GLP-1 receptor agonists have moved from niche diabetes therapies to some of the most commonly encountered medications in modern clinical practice. Drugs like semaglutide and tirzepatide are reshaping the management of obesity and cardiometabolic disease, with millions of patients now using them for sustained weight loss and risk reduction. In Canada, agents such as semaglutide (Ozempic®, Wegovy®), liraglutide (Victoza®, Saxenda®), and tirzepatide (Mounjaro®, Zepbound®) are rapidly becoming part of the everyday medication list.
For emergency physicians, this shift matters. We are now seeing the downstream effects of these therapies in real time. The same mechanisms that make GLP-1 receptor agonists effective, particularly delayed gastric emptying and altered gut motility, also create a predictable set of adverse effects and clinical presentations. From persistent gastrointestinal symptoms to aspiration risk and functional obstruction, these drugs are changing what walks through the ED doors.
This post breaks down what you need to know: how these medications work, what the key evidence shows, and most importantly, how to recognize and manage their complications in the emergency department.
Why this Matters: the Rise of GLP-1 Therapy
Obesity is a rapidly growing public health issue in Canada and a major driver of emergency department presentations. Recent national data show that obesity prevalence among Canadian adults increased from approximately 25% pre-pandemic to 33% between 2022–2024. [1,2]

[1,2]
When overweight categories are included, the burden becomes even more striking: since 2022, approximately 40% of Canadian men and 30% of women are classified as overweight, meaning that nearly three in four men and two in three women fall within BMI ranges associated with increased health risk. [1,2]

This has important clinical implications. Obesity is now recognized as a chronic, relapsing disease associated with increased risk of more than 200 conditions, including [3]:
- Type 2 diabetes
- Cardiovascular disease
- Obstructive sleep apnea
- Certain cancers
- Increased overall mortality
Beyond individual patient risk, the system-level burden is substantial. The estimated annual cost of obesity in Canada—including hospitalizations, medications, long-term care, and premature mortality—is approximately $27 billion per year. [4]
Against this backdrop, the demand for effective therapies has grown significantly. GLP-1 receptor agonists have emerged as a major advancement, offering clinically meaningful and sustained weight loss alongside improvements in glycemic control and cardiovascular outcomes. Their effectiveness has driven rapid uptake, with recent estimates suggesting that approximately 1.4 million Canadians are currently using a GLP-1 agent. [15]
As use continues to expand across both diabetes and obesity indications, emergency physicians are increasingly encountering patients on these therapies and their associated complications in the ED.
Mechanism: Why these drugs work (and why they cause problems)
Glucagon-like peptide-1 receptor agonists (GLP-1 RAs) were initially approved in 2005 for the treatment of type 2 diabetes. They were made to mimic endogenous GLP-1. [39, 40]
Endogenous GLP-1 is released from L-cells in the small intestine in response to food intake. It has a very short half-life, approximately 1–2 minutes, due to rapid degradation by dipeptidyl peptidase-4 (DPP-4). Despite this, it plays an important role in postprandial glucose regulation and energy balance.
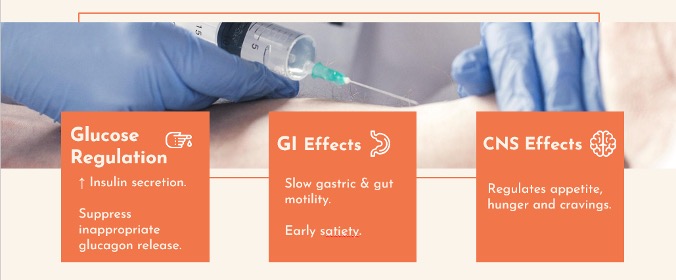
GLP-1 exerts several key physiologic effects:
- Increases glucose-dependent insulin secretion
- Suppresses inappropriate glucagon release
- Delays gastric emptying
- Promotes satiety and reduces appetite
Pharmacologic GLP-1 receptor agonists, including semaglutide, liraglutide, and tirzepatide, are engineered to resist DPP-4 degradation, allowing them to persist for hours to days. This prolongs and amplifies the physiologic effects of endogenous GLP-1.
