The management of cancer is drastically changing, with many novel medications and therapies emerging. As our approach to malignancy changes, there are a host of new complications that we need to be aware of in the Emergency Department (ED). Traditionally, cancer treatment has involved both Chemotherapy and Radiotherapy:
- Chemotherapy:
- Targets various steps of cell proliferation, such as DNA formation/function or the mitosis spindle
- Commonly given IV in a cyclic schedule
- Causes side effects such as nausea, emesis, hair loss, as well as bone marrow suppression
- Radiation therapy
- Uses controlled high-energy rays to treat tumours and other diseases of the body
- Works by damaging the DNA inside cells making them unable to divide and reproduce
- Abnormal cancer cells are more sensitive to radiation because they divide more quickly than normal cells
ED Physicians are well aware of these treatments and their side effects.
However, treatment of cancer using pharmacological approaches has changed extensively during the past two decades. Long years of basic and clinical research have started a shift from this broad systemic approach to more targeted anti-neoplastic treatments, which eliminate tumour cell populations effectively and with potentially milder side effects.
Checkpoint Inhibitors:
In this post, we will focus on the most commonly used immunotherapeutic agents, called checkpoint inhibitors. Checkpoint inhibitors have revolutionized the treatment of metastatic melanoma but, as we will get into, they are now becoming 2nd line agents for several different types of cancer.
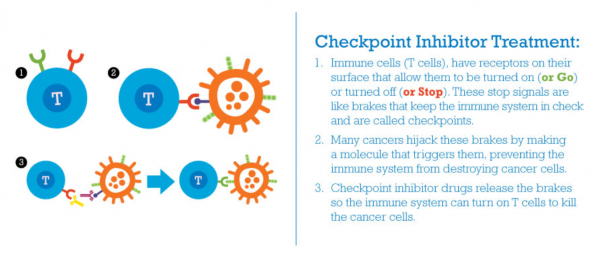
Mechanism of Action
- Tumours often act to evade T cell immune surveillance by activating T cell receptors, essentially turning on the brakes of the T cells
- Checkpoint inhibitors release these brakes allowing the body’s immune system to attack tumour cells
Current Environment
- In the US, checkpoint inhibitors are FDA approved for lymphoma, urothelial bladder cancer, and renal cell carcinoma among others
- In Canada:
- Clinical trials are ongoing in essentially all types of solid tumours as well hematological malignancies
Checkpoint inhibitors are becoming more and more common. They present a novel set of side effects which are quite different from standard cancer treatment complications.
Immune related adverse events (irAEs)
This is essentially the opposite pathophysiology of the usual oncology complications (i.e. neutropenia). It is very common to experience irAEs. According to Horvat et al. (2017), 1/3 of all patients on checkpoint inhibitors required systemic therapy for irAEs.
Checkpoint inhibitors can affect any organ system. The most common complications are:
- Skin-related
- Colitis
- Hepatotoxicity
- Pneumonitis
Ocular, dermatologic, and hepatotoxicity are also potential presentations to be aware of.
The majority of complications are mild but there are several life-threatening presentations which can mimic infectious processes where early administration of steroids will be life-saving.
Importantly, unlike the relatively predictable timelines associated with neutropenia and chemotherapy, the immune complications of checkpoint inhibitors have much more variable timelines. In fact, endocrinologic effects can appear from any point 6 weeks post-treatment and can even occur once treatment has been stopped.
