There were some notable papers published in trauma over the past 12 months or so. Here’s a look at three of them, which might help answer a few key questions in trauma management:
- When should you order a CT chest?
- What blood products should we be giving to trauma patients?
- Should we be giving mannitol or hypertonic saline to severe traumatic brain injury?
CLINICAL DECISION RULES FOR OBTAINING A CT CHEST
Derivation and Validation of Two Decision Instruments for Selective Chest CT in Blunt Trauma
Why Do We Care?
- We want to ensure we are not missing significant thoracic injuries in trauma patients
- However, CT chests take time, cost money, and expose patient to significant amounts of radiation.
- A chest CT is about 7 mSv of radiation, (compared to 14 mSv for an abdo/pelvis CT), which is roughly equivalent to 70 PA and lateral CXRs, or 350 trans-Atlantic flights
What’s Known Already?
- In a US study of nearly 10 000 blunt trauma patients, the yield of CT chest for detecting any thoracic injury after a normal CXR was 15%, with a total radiation dose of 59 mSv per injury, and a total radiation dose of 593 mSv per clinically major thoracic injury.
This means that for every 23 major injuries diagnosed on CT, potentially one cancer will be caused.
- Researchers have attempted coming up with clinical decision rules about obtaining chest CTs in trauma before, but they have been limited either by the need for multiple other plain x-rays, or by being small, retrospective rules that might be difficult to apply prospectively.
What is Unknown?
- How can we select which blunt trauma patients require a CT chest, and which are suitable for CXR alone?
What Does This Paper Tell Us?
Methods:
- Derivation and validation of 2 CDRs at 8 trauma centres (California, NJ, and Boston) from 2011-2014
- Inclusion criteria:
- Over 14 yrs of age
- Presented to ED with blunt trauma <6hrs
- Any chest imaging (CXR or CT chest) ordered in ED at discretion of providers
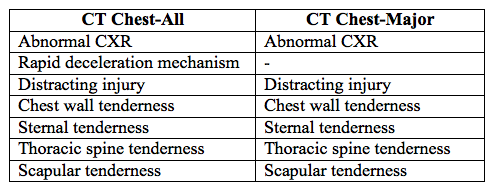
- Study sheets provided MDs with a list of 14 candidate CDR criteria (based on reviews of the literature and investigator consensus):
Outcomes:
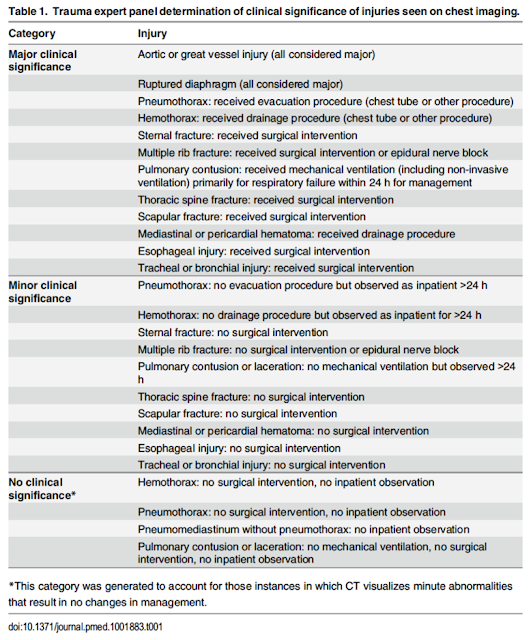
10 EPs and trauma surgeons classified potential thoracic injuries as insignificant, minor, or major (using need for intervention, observation, or hospitalization as a guide). Note that many of the minor clinical injuries were still observed for >24 hrs:
Sample size:
- Targeted sensitivities of 98% for major injury, and 95% for all injuries (with a 95% CI of +/- 2%)
- Assumed prevalence rates of 5% for major injury and 10% for all injuries
- calculated need to enrol 4570 patients (ended up enrolling 11 477 as a buffer)
Derivation and validation:
- Chest CT for all injuries (CT Chest-All) was derived first using recursive partitioning, with a sensitivity of 99% for major injuries, and 95% for all injuries
- Then criteria were removed to derive Chest CT-Major, with a sensitivity of 99% for major injury, but a lower acceptable threshold (90%) for all injuries
- Primary analysis was done in the group of patients that received both CXR and CT chest (31.2% in the derivation phase)
- However all patients were followed and a sensitivity analysis performed on the entire group of patients who received any imaging during the validation phase.
