Here we seek to review relatively common neurology presentations and some controversies surrounding their management. See our other ED-Neurology Reviews here including migraine therapies and stroke evidence.
To include Myasthenia Gravis, Guillian Barre Syndrome, and Multiple Sclerosis in the ED.
For each we will review:
- A brief overview of pathophysiology
- Clinical presentation and work-up
- Important management considerations for the ED physician
Myasthenia Gravis:
Epidemiology:
- Myasthenia gravis (MG) has a bimodal distribution. If affects predominately 20 to 30-year-old females and 60 to 80-year-old males.
- An epidemiological study by Breiner et al. in 2016 found a prevalence of 32/100,000 and incidence of 3.2/100,000 in Ontario. Prevalence was noted to be increasing possibly due to better treatment.
- It remains under-diagnosed due to the broad differential, subtle findings and fluctuating clinical course.
Pathophysiology:
- Myasthenia Gravis is an autoimmune neuromuscular junction disorder whereby antibodies block and destroy nicotinic acetylcholine receptors.
- This inhibits response to acetylcholine in the synapse preventing nerve signal transmission and muscle contraction.
- Other forms exist including: neonatal, neuromuscular junction mutations
Clinical features:
- Fatigable muscle weakness. It is not sensation of tiredness or acute onset of weakness.
- Features include:
- ocular findings (ptosis, diplopia)
- limb weakness (proximal> distal, upper extremity> lower extremity)
- respiratory symptoms (myasthenic crisis)
- bulbar symptoms (slurred speech, nasal speech after long conversations, jaw weakness after chewing, dysphagia, risk aspiration)
- Fun Fact: Sleepy from Snow White and the Seven Dwarfs© was supposedly based on a friend of Walt Disney who had Myasthenia Gravis.
Investigations
- Single breath testing consists of having a patient attempt to count as high as possible on a single breath in normal voice. On average a normal person can count to 50.
- In a prospective, cross sectional, single blind study by Elsheikh et al. in 2016 they found that single breath count less than 20 correlated with respiratory compromise as well as forced vital capacity.
- Normal FVC = 60-70ml/kg. They found single breath test # x 116 = actual FVC (ml). This study was in mild disease and in outpatient setting so difficult to extrapolate to the emergency department.
- Edrophonium testing (is no longer available in the ED) shows improvement in symptoms
Is there any evidence of clinical exam tools we can use in the ED which increases our suspicion of myasthenia gravis? A JAMA rational clinical exam series in 2005 completed a review.
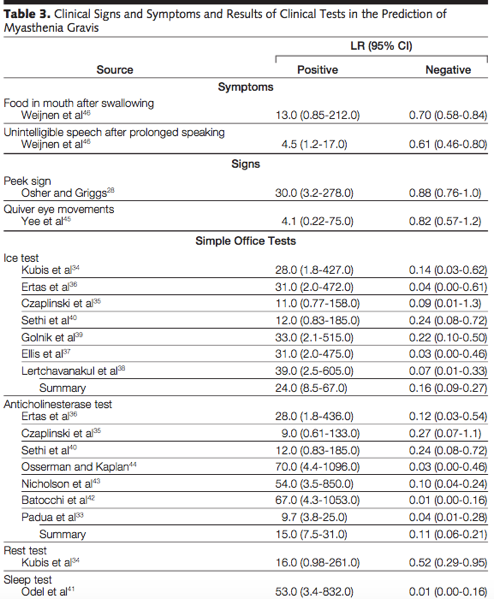
- Only ice test, anti-cholinesterase test, and a sleep test had a high negative likelihood ratio making them more helpful to rule out disease (see table 3 below). Limitations of this review included the small sample sizes and high prevalence of disease in the sample population making the pre-test odds higher.
- Ice Test = placing ice on eyelids for 2 minutes and examining if ptosis improved by 2 mm.
- Rest/Sleep Test = improvement in symptoms with sleep or rest
- Peek sign = voluntary closure of eyes resulting in involuntary separation of lid margins
Diagnosis and Differential:
- Most important step in the ED is consider it on the differential.