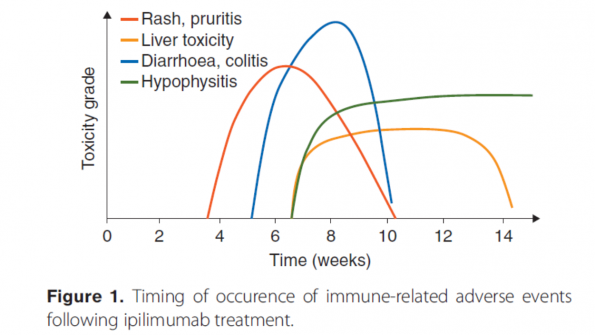
Some cases in particular can mimic other common ED presentations:
(1) Respiratory Distress/Pnuemonitis:
- Occurs in up to 4% of patients receiving checkpoint inhibitor therapy
- In patients presenting with cough or dyspnea, pneumonitis should be considered in the differential along with other potential causes
- Imaging of the chest should be arranged
- In the acute setting, management might need to cover all pathologies
- Moderate to severe symptoms require treatment with high-dose steroids
(2) Colitis:
- Common side effect of checkpoint inhibitors
- Infectious/C-diff work-up should still be sent as well as abdominal imaging as per clinical context
- However, early steroids in this population is critical if considering autoimmune colitis
(3) Endocrine:
- Patients can present with (1) hypo- or (2) hyperthyroid, (3) adrenal insufficiency, and (4) hypophysitis
- These can present at any point after exposure to checkpoint inhibitors
- Consider an autoimmune endocrine cause in any patient previously or currently on checkpoint inhibitors who presents with significant:
- Fatigue
- Headache
- Dizziness
- Dehydration
- Adrenal crisis or shock
- It is crucial not to miss the diagnosis of hypophysitis
- Often presents with non-specific symptoms: tiredness, headache, visual disturbance, nausea/vomiting, and myalgias
- Imaging to exclude brain metastases is required
- Biochemical assessment of pituitary hormones should be sent: TSH, T3, T4, ACTH, and Cortisol
- Symptomatic patients require timely treatment with steroids (and ongoing physiological steroid replacement) and replacement of any other relevant pituitary hormone axis affected
Take home:
- Obtain a good history to ascertain if there is any exposure to Immunotherapy/Checkpoint inhibitors
- Ask yourself if this presentation could be explained by an autoimmune phenomenon
If so:
- Make sure you assess the pituitary axis/thyroid if there is clinical suspicion
- Early steroids can be life-saving and prevent multiorgan failure
- irAEs can occur at variable points post exposure and even after treatment is stopped
Febrile Neutropenia
Febrile neutropenia (FN) is defined as:
- Oral temperature >38.5°C, or
- Oral temperature with two consecutive readings of >38.0°C for 2 h
And
- Absolute neutrophil count <0.5 × 109/L, or
- ANC expected to fall below 0.5 × 109/L
Febrile neutropenia is a very common ED presentation. At our centre, this often involves a work-up and referral to either Oncology or Hematology for admission. Is there a population that may benefit from avoiding inpatient admission?
- IDSA guidelines suggest risk stratifying to decide between IV antibiotics and admission versus PO antibiotics and discharge home with close follow-up
- IDSA divides patients into low risk and high risk:
- High risk: anticipated prolonged (≥7 days) and profound neutropenia (ANC <100 cells/mm3) and/or significant medical co-morbidities
- Low Risk: anticipated brief (<7 days duration) neutropenic periods or no/few comorbidities
Cochrane Review:
- The efficacy of oral (PO) versus intravenous (IV) antibiotic therapy in febrile neutropenic cancer patients was reviewed in 2013
- Common criteria used in the review included:
- Underlying diagnosis not leukemia, no severe soft tissue infection, NKDA, not pregnant, hemodynamic stability, no organ failure, tolerating PO, no pneumonia, no suspicion of central line infection
- Mortality rate was similar when comparing PO to IV antibiotic treatment
- RR 0.95, 95% CI 0.54 to 1.68, 9 trials, 1392 patients, median mortality 0%, range 0% to 8.8%
- Common criteria used in the review included:
Mamtani et al.:
- 2013 in Annals of Emergency Medicine
- Review of three studies of FN patients directly discharged from the ED
- All three studies used different criteria for low risk patients
- Malik et al. (1995)
- Randomized study done in India comparing PO ofloxacin to IV therapy
- 169 total patients were analyzed
- Defined treatment success as initial response to antibiotics, which was identical in both groups
- 3 patients died in the outpatient group
- One was non-compliant with treatment and died of sepsis, one from worsening pneumonia, and one from ICH
- There were 2 deaths in the inpatient group of unclear etiology
- 3 patients died in the outpatient group
- They concluded outpatient Rx was safe.
- Hidago et al. (1999)
- Spanish study comparing IV ceftazidime/amikacin vs. ofloxacin
- Discharged/PO group had a similar antibiotic success rate (treatment failure was defined as need for change of treatment)
- No deaths or major adverse outcomes in the low risk group
- Though 8 patients had to be admitted (3 for positive blood cultures and 5 for treatment failure)
- Elting et al. (2008)
- Retrospective study out of MD Anderson Cancer Center in Houston Texas
- At this centre, they started treating low risk patients as outpatients in the late 1990s and looked at 712 patients classified as low risk as per their algorithm
- Of the 589 who received PO antibiotics as outpatient, 121 had to return to be hospitalized. However, all of the patients responded and there no deaths.
- Malik et al. (1995)
Conclusion:
In select low risk patients, outpatient treatment with PO antibiotics is likely safe. However, these patients required rapid and close follow-up with their cancer specialist and must have the ability to return to the ED promptly. This decision would need to be done in conjunction with their cancer specialist.
Risk Stratification in Febrile Neutropenia
Is there a role for a risk stratification score to find the low risk population of those with febrile neutropenia?