- Patients who received no imaging were not followed (previous studies having suggested that the rate of injury in this group is negligible)
Results
- Of patients in the derivation phase, 31.2% had both CXR and CT chest
- Rates of injury in this groups
- Major 7.7%
- Minor/insignificant 27%
- Of patients in the validation phase, 48% had both CXR and CT chest
- Rates of injury in this groups
- Major 4.6%
- Minor/insignificant 22.3%
- CDRs:
- CT Chest-All (for detecting major AND minor injuries)
- Sensitivity 95.4%
- Specificity 25.5%
- NPV 93.9%
- CT Chest-Major (for detecting major injuries)
- Sensitivity 99.2%
- Specificity 31.7%
- NPV 99.9%
- In the validation population of this study, the authors predict that use of Chest CT-All would have avoided 25% of chest CTs, and Chest CT-Major would have avoided 37%
- Injuries missed:
- 1/120 major injuries in this study was not detected by either rule – a PTX that was treated with a chest tube in an 80yo male that fell down the stairs and had a SAH
- 31 minor injuries were missed by Chest CT-All and 64 were missed by Chest CT-Major (mostly rib fractures, sternal fractures, and pulmonary contusions).
- All were non-operative and non-interventional. 9 of these 65 ‘missed’ injuries were sent home directly from the ED, and none returned to the ED
Conclusions and Limitations
- This is a well-designed prospective derivation and validation of 2 CDRs to help decide what type of imaging a blunt trauma pt should undergo
- The derivation of dual CDRs allows for MDs to practice differently depending on their comfort in missing injuries that do not require intervention
- As per the authors themselves:
- This type of rule should not be used to determine whether a patient needs thoracic imaging or a CT chest in the first place (i.e. as a rule-in, as in some centres using the CDR that way is likely to increase CT use)
- But rather to determine whether you are able to forego a CT chest in a pt whom you have already decided needs some form of thoracic imaging (i.e. as a rule-out).
- In our centre therefore, it may be of less use (as our reflex is likely NOT to perform a CT chest unless there is a strong clinical suspicion)
- Some may disagree with the assessment of major and minor injuries (as some of the minor injuries were still admitted and observed for >24 hrs)
- The rules require external validation prior to wide adoption
Take-home and going forward:
- In blunt trauma patients for whom you have already determined a need for thoracic imaging, but are unsure if a CXR is sufficient, using the Nexus CT-Chest rules may help determine those patients in whom a CXR alone is adequate (though this is going to be a pretty small group, according to the rules).
- However, it is NOT specific enough to help you then determine which patients overall need a CT.
- Development of a ‘rule-in’ CDR that helps determine the more specific factors suggesting a pt needs a CT chest after initial CXR would be helpful
RATIOS OF BLOOD PRODUCTS IN MASSIVE TRANSFUSION
The PROPPR Trial.
Why do we care?
- 20-40% of trauma deaths that occur after hospitalization are due to massive haemorrhage
- Improved resuscitation and transfusion techniques may help reduce this rate of mortality
What’s known already?