- Remainder of work-up will be completed by neurology (serology antibody testing and electrophysiological confirmation with repetitive nerve stimulation)
- Two important conditions to consider on the differential include Botulism and Lambert-Eaton Syndrome
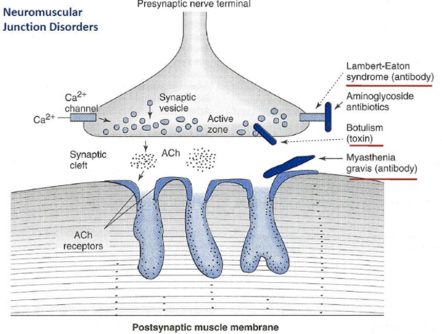
Lambert-Eaton Syndrome:
- 60% associated with malignancy (most common small cell lung cancer)
- antibodies block calcium influx
- Symptoms: leg weakness> arm weakness, rare bulbar symptoms, improves with repetitive use
- Treatment: Treat the underlying cause
Botulism:
- Caused by botulinum toxin
- Prevents exocytosis of Ach
- Sx: descending paralysis, affecting cranial nerves/bulbar first
- Rx: airway support, antitoxin
- 205 cases and 11 deaths between 1985-2005. Most in Nunavut, Quebec and BC. Associated with marine mammals, fish preparation
Treatment:
Why do we care about outpatient management?
Therapies for MG can make patients more likely to present to the ED with specific emergencies:
- Pyridostigmine (Mestinon) – can precipitate cholinergic crisis
- Chronic immunotherapies – means immunosuppression (at risk of opportunistic infections)
Additionally, when treating patients with MG for other illnesses we must be aware of the multitude of medications that can exacerbate the condition:
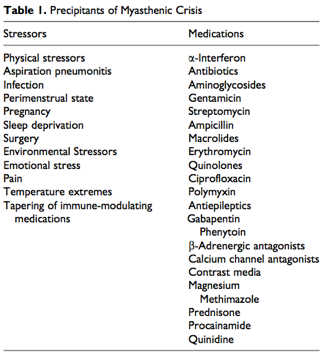
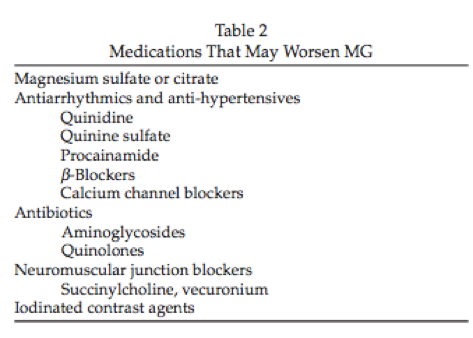
Myasthenic Crisis:
Definition: serious, life threatening exacerbation and potential airway compromise from respiratory/bulbar dysfunction.
- 15-20% of patients affected at least once in their life time.
- In 1/5 of patients it is the initial presentation of the disease
- Mortality rate has decreased from 42% to 4% since 1960.
Management
- Recognize the diagnosis, can often be forgotten especially if it is the initial presentation of the disease
- Look for precipitate (40% associated with respiratory infections)
- Differentiate from cholinergic crisis (often impossible to do). No case reports in literature
- Discontinue pyridostigmine – can worsen bronchial secretions and increase risk of arrhythmia
- Early involvement of neurology/ICU – they will decide treatment with IVIG or Plasma Exchange (equally effective)
- Monitor for Respiratory failure and need for intervention.
- Respiratory failure looks different in patients with myasthenia gravis. They do not always appear in classic failure as accessory muscles fatigue.
- Do not be falsely reassured by normal oxygen saturation. Hypoxia occurs late in neuromuscular respiratory failure.
- Be aware of the snapshot in time. Always remember the fatguability factor. They can look okay at first glance but evolve over time especially if other factors are involved (e.g. pneumonia, etc).