While these agents were initially developed for glycemic control, their impact extends beyond glucose regulation. By simultaneously targeting appetite regulation, satiety, gastric motility, and central reward pathways, they produce clinically meaningful and sustained weight loss.
Among these effects, delayed gastric emptying is particularly important. It contributes to early satiety and weight loss, but also underlies many of the adverse effects and emergency department presentations associated with these medications.
GLP-1 receptors are widely distributed throughout the body including the pancreas, gastrointestinal tract, and central nervous system, which helps explain both their broad therapeutic benefits and their diverse clinical effects. [7]
Effectiveness and Key Trials:
The rapid uptake of GLP-1 receptor agonists is largely driven by their consistent and clinically meaningful benefits across multiple large randomized trials.
Key trials demonstrate:
- SUSTAIN-1: HbA1c reduction ~1.45-1.55% with modest weight loss (~3.73–4.53 kg). [8]
- SURPASS-2: Tirzepatide (5, 10, 15 mg) reduced HbA1c by 2.01%–2.30%, compared to 1.86% with semaglutide (1 mg). Tirzepatide produced greater weight reduction than semaglutide, with effects increasing in a dose-dependent manner. [9]
- STEP-1: In patients with obesity without diabetes, semaglutide resulted in ~15% mean weight loss over 68 weeks. [10]
- SURMOUNT-1: Tirzepatide for the treatment of obesity produced ~20–22% weight loss over 72 weeks, among the largest effects seen in pharmacologic obesity treatment. [11]
- SELECT: SELECT was a large randomized, controlled trial that enrolled over 17,000 adults with established cardiovascular disease, an elevated BMI, but no diabetes. It showed that Semaglutide reduced major adverse cardiovascular events by 20% in patients with obesity without diabetes. [12, 13]
Taken together, these findings establish GLP-1 receptor agonists as among the most effective medical therapies for both diabetes and obesity, driving their rapid and widespread use. This also shifted GLP-1 therapy from being purely a metabolic medication to a cardiovascular disease–modifying therapy, even in patients without diabetes.
ED Relevant Adverse Effects
As use continues to expand, the same physiologic effects that drive the benefits of GLP-1 receptor agonists also underlie their adverse effects and ED presentations. Many of these complications stem from delayed gastric emptying and reduced gut motility. While most are mild and self-limited, a subset are clinically significant and relevant to emergency medicine. [37]
1. Gastrointestinal Effects (most common)
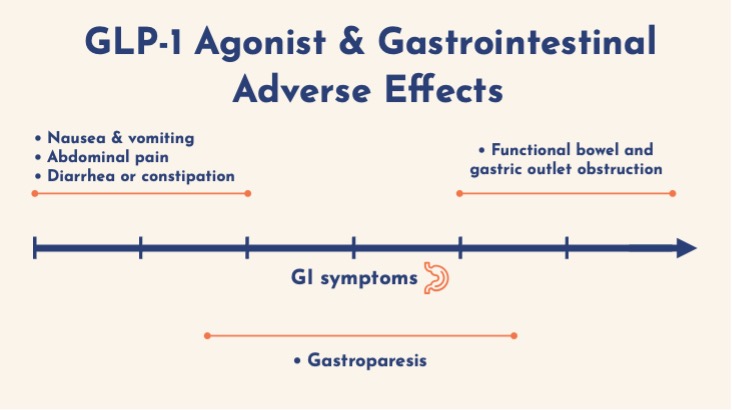
GI symptoms are the most frequent adverse effects and occur in a dose-dependent manner, typically within 48 hours of initiation or dose escalation. [16] Reported rates from trials include:
- Nausea: ~44%
- Vomiting: ~25%
- Diarrhea: ~31%
- Constipation: ~23%
- Dyspepsia: ~10%
These effects reflect slowed gastric motility and are usually mild, but can lead to clinical implications:
- Dehydration
- Electrolyte abnormalities
- ED presentations for poor oral intake.