- Traditional resuscitation techniques in haemorrhage involve the administration of crystalloid fluid and PRBCs, often slowly and targeting hemoglobin levels
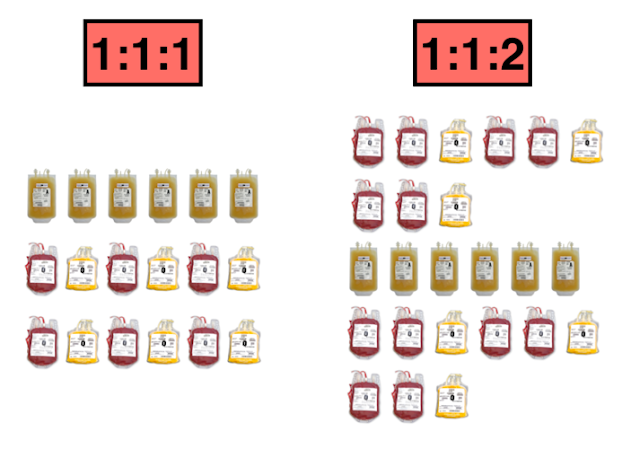
- ‘Damage control resuscitation’ is the rapid transfusion of blood products (plasma, platelets, and PRBCs) in a balanced ratio (usually 1:1:1) designed to approximate whole blood, as well as the prevention and correction of coagulopathy
- The goal is to better treat intravascular volume deficits, the acute coagulopathy of trauma, to preserve oxygen-carrying capacity, repair the endothelium, and prevent dilutional coagulopathy
- Retrospective military data of 246 trauma patients undergoing massive transfusion has previously shown that a transfusion ratio of 1:8 (plasma to PRBCs) is associated with a 65% mortality rate, compared to a mortality of 19% with ratios of 1:1.4
- Prospective observational data from the civilian world has shown similar, though perhaps less significant trends, with a 30-day mortality of 64% in patients receiving <1:2 (plasma to PRBCs) and 41% in patients receiving >1:2.
The PROMMTT study (2013) was a prospective observational study looking at 905 trauma patients admitted to hospital who required at least 3 units of blood products within 24 hrs (not the usual massive transfusion population that was looked at previously, in an effort to exclude the survivor bias that occurs when you require patients to receive at least 10 units)
- It showed reduced hazard ratios for death within the first 6 hrs when comparing low (<1:2) and high (>1:1) ratios of plasma:PRBCs or platelets:PRBCs (with hazard ratios going as low as 0.23 in the high ratio groups)
- However, the study was limited by poor standardization, and seemingly high heterogeneity in terms of how and when patients got blood products
What’s unknown?
Does a balanced transfusion strategy affect mortality when studied in a prospective, randomized fashion?
Does a balanced transfusion strategy affect mortality when studied in a prospective, randomized fashion?
What does this paper tell us?
Methods
- Randomized trial of 680 trauma patients requiring trauma team activation, administration of at least one unit of blood in first hour, and predicted to require massive transfusion (either by MD judgement or using the Assessment of Blood Consumption score) at 12 different Level 1 trauma centres in North America
- Excluded pregnancy, burns, inhalational injuries, CPR, and (most significantly) those who were not expected to survive past 1 hr on arrival to the ED.
- The two arms were composed of a 1:1:1 ratio and 1:1:2 ratio based on the PROMMTT study (which showed that clinicians clustered around these ratios anyways)
1:1:1 protocol:
- All containers included: 6 units of plasma, 1 dose of platelets (6 units on average), and 6 units of PRBCs
- Transfused in the following order: platelets first, then alternating RBC and plasma
1:1:2 protocol (composition and order changed from odd to even containers):
- Odd-numbered containers included: 3 units of plasma, 0 doses of platelets, 6 units of PRBCs
- Transfused in the following order: 2 units of PRBCs then 1 unit of plasma (repeated x 2)
- Even-numbered containers included: 3 units of plasma, 1 dose of platelets (6 units on average), and 6 units of PRBCs
- Transfused in the following order: platelets first, then alternating 2 units of PRBCs and 1 unit of plasma
- Stopping transfusion was as clinically indicated
Primary outcomes:
- 24hr and 30 day mortality
Secondary outcomes:
- Time to hemostasis (either judged by surgeon or in IR suite)
- Number and type of blood products used before hemostasis was achieved, and after hemostasis up the first 24hrs
- Complications
- Hospital, ventilator and ICU-free days
- Incidence of surgery
- Functional status at discharge/30 days
Results
Sample size of 580 planned to detect a clinically meaningful 10% difference in 24hr mortality and 12% difference in 30-day mortality (based on prior data) — increased to 680 based on the final observed mortality proportions to reach 95% power
