 Journal Club Summary
Journal Club Summary
Methodology Score: 2.5/5
Usefulness Score: 3/5
Ortiz M, et al.
Eur Heart J. 2016 Jun 28. pii: ehw230. [Epub ahead of print]
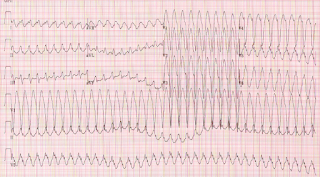
This RCT of patients with tolerated wide complex tachycardia found that IV Procainamide (10 mg/kg IV over 20 minutes) was better than IV Amiodarone (5 mg/kg over 20 minutes): less major cardiac adverse events, more efficacious in terminating tachycardia, and more efficacious in both endpoints in patients with structural heart disease. We found this article useful for deciding which chemical therapy should be first line in patients with stable ventricular tachycardia; however, we felt that the study would have been stronger if the physicians were blinded to the treatment drug.
By: Dr. Samantha Calder-Sprackman
Epi lesson:
Open label trials may be promoted as pragmatic trials but a lack of blinding to treatment allocation is a fundamental threat to internal validity. Blinding reduces ascertainment bias (the likelihood of differential assessment of outcome). It is not always possible to undertake blinding in a RCT, especially for surgical or procedural interventions. Drug studies can and must be blinded so readers should be very skeptical when this has not been done. For other studies, ask yourself if it was possible to blind and whether determination of outcome was free of bias.
By: Drs. Lisa Calder and Ian Stiell