More severe spectrum:
- Gastroparesis (HR ~1.6 vs other agents) [16]
- Functional obstruction (gastric or small bowel dilation without transition point)
ED management:
- Behavioural strategies & dietary strategies:
- Smaller meals, Eating slowly, Avoiding high-fat foods, alcohol & carbonation, High fiber diet.
- IV fluids, electrolyte correction
- Antiemetics
- Consider holding the GLP-1 agent
- NG decompression if severe
2. Airway and Aspiration Risk
Delayed gastric emptying has important implications for airway management. [17, 18, 19, 22]
Key evidence: [20, 21]
- A meta-analysis of >84,000 patients undergoing endoscopy showed:
-
- Increased retained gastric contents (OR ~5.56)
- No significant increase in aspiration events
Clinical implication:
Patients may have a “full stomach” despite appropriate fasting.
In the ED:
- Fasting history is unreliable
- Treat as higher aspiration risk especially if symptomatic
- Anticipate vomiting and difficult visualization
Practical considerations:
- Optimize positioning (ramped, head elevated, or slight reverse Trendelenburg)
- Use RSI when appropriate
- Minimize bag-mask ventilation
- Ensure suction is immediately available and functioning.
- Clearly communicate aspiration risk to the team to align preparation and roles
There are no formal ED-specific guidelines, but EM and anesthesia literature consistently emphasize increased vigilance rather than major practice change. [17, 18, 19, 22]
3. Biliary Disease (Common and Clinically Significant)
GLP-1 receptor agonists are associated with increased risk of gallbladder and biliary disease.
Evidence:
The strongest data come from a large meta-analysis involving over 100,000 patients. When compared with placebo or other therapies, GLP-1 agonists were associated with an increased risk of biliary disease [23]:
- Overall biliary disease: RR 1.37(95% CI 1.23–1.52)
- Cholelithiasis: RR 1.27 (95% CI 1.10–1.47)
- Cholecystitis: RR 1.36 (95% CI 1.14–1.62)
Mechanisms:
- Reduced gallbladder motility
- Rapid weight loss → increased cholesterol saturation and gallstone formation.
Clinical pearls:
- Risk highest within the first 6 months of therapy
- Presentation similar to typical biliary pathology
ED management:
Standard approach to biliary disease (analgesia, antibiotics if indicated, surgical consultation).
4. Pancreatitis
Initial concerns arose from case reports and post-marketing data. However:
- Larger meta-analyses show no significant increase in risk after adjusting for confounders [25]
- Incidence appears similar to other diabetes therapies [25]
Much of the observed signal likely reflects:
- Baseline risk (obesity, T2DM)
- Secondary gallstone disease
In summary, pancreatitis in GLP-1 users is a rare and possible complication, but current evidence does not support a substantial increase in risk directly related to GLP-1 agents. That being said, vigilance is warranted particularly in patients with biliary disease, prior pancreatitis, or rapid recent weight loss. Cases of pancreatitis can be managed according to standard ED practice and logically, the GLP-1 agent should be stopped. [26, 28]
5. Metabolic: Euglycemic DKA
The risk of euglycemic DKA is very uncommon, but it’s a clinically important and real complication to be aware of in patients using GLP-1 agonists. [29, 30, 31]
Risk factors:
- Diabetes (especially in combination with SGLT2 inhibitors use)
- Reduced oral intake and severe GI symptoms
- Dehydration
Presentation:
- Nausea, vomiting, abdominal pain
- Anion gap metabolic acidosis
- Elevated ketones
- Normal or mildly elevated glucose
Key point:
EDKA has not been observed in non-diabetic patients in large cohort data. [31]
The takeaway is vigilance: any GLP-1 user with diabetes especially on combined therapy with SGLT2 inhibitors, who presents with persistent vomiting or reduced oral intake deserves consideration for EDKA. Early diagnosis and standard DKA management are key to preventing serious outcomes.