- 4 patients lost to 30 day follow-up: sensitivity analysis for all possible outcomes was computed and a range of values incorporated
- P values were adjusted for 2 interim analyses (p=0.044)
- 11 185 patients assessed for eligibility, 10 505 excluded (most did not receive at least one unit of blood, were not direct transfers, or were not predicted to need massive transfusion – 130 were excluded because they were expected to die within an hr)
- 75% of patients required IR or OR within 2hrs
PRIMARY OUTCOME:
- 24hr mortality:
- 1:1:1 – 12.7%
- 1:1:2 – 17.0% (NS)
- Exsanguination (the main cause of death) in the 1st 24hrs WAS reduced significantly (9.2% vs. 14.6%, p=0.03)
- 30 day mortality:
- 1:1:1 – 22.4%
- 1:1:2 – 26.1% (NS)
Sensitivity analysis done to exclude patients who were given blood products out of the pre-specified order yielded similar mortality results
More patients in the 1:1:1 group achieved hemostasis (86.1% vs. 78.1%) [This is an unblinded outcome]
- In the 1st 24hrs, the 1:1:1 group received more total blood products (a median of 25.5 units of blood products, compared to 19 units in the other group), but similar total amount of PRBCs
- 1:1:1 got 7 units plasma, 12 units platelets, 9 units PRBCs
- 1:1:2 got 5 units plasma, 6 units platelets, and 9 units PRBCs
Also note, the patients did NOT actually receive the pre-specified ratios, for the most part:
- The 1:1:1 group actually ended up receiving something more closely approximating 1:1.5:1 (ie. more platelets)
- The 1:1:2 group actually ended up receiving something more closely approximating 2:1:4 (ie. less platelets)
This occurred, according to the authors, because after the intervention period there was a catching up of products based on lab-directed care
There were no significant differences in complications at 30 days (ARDS, MODS, VTE, sepsis, and transfusion-related complications), though the overall rate of complications was high (89%))
Conclusions and Limitations
While this trial failed to detect a statistically significant mortality outcome when comparing the two transfusion protocols, it DID show a trend for both, and it DID show a statistically significant reduction in death by exsanguination, without an apparent increase in adverse effects
The authors suggest that if they had been permitted to move the endpoints much earlier (ie. mortality within 3-4 hrs) that they might have demonstrated a more robust mortality difference (as that is when most exsanguination deaths occurs)
As well, the authors were unable to exclude unsurvivable brain injuries, which made up a lot of the overall mortality (although we likely can’t reliably exclude this group when deciding on initiating massive transfusions either)
The trial was also underpowered (or overly ambitious) to detect mortality differences of less than 10% (given the observed difference of 4.2%, the study would have needed to enrol nearly 3000 patients to detect a significant result)
The authors conclude that a 1:1:1 protocol is reasonable while patients are actively bleeding or interventions are taking place, followed by lab-guided treatment once hemostasis has been achieved
Take-homes and going forward:
There is probably a trend towards improved mortality with transfusion ratios more closely approximating whole blood, and the PROPPR investigators may simply have overestimated the effect size (ie. it is not more than 10%), or they may have unfortunately compared two strategies that are too similar to tease out really significant differences (remember, the retrospective military studies were comparing 1:1.4 and 1:8!)
Perhaps transfusion care will go the way of sepsis care in the future, with the realization that early administration of something similar to whole blood is the key, rather than a rigid ratio
Questions going forward:
- How can we better predict what patients should be included in a massive transfusion protocol to begin with?
- What is the role of laboratory guided transfusion protocols?
HYPERTONIC SALINE IN SEVERE TRAUMATIC BRAIN INJURY
HTS in Severe TBI: Systematic Review and Meta-analysis
Why do we care?