Controversies in the Treatment of Myasthenic Crisis
- Should we give steroids?
- Lacomis et al. completed a systemic review which showed that myasthenic crisis was precipitated in 8-19% following steroids and almost ½ patients worsen on steroids.
- Therefore, no steroids acutely. Neurology will decide on timing of initiation
- Is there a role for BiPAP?
- Three retrospective studies (Rabinstein 2002, Seneviratne 2008, Wu 2009) found BiPAP can be an effective mode of ventilation and prevent intubation therefore seems reasonable to attempt as an initial therapy.
- However, if severe bulbar dysfunction and unable to handle secretions BiPAP is not a good option.
- Can we use PFTs/ Blood gases to predict need for intubation?
- Many texts attempt to use criteria for intubation from literature for Guillian-Barre Syndrome using a 20/30/40 rule:
- Forced vital capacity FVC < 20ml/kg
- Maximal inspiratory pressure < 30
- Maximal Expiratory Pressure < 40
- Thieben et al (2005) retrospective analysis of 42 patients found vital capacity (VC) is a poor predictor for need for intubation.
- Approximately three-fourths of the patients did not require ventilation had a VC of less than 20 ml/kg. This was supported in an additional study by Thomas et al (2001).
- Rieder et al (1995) found in their retrospective analysis that repeated measurement of VC is a poor predictor for need for mechanical ventilation.
- Conclusion: No strict PFT cut offs exist to predict need for intubation.
- These studies were obviously limited by physician judgment for need for intubation and we are unable to determine how PFT values or trends influenced their decisions.
- These studies do suggest that respiratory support is unlikely to be required in patients with vital capacity > 20ml/kg.
- Many texts attempt to use criteria for intubation from literature for Guillian-Barre Syndrome using a 20/30/40 rule:
- When proceeding with intubation, which neuromuscular blocker is preferred?
- Most guidelines are from the anesthesia literature
- If using non-depolarizing agent (Rocuronium) the recommendation is to use incremental small doses as patients are extremely sensitive and the duration of effect is unknown
- How does this apply to the ED however?
- De Haes (2002) showed rocuronium is safe for use in these patients. The duration of effect of rocuronium probably does not matter in the ED. No practice variation.
- How does this apply to the ED however?
- If using depolarizing agent (Succinylcholine) literature suggests that patients can be resistant to blockade.
- How does this affect our practice?
- Eisenkraft et al (1988) took 15 patients and measured fast twitch depression/ degree of muscle blockade with increasing doses of succinylcholine and found resistance to succinycholine. They extrapolated their data to require 2-2.6 times normal dose to have effective neuromuscular blockade.
- In the ED, if we use we are using typical doses of succinycholine for rapid sequence intubation (1.5 mg/kg) then most patients with myasthenia gravis will have almost 100% first twitch depression. So you can consider using slightly higher dose but probably not needed in rapid sequence intubation.
- How does this affect our practice?
Summary of Myasthenia Gravis:
- Put myasthenia gravis on your differential
- Caution with prescribing certain medications
- These patients are at high risk of deterioration and do not present with typical respiratory distress. Early involvement of neurology/ICU is critical.
- Treat the patient and not the PFTs
Guillian-Barre Syndrome
Epidemiology:
- Incidence 2/100,000
- Males>Females, increase with age
Pathophysiology:
- Exact pathophysiology unknown. Thought to be immune mediated reaction against myelin or axons in the peripheral nervous system
- 2/3 associated with antecedent respiratory tract infection or diarrheal illness (most common Campylobacter jejuni)
- Do vaccines cause GBS?
- Bottom line: no, you have a higher chance of getting GBS from influenza than from the vaccine. The CDC, as a precaution, recommends against immunizations in the acute period of GBS up to one year. Future avoidance is suggested to be weighed against potential risk of not being immunized if GBS symptoms occurred within 6 weeks of vaccination.