6. Renal – Acute Kidney Injury
Acute kidney injury is an uncommon but recognized complication of GLP-1 receptor agonists. [33, 34, 35, 36]
Mechanism:
- Typically pre-renal, occurring in the setting of volume depletion from GI losses (nausea, vomiting, diarrhea, reduced oral intake)
- More frequently seen at higher, obesity-level doses
- Although generally reno-protective long term, post-marketing data describe acute rises in creatinine following days to weeks of significant GI symptoms
Risk factors:
- Underlying chronic kidney disease
- Concomitant use of ACE inhibitors, ARBs, diuretics, or SGLT2 inhibitors
ED Management:
- IV fluids
- Hold GLP-1 and contributing medications
- Usually reversible with supportive care
Future Directions
GLP-1 receptor agonists are rapidly evolving beyond their original indications in diabetes and obesity. Ongoing research is exploring their potential role across a range of conditions, including cardiovascular disease prevention, metabolic liver disease, and addictions medicine. Early data are promising, but these applications remain under active investigation.
At the same time, the therapeutic landscape is expanding. Newer agents including dual and triple incretin agonists are demonstrating even greater metabolic effects in clinical trials. Oral formulations are also emerging, which may improve accessibility and further increase uptake.
As use continues to grow, emergency physicians will encounter these medications more frequently across a broader patient population. Understanding their mechanisms, benefits, and associated risks will remain essential as their role in clinical medicine continues to expand.
Key Points:
- GI symptoms are the most common adverse effects, ranging from expected nausea and vomiting to gastroparesis and, rarely, functional obstruction
- Delayed gastric emptying has important airway implications and a cautious approach to airway management is warranted
- Biliary disease is one of the most clinically significant complications, particularly early in treatment and with rapid weight loss
- Pancreatitis is rare, and current evidence does not support a strong causal relationship
- EDKA and AKI are uncommon but important to recognize in the right context
Conclusions:
GLP-1 receptor agonists have transformed the management of diabetes and obesity, with clear benefits in weight loss, glycemic control, and cardiovascular outcomes. As their use continues to expand, emergency physicians are increasingly encountering their downstream effects.
The same physiology that drives these benefits, particularly delayed gastric emptying, also explains many of the complications seen in the ED. Recognizing these patterns is key to identifying at-risk patients, anticipating complications, and guiding safe management.
As more patients are treated with these therapies, it is important to understand how they alter clinical presentations and risk for adverse effects. This will support more informed, anticipatory, and patient-centered care in the emergency department.
References
1. C. Government of Canada, “The Daily — The prevalence of overweight and obesity is on the rise in Canada: New results from the Canadian Health Measures Survey, 2022 to 2024.” Accessed: Nov. 16, 2025. [Online]. Available: https://www150.statcan.gc.ca/n1/daily-quotidien/251002/dq251002b-eng.htm
[2] “Press release: New Pediatric Obesity Clinical Practice Guideline | Obesity Canada.” Accessed: Nov. 16, 2025. [Online]. Available: https://obesitycanada.ca/news/2025-pediatric-obesity-cpg/?utm
[3] P. H. A. of Canada, “Obesity Statistics in Canada: Report.” Accessed: Nov. 16, 2025. [Online]. Available: https://www.canada.ca/en/public-health/services/publications/healthy-living/obesity-statistics-canada.html
[4] “Cost of Inaction on Obesity,” Obesity Canada. Accessed: Nov. 23, 2025. [Online]. Available: https://obesitycanada.ca/understanding-obesity/cost-of-inaction/
[5] P. Aparicio-Martinez, A.-J. Perea-Moreno, M. P. Martinez-Jimenez, M. D. Redel-Macías, C. Pagliari, and M. Vaquero-Abellan, “Social Media, Thin-Ideal, Body Dissatisfaction and Disordered Eating Attitudes: An Exploratory Analysis,” Int. J. Environ. Res. Public. Health, vol. 16, no. 21, p. 4177, Nov. 2019, doi: 10.3390/ijerph16214177.
