience and system efficiency.
Beyond its practical application, mBIG also highlights the evolving role of emergency medicine in the care of traumatic brain injury. The development of BIG and mBIG, as well as new emerging tools such as QueBIC, reflect growing momentum toward more standardized, evidence-based TBI management.
Emergency physicians are uniquely positioned to lead this shift. Familiarity with these guidelines has the potential to enable more consistent care, facilitate collaboration across specialties, and support patient-centered implementation strategies. The approach to traumatic brain injury is evolving. The Modified Brain Injury Guidelines bring us closer to a safer, more efficient, and more unified approach. BIG and mBIG are gaining traction and may soon represent a meaningful and lasting practice shift in how emergency medicine manages low-risk TBI.
References
- H. A. of Canada, “Traumatic Brain Injuries – Canada.ca.” Accessed: Apr. 21, 2025. [Online]. Available: https://health-infobase.canada.ca/brain-injuries/
[2] B. Joseph et al., “The BIG (brain injury guidelines) project: defining the management of traumatic brain injury by acute care surgeons,” J. Trauma Acute Care Surg., vol. 76, no. 4, pp. 965–969, Apr. 2014, doi: 10.1097/TA.0000000000000161.
[3] B. Joseph et al., “Validating the Brain Injury Guidelines: Results of an American Association for the Surgery of Trauma prospective multi-institutional trial,” J. Trauma Acute Care Surg., vol. 93, no. 2, pp. 157–165, Aug. 2022, doi: 10.1097/TA.0000000000003554.
[4] “Abstract 5: Validating the Brain Injury Guidelines: A Multi-Institutional Trial,” EM:RAP. Accessed: Apr. 15, 2025. [Online]. Available: https://www.emrap.org/episode/ema2023april/abstract5
[5] A. D. Khan et al., “Multicenter assessment of the Brain Injury Guidelines and a proposal of guideline modifications,” Trauma Surg. Acute Care Open, vol. 5, no. 1, p. e000483, 2020, doi: 10.1136/tsaco-2020-000483.
[6] A. D. Khan et al., “A multicenter validation of the modified brain injury guidelines: Are they safe and effective?,” J. Trauma Acute Care Surg., vol. 93, no. 1, pp. 106–112, Jul. 2022, doi: 10.1097/TA.0000000000003633.
[7] “EM:RAP – Neurocritical Care Mailbag: TBI Management – MVCs and TBIs,” EM:RAP. Accessed: Apr. 15, 2025. [Online]. Available: https://www.emrap.org/episode/mvcsandtbis/neurocriticalca8
[8] “Abstract 9: Low-Risk TBI in Rural Level IV Trauma Center: ID and Management,” EM:RAP. Accessed: Apr. 15, 2025. [Online]. Available: https://www.emrap.org/episode/ema2024april/abstract9lowris
[9] “Abstract 7: Brain Injury Guideline: Observation in Small Intracranial Hemorrhage,” EM:RAP. Accessed: Apr. 15, 2025. [Online]. Available: https://www.emrap.org/episode/ema2024march/abstract7braini
[10] B. Joseph et al., “Prospective validation of the brain injury guidelines: managing traumatic brain injury without neurosurgical consultation,” J. Trauma Acute Care Surg., vol. 77, no. 6, pp. 984–988, Dec. 2014, doi: 10.1097/TA.0000000000000428.
[11] K. de Wit, Z. Merali, Y. K. Kagoma, and É. Mercier, “Incidence of intracranial bleeding in seniors presenting to the emergency department after a fall: A systematic review,” Injury, vol. 51, no. 2, pp. 157–163, Feb. 2020, doi: 10.1016/j.injury.2019.12.036.
[12] S. Kannan, C. S. Gillespie, K. S. Lee, I. Phang, and C. J. McMahon, “Diagnostic utility of Brain Injury Guidelines (BIG): systematic review and meta-analysis for prediction of neurosurgical intervention in traumatic brain injury,” Brain Inj., vol. 38, no. 13, pp. 1093–1100, Nov. 2024, doi: 10.1080/02699052.2024.2375593.
[13] A. B. Kay, S. A. Malone, J. R. Bledsoe, S. Majercik, and D. S. Morris, “First steps toward a BIG change: A pilot study to implement the Brain Injury Guidelines across a 24-hospital system,” Am. J. Surg., vol. 226, no. 6, pp. 845–850, Dec. 2023, doi: 10.1016/j.amjsurg.2023.07.002.
[14] J. S. Lee et al., “To observe or not to observe: Evaluation of the modified brain injury guideline management of small volume intracranial hemorrhage,” Am. J. Surg., vol. 226, no. 6, pp. 808–812, Dec. 2023, doi: 10.1016/j.amjsurg.2023.06.023.
[15] G. E. Martin et al., “Safety and efficacy of brain injury guidelines at a Level III trauma center,” J. Trauma Acute Care Surg., vol. 84, no. 3, pp. 483–489, Mar. 2018, doi: 10.1097/TA.0000000000001767.
[16] J. Morgenstern, “The Brain Injury Guidelines: Can we avoid talking to neurosurgeons?,” First10EM. Accessed: Apr. 15, 2025. [Online]. Available: https://first10em.com/the-brain-injury-guidelines-can-we-avoid-talking-to-neurosurgeons/
[17] J. F. Sweeney, K. Nath, N. C. Field, T. A. Harland, and M. A. Adamo, “Utilization of the Modified Brain Injury Guidelines by Neurosurgeons to Improve Traumatic Brain Injury Patient Throughput at a Level I Trauma Center: A Retrospective Observational Study,” World Neurosurg., vol. 191, pp. e227–e234, Nov. 2024, doi: 10.1016/j.wneu.2024.08.098.
[18] R. V. Nene et al., “Identification and management of low-risk isolated traumatic brain injury patients initially treated at a rural level IV trauma center,” Am. J. Emerg. Med., vol. 78, pp. 127–131, Apr. 2024, doi: 10.1016/j.ajem.2024.01.014.
[19] D. Roberge Bouchard et al., “Outcomes of Traumatic Brain Injury Patients Managed in a Non-Intensive Care Unit Setting,” J. Surg. Res., vol. 302, pp. 679–684, Oct. 2024, doi: 10.1016/j.jss.2024.07.113.
[20] J.-N. Tourigny et al., “Derivation of the Quebec Brain Injury Categories for complicated mild traumatic brain injuries,” Jul. 2024, doi: 10.3171/2024.4.JNS24183.
[21] Dr. Marcel Émond, Université Laval, “Minor Complex TBI: the QueBIC?”, oral presentation, 2024. Shared slides used with permission.


