Journal Club Summary
 Methodology Score: 3.5/5
Methodology Score: 3.5/5 Usefulness Score: 3.5 /5
Talan DA, et al.
N Engl J Med. 2016 Mar 3;374(9):823-32
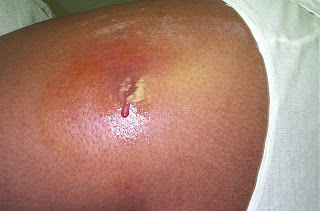
This RCT conducted at 5 US emergency departments including over 1200 outpatients aged over 12 years compared treatment of TMP-SMX, 320 mg/1600 mg twice daily for 7 days with placebo for drained skin abscesses ≥2 cm in diameter. The cure rate 7 to 14 days after treatment end was higher in the TMP-SMX group (80.5 versus 73.6 percent). Based on these findings, abscess size ≥2 cm in diameter may be a useful threshold for guiding decisions regarding use of antibiotic therapy after I&D. It should be noted that wound cultures were positive for MRSA in 45 percent of cases, which is surprisingly high and may not apply to most populations. (N.B. percent of MRSA+ swabs at TOH is 18%).
Some limitations of the study included medication adherence, assumed failure of cure when patients were lost to follow up, unclear allocation concealment, and performing multiple comparisons of secondary outcomes without statistical adjustment for such comparisons.
By: Dr. Julie Kim
Epi lesson:
It is not uncommon for a manuscript to report several secondary outcomes. The number of secondary comparisons is directly proportional to the chance that one of them will end-up being statistically significant by chance alone. To account for this, statisticians should make it proportionally more difficult to find such a statistical difference. The Bonferroni correction suggests that the level of significance (alpha error, 0.05) should be divided by the number of comparisons made i.e. 0.05/5 comparisons = new alpha of 0.01.
By: Dr. Christian Vaillancourt