[6] E. Hutchinson, “12 Celebs Who Totally Transformed After Taking Ozempic Or Other GLP-1’s,” Women. Accessed: Nov. 16, 2025. [Online]. Available: https://www.women.com/1857718/celebs-totally-transformed-taking-ozempic-other-glp-1/
[7] “Pharmacology Rounds: GLP-1 Receptor Agonists Part 1,” EM:RAP. Accessed: Nov. 19, 2025. [Online]. Available: https://www.emrap.org/episode/ucmax2025may/pharmacologyrou4
[8] C. Sorli et al., “Efficacy and safety of once-weekly semaglutide monotherapy versus placebo in patients with type 2 diabetes (SUSTAIN 1): a double-blind, randomised, placebo-controlled, parallel-group, multinational, multicentre phase 3a trial,” Lancet Diabetes Endocrinol., vol. 5, no. 4, pp. 251–260, Apr. 2017, doi: 10.1016/S2213-8587(17)30013-X.
[9] J. P. Frías et al., “Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes,” N. Engl. J. Med., vol. 385, no. 6, pp. 503–515, Aug. 2021, doi: 10.1056/NEJMoa2107519.
[10] J. P. H. Wilding et al., “Once-Weekly Semaglutide in Adults with Overweight or Obesity,” N. Engl. J. Med., vol. 384, no. 11, pp. 989–1002, Mar. 2021, doi: 10.1056/NEJMoa2032183.
[11] “Tirzepatide Once Weekly for the Treatment of Obesity | New England Journal of Medicine.” Accessed: Nov. 19, 2025. [Online]. Available: https://www.nejm.org/doi/full/10.1056/NEJMoa2206038?utm_source=chatgpt.com
[12] A. M. Lincoff et al., “Semaglutide and Cardiovascular Outcomes in Obesity without Diabetes,” N. Engl. J. Med., vol. 389, no. 24, pp. 2221–2232, Dec. 2023, doi: 10.1056/NEJMoa2307563.
[13] D. H. Ryan et al., “Long-term weight loss effects of semaglutide in obesity without diabetes in the SELECT trial,” Nat. Med., vol. 30, no. 7, pp. 2049–2057, Jul. 2024, doi: 10.1038/s41591-024-02996-7.
[14] “Nearly 12 Percent of Americans Have Used GLP-1 Weight Loss Drugs; Medications Are Most Used by Women Aged 50 to 64.” Accessed: Nov. 23, 2025. [Online]. Available: https://www.rand.org/news/press/2025/08/nearly-12-percent-of-americans-have-used-glp-1-weight.html
[15] S. M. Arif Hafsa, “Roughly 1M Canadians use Ozempic, similar drugs for weight loss, diabetes: study,” CTVNews. Accessed: Nov. 19, 2025. [Online]. Available: https://www.ctvnews.ca/atlantic/article/roughly-1m-canadians-use-ozempic-similar-drugs-for-weight-loss-diabetes-study/
[16] C. Niu et al., “Gastrointestinal and Hepatobiliary Safety of Glucagon-Like Peptide-1 Receptor Agonists in Patients With Type 2 Diabetes,” Am. J. Gastroenterol., Sep. 2025, doi: 10.14309/ajg.0000000000003760.
[17] G. B. Mizubuti, A. M.-H. Ho, and L. M. da Silva, “Implications of GLP-1 agonist use on airway management,” Am. J. Emerg. Med., vol. 82, pp. 190–191, Aug. 2024, doi: 10.1016/j.ajem.2024.05.018.
[18] W. Dixon, “Use of Glucagon-Like Peptide-1 Agonists and Increased Risk of Procedural Sedation and Endotracheal Intubation in the Emergency Department,” Ann. Emerg. Med., vol. 84, no. 2, pp. 226–227, Aug. 2024, doi: 10.1016/j.annemergmed.2024.03.007.
[19] M. Sherwin, J. Hamburger, D. Katz, and S. DeMaria, “Influence of semaglutide use on the presence of residual gastric solids on gastric ultrasound: a prospective observational study in volunteers without obesity recently started on semaglutide,” Can. J. Anaesth. J. Can. Anesth., vol. 70, no. 8, pp. 1300–1306, Aug. 2023, doi: 10.1007/s12630-023-02549-5.