- Increased ICP in severe traumatic brain injury (sTBI) is strongly associated with mortality
- Osmotic agents are used in sTBI with signs of increased ICP such as:
- A herniation syndrome
- Cushing’s reflex
- Progressive neurologic deterioration not attributable to an extracranial cause
The idea is that creating an osmotic gradient will keep more fluid intravascular, and that this will reduce cerebral edema, and therefore ICP
Mannitol is the classic osmotic agent used in sTBI, and may provide other beneficial effects as well (may be a systemic volume expander and improve overall BP, reduce blood viscosity and therefore improve cerebral blood flow, and act as a free radical scavenger)
- However, it may also have detrimental effects:
- May produce AKI
- May actually produce HYPOtension in large doses (due to diuresis)
- May cause paradoxical bleeding into a cerebral hematoma by reducing the local tamponade
Hypertonic saline (HTS) has long been proposed as an alternative osmotic agent, primarily due to its smaller volume and lower risk of inducing hypotension
It also has a proposed alternate mechanism besides a direct osmolar effect:
- It may increase systemic MAP, which leads to cerebral vasoconstriction (in a brain where auto-regulation persists), and thus a reduction in ICP
It’s important to note that HTS may also have adverse effects associated with it:
- May also produce AKI
- Risk of cerebral pontine myelinolysis
- Hypernatremia
- May lead to RBC lysis
- May worsen coagulopathy
What’s known already?
Mannitol is traditionally the most widely used osmotic agent (83% of centres in the US, and 100% of neurosurgical centres in the UK)
A recent survey of EPs in Quebec, on the other hand, suggested that 53% use HTS instead
- The Brain Trauma Foundation and American Association of Neurological Surgeons clinical practice guidelines give a Level II recommendation that mannitol is effective for control of raised ICP.
- They note that current evidence is not sufficient to make recommendations about HTS, as does the 2008 ATLS guidelines
Studies on both mannitol and HTS are, in general, fairly poor
A Cochrane review of mannitol in acute TBI found only four RCT’s worth reviewing. Only one of them compared mannitol and hypertonic saline – in a trial of 20 ICU patients.
While the HTS group had fewer episodes of raised ICP, shorter episodes, and required less CSF drainage, it is too small a group to make any claims regarding patient-centred morbidity or mortality
What’s unknown?
What is the cumulative evidence for using HTS in sTBI
What does this paper tell us?
Methods
- The largest systematic review and meta-analysis of of RCTs studying HTS in sTBI to date
- Reviewed a wide variety of databases and grey literature, coming up with 11 studies (1820 pts) of adults with a sTBI (GCS <8), randomly assigned to receive HTS vs any other type of solution
- Primary outcomes looked at by the investigators were death and ICP control
- They wanted to look at secondary outcomes of neurological outcome at discharge, LOS, but almost none of the studies reported these
- They also looked at adverse events as reported in the studies
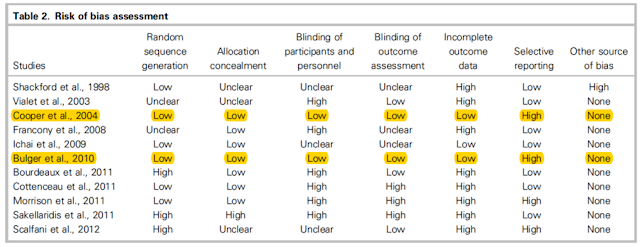
- Used the Cochrane Collaboration tool for risk of bias
Results
- Of the 11 studies, only 3 enrolled more than 100 pts
- Only 2 studies were considered at low risk of overall bias (only missing one of the criteria on the Cochrane risk of bias tool
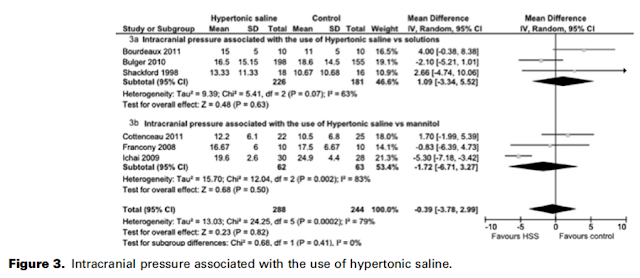
Four studies looked at mortality, but only one of them was in comparison with mannitol (the Vialet trial) – the others all compared single bolus HTS with either NS or RL. None of them showed a mortality benefit:
When assessing for control of ICP, there was no significant improvement with HTS as compared to any other solution (mannitol or NS/RL), setting:
- All the studies monitored osmolality and natremia, but not in a standardized way that would permit meta-analysis.