- Multiple forms of GBS exist:
- 90% classical acute inflammatory demyelinating polyneuropathy (AIDP)
- Other forms:
- Axonal
- Chronic inflammatory demyelinating polyneuropathy (CIDP)
- Miller Fischer varient (areflexia, ataxia, ophthalmoplegia)
Clinical:
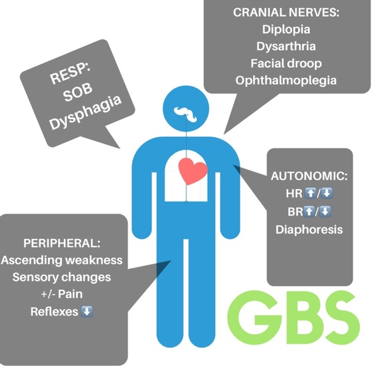
- Most important cause of acute flaccid paralysis after polio
- Presents 2-4 weeks after GI/resp illness
- Often ascending/symmetric muscle weakness, areflexia however can present atypically
- Sensory symptoms often mild, not always reproducible on physical exam. Often precede weakness.
- Generally begin in fingers/toes and stop at wrists/ankles
- In a prospective, longitudinal study 89% of patients had pain at some time during their illness. (Fokke 2014)
- If the patient is febrile, has bowel/bladder dysfunction or you find a clear sensory level or asymmetric weakness look for other causes.
Diagnosis/Work-Up:
- Lumbar puncture may be considered in ED. If febrile can help r/u west nile or lyme disease. Findings in GBS often normal initially. Can see elevation in CSF protein > 0.55g/L with N WBC.
- Peripheral neuropathy work-up, EMG studies will be organized by neurology
- Our job in the ED is to rule out brain stem CVA (acute onset) or acute spinal cord compression (MRI spine). The plantar reflexes in patient with GBS should be down going unless prior history of CVA/UMN disorder)
Treatment:
- Consider the differential diagnosis – r/o brain stem CVA/ acute spinal cord compression PRN
- Early involvement of neurology/ICU. They will decide on IVIG/Plasma Exchange (equal efficacy)
- Manage the pain – no studies to support specified pharmacotherapy. Gabapentin and carbamazepine have shown some promise.
Do we give steroids?
Cochrane Review by Hughes et al (2016) showed that according to moderate quality evidence, corticosteroids given alone do not speed recovery or affect long term outcomes. Conclusion: no role for steroids in the ED.
Can we use PFTs for prediction for intubation?
Thirty percent of patients with GBS will require intubation. However, there is limited literature available on this topic.
Lawn (2001) completed a retrospective study of 114 patients with severe GBS admitted to the ICU. They found that mechanical ventilation was likely to occur in those with:
- Rapid disease progression
- Bulbar dysfunction
- Bilateral facial weakness
- Dysautonomia.
Factors associated with progression to respiratory failure included:
- Vital capacity of less than 20 mL/kg
- Maximal inspiratory pressure less than 30 cm H2O
- Maximal expiratory pressure less than 40 cm H2O
- A reduction of more than 30% in vital capacity, maximal inspiratory pressure, or maximal expiratory pressure.
The conclusion drawn from this study is that we can predict deterioration and plan for elective intubation. At no point in the manuscript was the sensitivity or specificity of the combined rule actually evaluated. This is where the 20/30/40 rule is based.
Sharshar et al (2003) analyzed 722 patients and found that a mixture of predictors increased the likelihood of intubation (> 3 predictors = > 85% likelihood for mechanical ventilation). These include:
- Onset to admission < 7 days
- Unable to stand
- Unable to cough
- VC < 20
- Abnormal liver enzymes
- Unable to lift elbows
- Unable to lift head
Walgaard et al (2010) attempted to develop a prognostic model to accurately predicts respiratory insufficiency in the early stage of GBS called the EGRIS (Erasmus GBS Respiratory Insufficiency Score). They found that the most important predictors of need for mechanical ventilation included:
- Number of days between onset of weakness and hospital admission
- The MRC (Medical Research Council) sum score (muscle power)
- The presence of facial or bulbar weakness.