[20] S. Q. Silveira et al., “Relationship between perioperative semaglutide use and residual gastric content: A retrospective analysis of patients undergoing elective upper endoscopy,” J. Clin. Anesth., vol. 87, p. 111091, Aug. 2023, doi: 10.1016/j.jclinane.2023.111091.
[21] A. Facciorusso et al., “Effects of Glucagon-Like Peptide-1 Receptor Agonists on Upper Gastrointestinal Endoscopy: A Meta-Analysis,” Clin. Gastroenterol. Hepatol., vol. 23, no. 5, pp. 715-725.e3, Apr. 2025, doi: 10.1016/j.cgh.2024.07.021.
[22] “American Society of Anesthesiologists Consensus-Based Guidance on Preoperative Management of Patients (Adults and Children) on Glucagon-Like Peptide-1 (GLP-1) Receptor Agonists.” Accessed: Nov. 20, 2025. [Online]. Available: https://www.asahq.org/about-asa/newsroom/news-releases/2023/06/american-society-of-anesthesiologists-consensus-based-guidance-on-preoperative
[23] L. He et al., “Association of Glucagon-Like Peptide-1 Receptor Agonist Use With Risk of Gallbladder and Biliary Diseases: A Systematic Review and Meta-analysis of Randomized Clinical Trials,” JAMA Intern. Med., vol. 182, no. 5, pp. 513–519, May 2022, doi: 10.1001/jamainternmed.2022.0338.
[24] D. Woronow, C. Chamberlain, A. Niak, M. Avigan, M. Houstoun, and C. Kortepeter, “Acute Cholecystitis Associated With the Use of Glucagon-Like Peptide-1 Receptor Agonists Reported to the US Food and Drug Administration,” JAMA Intern. Med., vol. 182, no. 10, pp. 1104–1106, Oct. 2022, doi: 10.1001/jamainternmed.2022.3810.
[25] J. Wen et al., “Evaluating the Rates of Pancreatitis and Pancreatic Cancer Among GLP-1 Receptor Agonists: A Systematic Review and Meta-Analysis of Randomised Controlled Trials,” Endocrinol. Diabetes Metab., vol. 8, no. 5, p. e70113, Sep. 2025, doi: 10.1002/edm2.70113.
[26] A. E. Mehta, L. D. Lomeli, and K. M. Pantalone, “Glucagon-like peptide-1 receptor agonists and pancreatitis: A reconcilable divorce,” Cleve. Clin. J. Med., vol. 92, no. 8, pp. 483–489, Aug. 2025, doi: 10.3949/ccjm.92a.24113.
[27] “ACG 2022 Annual Meeting.” Accessed: Nov. 21, 2025. [Online]. Available: https://www.eventscribe.net/2022/ACG2022/fsPopup.asp?PresentationID=1140511&mode=presinfo
[28] L. Li et al., “Incretin treatment and risk of pancreatitis in patients with type 2 diabetes mellitus: systematic review and meta-analysis of randomised and non-randomised studies,” BMJ, vol. 348, p. g2366, Apr. 2014, doi: 10.1136/bmj.g2366.
[29] J. Zhang, Y. Ma, Q. Zu, X. Wang, and Y. Zhang, “GLP-1 receptor agonist-induced diabetic ketoacidosis: A case report,” Medicine (Baltimore), vol. 103, no. 39, p. e39799, Sep. 2024, doi: 10.1097/MD.0000000000039799.
[30] Z. Yang et al., “The association between GLP-1 receptor agonist and diabetic ketoacidosis in the FDA adverse event reporting system,” Nutr. Metab. Cardiovasc. Dis. NMCD, vol. 32, no. 2, pp. 504–510, Feb. 2022, doi: 10.1016/j.numecd.2021.10.003.