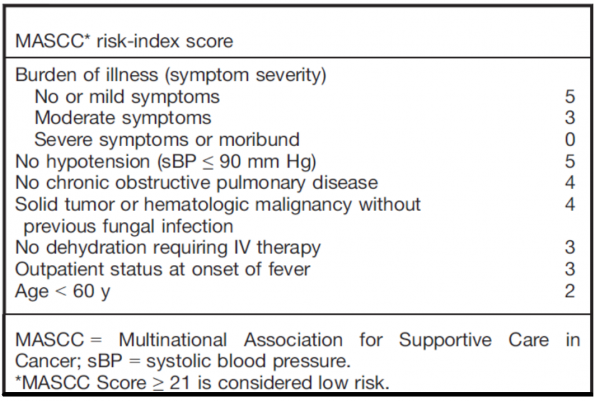
- The MASCC score
- Derived and validated in 2000 rule to determine low risk patients appropriate for outpatient treatment
- Endorsed by the IDSA and by ASCO to guide appropriateness for outpatient treatment
- But:
- Not externally validated
- Sensitivity and specificity actually quite poor (68% and 71%, respectively)
- Baugh et al. (2017)
- Academic Emergency Medicine; studied a tertiary care center in the US affiliated with a large cancer center
- Retrospective chart review examining compliance with the IDSA and ASCO guidelines
- Not surprisingly, found that very few physicians were following the IDSA guidelines
- Guidelines were followed in only 8 of 44 low risk cases
- Coyne et al. (2017)
- Retrospectively compared the MASCC score and the CISNE for stratifying febrile neutropenic patients
- CISNE score performed excellently
- 53 total patients
- The only negative outcome was 1 positive blood culture result
- However, the vast majority of patients were admitted and received IV antibiotics so it is difficult to apply this to our patients
- This group is planning further prospective studies
Conclusion:
As of yet there is no proven clinical risk tool to stratify febrile neutropenic patients, local practice patterns play a significant role in the management of these patients, but individual infectious disease risk stratification is of the greatest utility.
References
- Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer. 2012 Mar 22;12(4):252-64.
- Haanen JBAG et al. ESMO Guidelines Committee. Management of toxicities from immunotherapy: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2017 Jul 1;28(suppl_4):iv119-iv142.
- Weber JS et al. Toxicities of Immunotherapy for the Practitioner. J Clin Oncol. 2015 Jun 20;33(18):2092-9.
- Horvat TZ et al. Immune-Related Adverse Events, Need for Systemic Immunosuppression, and Effects on Survival and Time to Treatment Failure in Patients With Melanoma Treated With Ipilimumab at Memorial Sloan Kettering Cancer Center. J Clin Oncol. 2015 Oct 1;33(28):3193-8.
- Freifeld et al. Clinical practice guideline for the use of antimicrobial agents in neutropenic patients with cancer: 2010 update by the infectious diseases society of america. Infectious Diseases Society of America. Clin Infect Dis. 2011 Feb 15;52(4):e56-93.
- Vidal et al. Oral versus intravenous antibiotic treatment for febrile neutropenia in cancer patients. Cochrane Database Syst Rev. 2013 Oct 9;(10):CD003992.
- Mamtani et al. Can we safely discharge low-risk patients with febrile neutropenia from the emergency department? Ann Emerg Med. 2014 Jan;63(1):48-51.
- Malik et al. Feasibility of outpatient management of fever in cancer patients with low-risk neutropenia: results of a prospective randomized trial. Am J Med. 1995 Mar;98(3):224-31.
- Hidalgo et al. Outpatient therapy with oral ofloxacin for patients with low risk neutropenia and fever: a prospective, randomized clinical trial. Cancer. 1999 Jan 1;85(1):213-9.
- Elting et al. Outcomes and cost of outpatient or inpatient management of 712 patients with febrile neutropenia. J Clin Oncol. 2008 Feb 1;26(4):606-11.
- Klastersky et al. Outpatient oral antibiotics for febrile neutropenic cancer patients using a score predictive for complications. J Clin Oncol. 2006 Sep 1;24(25):4129-34.
- Coyne et al. Application of the MASCC and CISNE Risk-Stratification Scores to Identify Low-Risk Febrile Neutropenic Patients in the Emergency Department. Ann Emerg Med. 2017 Jun;69(6):755-764.
- Bitar RA. Utility of the Multinational Association for Supportive Care in Cancer (MASCC) Risk Index Score as a Criterion for Nonadmission in Febrile Neutropenic Patients with Solid Tumors. Perm J. 2015 Summer;19(3):37-47.
- Baugh et al. Emergency Department Management of Patients with Febrile Neutropenia: Guideline Concordant or Overly Aggressive? Acad Emerg Med. 2017 Jan;24(1):83-91.