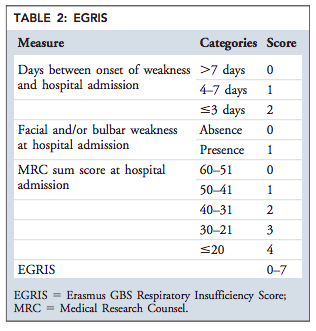
From Walgaard C, Lingsma HF, Ruts L, et al. Prediction of respiratory insufficiency in Guillain-Barré syndrome. Ann Neurol 2010; 67:781.
This tool had good discriminative ability (AUC 0.84). How to apply the EGRIS in clinical practice? Even with a EGRIS score of 2 (low risk group) the risk of intubation is 4%. This outlines the variability clinical course. Therefore, this tool is easy to use but not really clinically helpful.
No evidence for pulse oximetry/ABGs correlated to need for intubation. BiPAP has not been shown to be a helpful adjunct in GBS (limited evidence).
Neurology and ICU pearl: Trend is more meaningful in adjunct with clinical exam. Many pitfalls with use of bedside PFTs, Normally on admission at The Ottawa Hospital we monitor q 4 hours with PFTs x 24 h then patient dependent.
How can we manage autonomic dysfunction?
Degree of weakness does not always correlate to autonomic dysfunction. Patient present with number of cardiovascular complications. The most common being sinus tachycardia and blood pressure variability. They tend to be extremely sensitive to any stressor (e.g. anemia, positional change, medication changes, hypoxia etc).
According to the BMJ Best practice guidelines in 2016 and Cardiovascular Complications in Guillain Barre Syndrome Review by Mukerji in 2009 it is recommended to only treat tachyarrhythmias if life threatening. Bradyarrhythmias occur in up to 50% and there are no guidelines for pacemaker insertion. If patient is hypotensive a fluid challenge is advised before low dose vasopressor therapy. If vasopressor therapy is required it is recommended to start low with careful titration. There is no evidence of a target MAP but intra-arterial blood pressure monitoring is advised. If hypertension develops IV labetalol or nitroprusside is initially recommended.
Anesthesia concerns – use of succinylcholine:
Only case reports and theoretical risks exist of succinylcholine causing life threatening hyperkalemia. Fergusson et al. described four cases of VT post succinylcholine, Reilly et al. described one case where potassium increased from 4.3 to 8.6. The risk may even extend after the disorder has run its course as was documented in one case report. Vecuronium and rocuronium, both of which have minimal cardiovascular effects are recommended.
Summary of GBS:
- Rule out acute brainstem CVA/spinal cord compression
- Early involvement of neurology/ICU is recommended
- Do not use PFTs in isolation for decision for intubation. PFT trend in conjunction with the clinical picture may be more helpful
- Monitor for autonomic instability however just watching may be a better management option than treating.
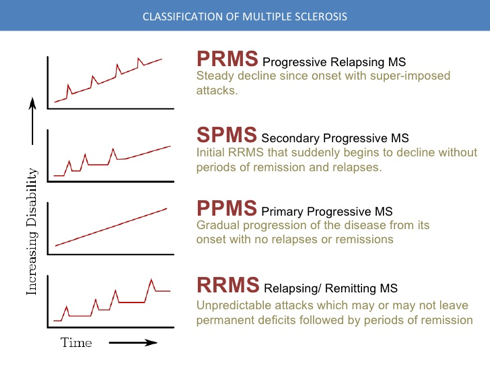
Multiple Sclerosis (Brief overview)
Epidemiology:
- Females > Males
- Age 20-40 years
- Inverse relationship between sun exposure, serum Vitamin D levels and prevalence
- Genetic link
Pathophysiology:
- Exact etiology unknown. Most widely accepted mechanism is an inflammatory, immune mediated disease affecting central nervous system resulting in demyelination and axonal degeneration.