[31] H. A. Thaibah, O. J. F. Banji, D. Banji, and T. M. Alshammari, “Diabetic Ketoacidosis and the Use of New Hypoglycemic Groups: Real-World Evidence Utilizing the Food and Drug Administration Adverse Event Reporting System,” Pharm. Basel Switz., vol. 18, no. 2, p. 214, Feb. 2025, doi: 10.3390/ph18020214.
[32] Y.-N. Huang et al., “Long-term safety and efficacy of glucagon-like peptide-1 receptor agonists in individuals with obesity and without type 2 diabetes: A global retrospective cohort study,” Diabetes Obes. Metab., vol. 26, no. 11, pp. 5222–5232, Nov. 2024, doi: 10.1111/dom.15869.
[33] A. Gandhi, N. Bhatt, and A. Parhizgar, “Comparative Renal Safety of Tirzepatide and Semaglutide: An FDA Adverse Event Reporting System (FAERS)—Disproportionality Study,” J. Clin. Med., vol. 14, no. 21, p. 7678, Oct. 2025, doi: 10.3390/jcm14217678.
[34] T. D. Filippatos, T. V. Panagiotopoulou, and M. S. Elisaf, “Adverse Effects of GLP-1 Receptor Agonists,” Rev. Diabet. Stud. RDS, vol. 11, no. 3, pp. 202–230, 2014, doi: 10.1900/RDS.2014.11.202.
[35] D. J. Leehey, M. A. Rahman, E. Borys, M. M. Picken, and C. E. Clise, “Acute Kidney Injury Associated With Semaglutide,” Kidney Med., vol. 3, no. 2, pp. 282–285, Jan. 2021, doi: 10.1016/j.xkme.2020.10.008.
[36] “Semaglutide-associated kidney injury | Clinical Kidney Journal | Oxford Academic.” Accessed: Nov. 20, 2025. [Online]. Available: https://academic.oup.com/ckj/article/17/9/sfae250/7733090?utm_source=chatgpt.com&login=false
[37] B. Long, J. Pelletier, A. Koyfman, and R. E. Bridwell, “GLP-1 agonists: A review for emergency clinicians,” Am. J. Emerg. Med., vol. 78, pp. 89–94, Apr. 2024, doi: 10.1016/j.ajem.2024.01.010.
[38] D. Mozaffarian et al., “Nutritional priorities to support GLP-1 therapy for obesity: a joint Advisory from the American College of Lifestyle Medicine, the American Society for Nutrition, the Obesity Medicine Association, and The Obesity Society,” Am. J. Clin. Nutr., vol. 122, no. 1, pp. 344–367, Jul. 2025, doi: 10.1016/j.ajcnut.2025.04.023.
[39] “September Mailbag: Semaglutide for Weight Loss,” EM:RAP. Accessed: Nov. 05, 2025. [Online]. Available: https://www.emrap.org/episode/emrap20238/september1
[40] “Pharmacology Rounds: GLP-1 Receptor Agonists Part 2,” EM:RAP. Accessed: Nov. 20, 2025. [Online]. Available: https://www.emrap.org/episode/ucmax2025may/pharmacologyrou5
[41] R. Pradhan, F. Montastruc, V. Rousseau, E. Patorno, and L. Azoulay, “Exendin-based glucagon-like peptide-1 receptor agonists and anaphylactic reactions: a pharmacovigilance analysis,” Lancet Diabetes Endocrinol., vol. 8, no. 1, pp. 13–14, Jan. 2020, doi: 10.1016/S2213-8587(19)30382-1.
[42] F. Celletti, J. Farrar, and L. De Regil, “World Health Organization Guideline on the Use and Indications of Glucagon-Like Peptide-1 Therapies for the Treatment of Obesity in Adults,” JAMA, Dec. 2025, doi: 10.1001/jama.2025.24288.
[44] The Thin Line. Accessed: Dec. 03, 2025. [Online]. Available: https://www.audible.ca/pd/The-Thin-Line-Audiobook/B0DQ94G2GH (Written by Scaachi Koul)



Thanks Camille – I enjoyed your article