Journal Club Summary
Usefulness: 3/5
Kim K, Kim YH, Kim SY, Kim S, Lee YJ, Kim KP, Lee HS, Ahn S, Kim T, Hwang SS, Song KJ, Kang SB, Kim DW, Park SH, Lee KH.
N Engl J Med. 2012 Apr 26;366(17):1596-605
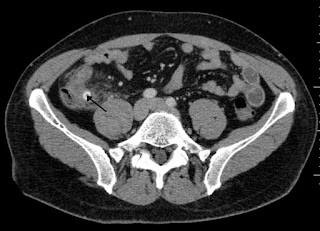
This single-centre randomized non-inferiority trial found that low-dose abdominal CT was non-inferior to standard dose in terms of negative appendectomy rates with an absolute increase of 0.3% in the low dose group (0.3, 95% confidence interval -3.8 to 4.6). The group felt this was not a practice changing study, as it did not include obese patients; there was a high risk of bias as the interpreting radiologists were not blinded, and it was not applicable to our clinical practice in Canada where we often obtain an ultrasound before CT.
By: Dr. Hanzla Tariq
(Presented January 2013)
Epi Lesson:
Non-inferiority trials are distinct from superiority trials such that they are designed to determine whether a given intervention is non-inferior by a pre-specified margin compared to a control. This is not the same as equivalence and a key section of the methods to examine is the sample size calculation where the non-inferiority margin is specified. Ideally, researchers explain how this margin was determined (based on previous placebo controlled trials, consensus of experts). The critical reader will ask themselves if they feel this margin is truly clinically significant.
By: Dr. Lisa Calder