- Other possible etiology – linked to virus ?EBV
Clinical:
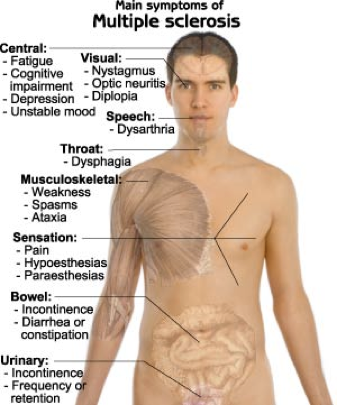
Optic neuritis:
- Presenting feature of multiple sclerosis in 20% of patients
- Due to inflammation, demyelination, or degeneration of the optic nerve.
- Clinical:
- monoculuar visual loss (often central)
- severe eye pain (92%).
- May be assoc with photophobia, change in colour vision, flashes of light, papillitis, afferent pupillary defect
- Consider referral to both neurology and ophthalmology
- Should we use steroids?
- Cochrane review in 2012 showed no benefit in long term outcomes with either IV or oral steroids.
- The Optic Neuritis Treatment Trial was a 2008 RCT comparing IV steroid versus oral steroid versus placebo in the treatment of optic neuritis. It showed IV methyprednisolone accelerated recovery and reduced risk of conversion to MS within 2 years. However, long term outcomes showed no benefit.
Diagnosis:
- Diagnosis should not be an emergency department diagnosis. But it should be on the DDX. Only a consultant neurologist should make this diagnosis.
- Main thing is recognition and appropriate referral
- In ED need to rule out acute transverse myelitis or acute spinal cord compression
- MRI findings of MS including T2 hyperintensities are common and are non-specific
- Lumbar punctures no longer routine. Can see oligoclonal bands (in 90-95% of patients) and IgG CSF production (in 70-90%)
- Neurologist use McDonald criteria for diagnosis of multiple sclerosis.
- Newly updated in 2010 allows diagnosis of MS even after a first clinical episode.
Imaging:
- MRI is the test of choice. Obviously MRIs are not openly available in the ED. Often these patients with new neurological symptoms get a non-contrast CT. If you see an open ring sign it has a specificity of 85-94% for demyelinating condition in a retrospective case series. Ring enhancement is often closed in infectious or malignant etiology.
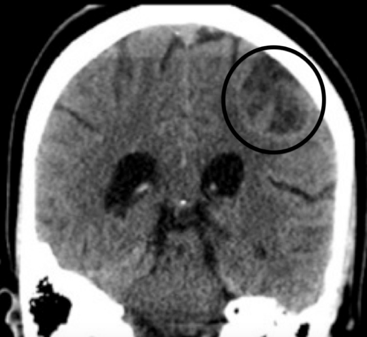
How often are we missing this diagnosis:
Farber et al (2013) in a retrospective observational study out of Mount Sinai found that 61% were diagnosed at the time of ED visit or on first admission.
Treatment of Acute MS attacks:
- Most MS attacks are not seen in the emergency department. They are managed as an outpatient by their own neurologist. In a true MS attack, neurology should be involved.
- The mainstay of treatment is IV steroids.
- As mentioned above, a Cochrane review by Burton et al in 2012 demonstrated that IV methyprednisolone was found to speed recovery but had no effect on long term outcome or repeat attacks.
- Le Page et al. (2015) in a multicentre, double blind, RCT found that oral methylprednisolone 1000mg was non-inferior to IV.
- Outside of acute MS attacks treatment with disease modifying agents should be left to the discretion of the neurologist.
- An exciting field here in Ottawa with Dr. Mark Freedman and Dr. Harold Atkins have shown a breakthrough with immunosuppression and stem cell transplants for prevention of relapse in 24 patients
- Most common what we see in the ED are pseudo-exacerbations. Which is defined as a worsening of symptoms in the setting of an external complicating factor that is not related to an actual new CNS lesion.
- In this case, there is a clear precipitant that needs to be identified.
- Main cause is urinary tract infections.