- All studies noted some incidence of hypernatremia, but most did not monitor for any related clinical adverse events
Conclusions and Limitations
The authors conclude that there is no apparent clinical benefit with the use of HTS to other fluids in treating ICP in sTBI
They note that 6 previous systematic reviews have been done before theirs, and note that 5 of them suggested a trend to reduction in ICP existed when using HTS.
However, all those reviews included fewer overall studies, and many included either non-RCT evidence (retrospective and observational data), or other neurosurgical conditions besides sTBI (eg. tumour, stroke), thus increasing clinical heterogeneity
The authors also acknowledge the high risk of bias in the included studies and the limited number of studies overall
Take-homes and going forward:
In this study, the largest review of HTS in sTBI to date, there is no observable mortality benefit or effect on intracranial pressure with the use of HTS when compared to other solutions
The evidence is limited primarily by a lack of standardization of doses of HTS, as well as a lack of focus on patient-centred outcomes (mortality and disability)
Questions that still need to be answered:
- What is the ideal concentration of HTS to use? What is the ideal agent in the ED? What effect do either mannitol or HTS actually have on mortality and disability?
In practice:
Probably reasonable to use either agent, although the balance between evidence and practice guidelines likely still favours mannitol
A strategy that chooses according to adverse effects is likely the most reasonable strategy (ie. avoiding mannitol in patients with AKI or hypotension, avoiding HTS in patients prone to demyelinolysis or who are coagulopathic).
Dr. Francis Bakewell is a 5th year Emergency Medicine Resident at the University of Ottawa and The Ottawa Hospital. He has a particular interest in education, trauma and communication.
Edited by Dr. Robert Suttie, PGY2 Emergency Medicine at the University of Ottawa.
References:
1. http://www.XRayRisk.com
2. Rodriguez RM, Baumann BM, Raja AS, Langdorf MI, Anglin D, Bradley RN, et al. Diagnostic yields, charges, and radiation dose of chest imaging in blunt trauma evaluation. Acad Emerg Med. 2014; 6:644–650
3. Brink M, Deunk J, Dekker HM, Edwards MJ, Kool DR, van Vugt AB, et al. Criteria for the selective use of chest computed tomography in blunt trauma patients. Eur Radiol. 2010; 20:818–828. doi: 10.1007/s00330-009-1608-y PMID: 19760233
4. Payrastre J, Upadhye S, Worster A, et al. The SCRAP Rule: The derivation and internal validation of a clinical decision rule for computed tomography of the chest in blunt thoracic trauma. CJEM. 2012; 14(6): 344-353
5. Tisherman SA, Schmicker RH, Brasel KJ, et al. Detailed description of all deaths in both the Shock and Traumatic Brain Injury Hypertonic Saline Trials of the Resuscitation Outcomes Consortium [published online July 28, 2014]. Ann Surg. doi:10.1097/SLA.0b013e3181df0401.
6. Borgman MA, Spinella PC, Perkins JG, et al. The ratio of blood products transfused affects mortality in patients receiving massive transfusions at a combat support hospital. J Trauma. 2007;63(4): 805-813.
7. Shaz BH, Dente CJ, Nicholas J, et al. Increased number of coagulation products in relationship to red blood cell products transfused improves mortality in trauma patients. Transfusion. 2010;50 (2):493-500.
8. Giulioni M, Ursino M. Impact of cerebral perfusion pressure and autoregulation on intracranial dynamics: a modeling study. Neurosurgery 1996;39(5):1005-15.
9. Ghajar J, Hariri RJ, Narayan RK, et al. Survey of critical care management of comatose, head-injured patients in the United States. Critical Care Medicine 1995;23(3):560-7
10. Jeevaratnam DR, Menon DK. Survey of intensive care of severely head injured patients in the United Kingdom. BMJ. 1996; 312:994-7
11. Berger Pelletier E, Emond M, Lauzier F, et al. Hyperosmolar Therapy in Severe Traumatic Brain Injury: A Survey of Emergency Physicians from a Large Canadian Province. PLoS One. 2014 Apr 22;9(4):e95778. doi: 10.1371/journal.pone.0095778. eCollection 2014