- The mainstay in management is treat the underlying cause
- No benefit with steroids. Often internal medicine is seeing these patients for admission.
Important note for use of succinylcholine in multiple sclerosis:
- Case report evidence and theoretical risk of hyperkalemia and cardiac arrhythmias. Advised to avoid use.
Summary of Multiple Sclerosis:
- Do not diagnosis multiple sclerosis in the ED
- Rule out mimickers such as acute spinal cord compression
- Often pseudoexacerbations of MS in ED. Look for precipitant
- Steroids help speed recovery but have little effect on overall outcome
References
- Breiner A, Widdifield J, Katzberg HD, et al. Epidemiology of myasthenia gravis in Ontario, Canada. Neuromuscul Disord 2016; 26:41.
- Vincent A. Unravelling the pathogenesis of myasthenia gravis. Nat Rev Immunol 2002; 2:797.
- Mantegazza R, Beghi E, Pareyson D, et al. A multicentre follow-up study of 1152 patients with myasthenia gravis in Italy. J Neurol 1990; 237:339
- Conti-Fine BM, Milani M, Kaminski HJ. Myasthenia gravis: past, present, and future. J Clin Invest 2006; 116:2843.
- Scherer K, BedlackRS. Does this patient have myasthenia gravis? JAMA 2005;293 (15):1906-14
- Silvestri NJ, Wolfe GI. Myasthenia gravis. Semin Neurol 2012; 32:215.
- Meriggioli MN, Sanders DB. Autoimmune myasthenia gravis: emerging clinical and biological heterogeneity. Lancet Neurol 2009; 8:475.
- Mahadeva B, Phillips LH 2nd, Juel VC. Autoimmune disorders of neuromuscular transmission. Semin Neurol 2008; 28:212.
- Farrugia ME, Vincent A. Autoimmune mediated neuromuscular junction defects. Curr Opin Neurol 2010; 23:489.
- Wendell LC, Levine JM. Myasthenic crisis. Neurohospitalist 2011; 1:16.
- Jani-Acsadi A, Lisak RP. Myasthenic crisis: guidelines for prevention and treatment. J Neurol Sci 2007; 261:127.
- Chaudhuri A, Behan PO. Myasthenic crisis. QJM 2009; 102:97.
- Rabinstein AA, Wijdicks EF. Warning signs of imminent respiratory failure in neurological patients. Semin Neurol 2003; 23:97.
- Seneviratne J, Mandrekar J, Wijdicks EF, Rabinstein AA. Noninvasive ventilation in myasthenic crisis. Arch Neurol 2008; 65:54.
- Wu JY, Kuo PH, Fan PC, et al. The role of non-invasive ventilation and factors predicting extubation outcome in myasthenic crisis. Neurocrit Care 2009; 10:35.
- Varelas PN, Chua HC, Natterman J, et al. Ventilatory care in myasthenia gravis crisis: assessing the baseline adverse event rate. Crit Care Med 2002; 30:2663.
- Sanders DB, Wolfe GI, Benatar M, et al. International consensus guidance for management of myasthenia gravis: Executive summary. Neurology 2016; 87:419.
- Gajdos P, Chevret S, Toyka KV. Intravenous immunoglobulin for myasthenia gravis. Cochrane Database Syst Rev 2012; 12:CD002277.
- Gilhus NE. Neuromuscular disease: acute treatment for myasthenia gravis. Nat Rev Neurol 2011; 7:132.
- Masters W, Bagshaw O. Review Article: Anesthetic Considerations in Paediatric Myasthenia Gravis. Autoimmune Diseases 2011; 250561
- Lacomis D. Myasthenic crisis. Neurocrit Care. 2005;3:189-194.
- Thieben M et al. Pulmonary Function Tests and Blood Gases in Worsening Myasthenia Gravis. Muscle Nerve. 2005; 32: 664-667
- Rieder P et al. The repeated measurement of vital capacity is a poor predictor of the need for mechanical ventilation in myasthenia gravis. Intensive Care Medicine. 1995; 21 (8): 663-668
- Eisenkraft JB, Book WJ, Mann SM, et al. Resistance to succinylcholine in myasthenia gravis: a dose-response study. Anesthesiology 1988; 69:760.
- Baraka A, Baroody M, Yazbeck V. Repeated doses of suxamethonium in the myasthenic patient. Anaesthesia 1993; 48:782.
- De Haes, J. H. Proost, J. B. M. Kuks, D. C. Van den Tol, and J. M. K. H. Wierda, “Pharmacokinetic/pharmacodynamic modeling of rocuronium in myasthenic patients is improved by taking into account the number of unbound acetylcholine receptors,” Anesthesia and Analgesia, vol. 95, no. 3, pp. 588– 596, 2002.
- Sejvar JJ, Baughman AL, Wise M, Morgan OW. Population incidence of Guillain-Barré syndrome: a systematic review and meta-analysis. Neuroepidemiology 2011; 36:123.
- Rees JH, Soudain SE, Gregson NA, ghghes RA. Campylobacter jejuni infection and Guillain-Barré syndrome. N Engl J Med 1995; 333:1374.
- Baxter R, Bakshi N, Fireman B, et al. Lack of association of Guillain-Barré syndrome with vaccinations. Clin Infect Dis 2013; 57:197.
- Grohskopf, Lisa A.1 Prevention and Control of Seasonal Influenza with Vaccines Recommendations of the Advisory Committee on Immunization Practices — United States, 2016-17 Influenza Season. MMWR Recomm Rep. 2016 Aug 26;65(5):1-54. Ropper AH. The Guillain-Barré syndrome. N Engl J Med 1992; 326:1130.
- Fokke C, van den Berg B, Drenthen J, et al. Diagnosis of Guillain-Barré syndrome and validation of Brighton criteria. Brain 2014; 137:33.
- Asbury AK, Cornblath DR. Assessment of current diagnostic criteria for Guillain-Barré syndrome. Ann Neurol 1990; 27 Suppl:S21.
- Lawn ND, Fletcher DD, Henderson RD, et al. Anticipating mechanical ventilation in Guillain-Barré syndrome. Arch Neurol 2001; 58:893.
- Sharshar T, Chevret S, Bourdain F, et al. Early predictors of mechanical ventilation in Guillain-Barré syndrome. Crit Care Med 2003; 31:278.
- Walgaard C, Lingsma HF, Ruts L, et al. Prediction of respiratory insufficiency in Guillain-Barré syndrome. Ann Neurol 2010; 67:781.
- Rajabally YA, Uncini A. Outcome and its predictors in Guillain-Barre syndrome. J Neurol Neurosurg Psychiatry 2012; 83:711.
- Alonso A, Hernán MA. Temporal trends in the incidence of multiple sclerosis: a systematic review. Neurology 2008; 71:129.
- Koch-Henriksen N, Sørensen PS. The changing demographic pattern of multiple sclerosis epidemiology. Lancet Neurol 2010; 9:520.
- Dunn SE, Steinman L. The gender gap in multiple sclerosis: intersection of science and society. JAMA Neurol 2013; 70:634.
- Polman CH, Reingold SC, Banwell B, et al. Diagnostic criteria for multiple sclerosis: 2010 revisions to the McDonald criteria. Ann Neurol 2011; 69:292.
- Brownlee WJ, Hardy TA, Fazekas F, Miller DH. Diagnosis of multiple sclerosis: progress and challenges. Lancet 2016.
- Galea I, Ward-Abel N, Heesen C. Relapse in multiple sclerosis. BMJ 2015; 35
- Burton A et al, Oral versus intravenous steroids for treatment of relapses in multiple sclerosis. Cochrane Database Syst Rev. 2012;120
- Aul E et al, Oral versus intravenous high-dose methylprednisolone for treatment of relapses in patients with multiple sclerosis (COPOUSEP): a randomised, controlled, double-blind, non-inferiority trial. Lancet 2015