Meningitis is defined as the inflammation of the membranes of the brain and/or spinal cord. The incidence of bacterial meningitis is in the range of 5-10 cases per 100,000 patients per year1. Despite modern medical therapy, it is associated with a case fatality rate of 10-20% and results in unfavourable neurological outcomes in up to 35% of patients2.
This post will delve into new evidence regarding several decision points clinicians often encounter when working up and managing patients with suspected meningitis.
Optimizing the clinical exam for meningitis
Traditionally, we have been taught to examine for Kernig’s and Brudzinski’s signs when examining patients with suspected meningitis.
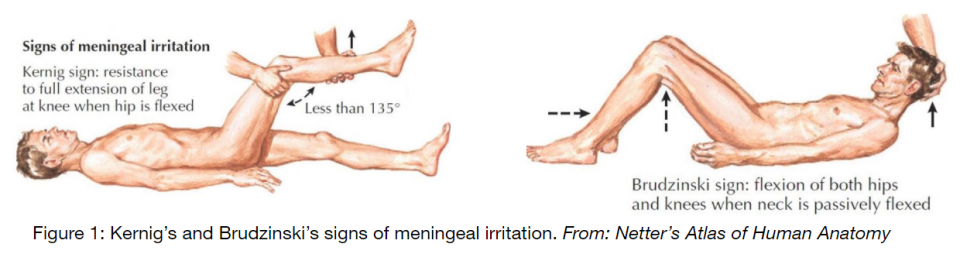
The JAMA rational clinical exam series (1999) highlights the limited supporting evidence and low sensitivity (<10%) of these classic signs3. However, they do recommend testing for jolt accentuation of the headache, defined as the worsening of the patient’s headache as their head is rotated left to right 2-3 times per second. The authors highlighted that a negative jolt accentuation could rule out meningitis at the bedside in non-ill appearing patients. This conclusion was based on the findings of a single cohort study (n = 54) which demonstrated a 97.1% sensitivity and 60% specificity in diagnosing meningitis.
Multiple studies with larger cohorts (n = 118-530) have since examined jolt accentuation, demonstrating sensitivities and specificities ranging from 21-71% and 37-82%, respectively4–6. Considering the poor test characteristics of jolt accentuation in these contemporary studies, it cannot be relied upon to rule out meningitis prior to lumbar puncture.
In a registry study of 696 patients with confirmed bacterial meningitis, van de Beek et al. demonstrated that ≥2 of fever, headache, neck stiffness, and altered mental status were present in 95% of cases2. Unfortunately, this may not be very helpful in ruling out meningitis on clinical grounds, given the limited number of patients in which we consider meningitis with 1 or less of the above characteristics.
Take home points:
- Traditional physical exam maneuvers are unhelpful
- Jolt accentuation of the headache is unhelpful
- Meningitis may be ruled out clinically if the patient complains of ≤1 of:
- Fever
- Headache
- Neck stiffness
- Decreased level of consciousness
Distinguishing aseptic from bacterial meningitis
The incidence of aseptic meningitis is greater than it’s bacterial variant in North America7. Only 66% of patients with aseptic meningitis are diagnosed with a specific pathogen; viruses account for the large majority of these, which include enterovirus, herpes simplex virus 2, and varicella zoster virus8. Non-viral causes of aseptic meningitis must be considered for at-risk patients, including tuberculosis, cryptococcus, partially-treated bacterial meningitis, malignancy, medications (NSAIDs, TMP-SMX, IVIG), and autoimmune diseases. This section will focus on the viral subset of aseptic meningitis.
The clinical presentation of both viral and bacterial meningitis is often indistinguishable. However, alterations in level of consciousness are absent in viral meningitis, which should trigger concern for either bacterial meningitis or encephalitis.
The first step in differentiating bacterial and viral meningitis is the lumbar puncture. Table 1 demonstrates classic CSF findings.
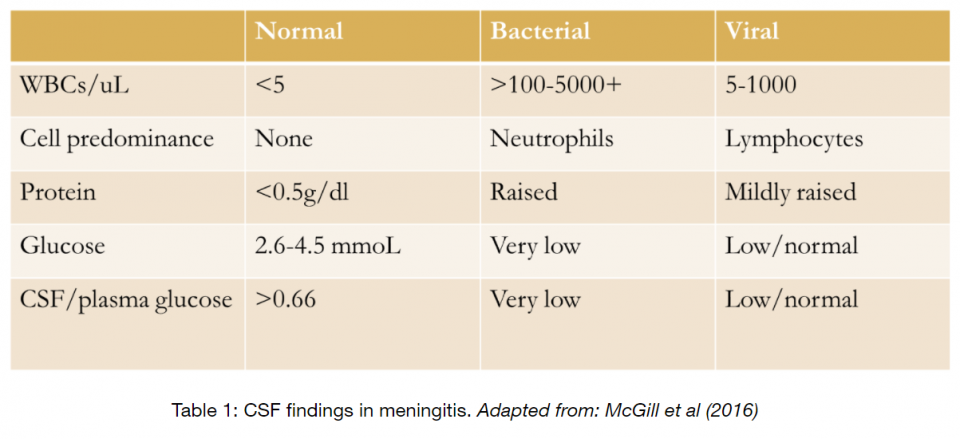
Importantly, there is some overlap in the above table. The CSF:plasma glucose ratio has been found to be highly discriminatory, where a threshold of 0.36 has a 93% sensitivity and specificity for bacterial meningitis9.
Another useful tool is CSF lactate; when applied to patients without antibiotic pre-treatment, a CSF lactate of <35mg/dl (3.9mmol/L) yields a sensitivity and specificity of 93% and 96%, respectively10.
Viral PCR is now considered the gold standard for diagnosing viral meningitis. While this will not be of use to us in the ED given its long turnaround time, international guidelines recommend that the CSF be tested for enterovirus, HSV2, and VZV when viral meningitis is suspected, given that a positive result allows inpatient teams to stop antibiotics and thus shorten hospital stay9.
Treatment options for viral meningitis lack evidence. There are no specific therapies for enterovirus and most patients recover quickly without sequelae7. No RCTs have tested antiviral medications (eg. acyclovir) in patients with HSV2 or VZV meningitis; however, outcomes are excellent regardless of treatment11.
Is there a population in which we can avoid performing an LP entirely? This is tempting in the well-appearing febrile patient presenting with a headache-predominant viral illness and neck stiffness. Unfortunately, there is currently no evidence to justify this practice.
For further information on LP technique and CSF interpretation see this article from JAMA’s Rational Clincal Exam Series
Take home points
-
- Viral meningitis is common and benign
- Consider other causes of aseptic meningitis for at-risk patients
- Consider CSF lactate in your workup
- Save an extra vial of CSF for viral studies
- Treatment is supportive for viral meningitis
- We do not yet know of a subgroup in which we can safely avoid LP
- Viral meningitis is common and benign
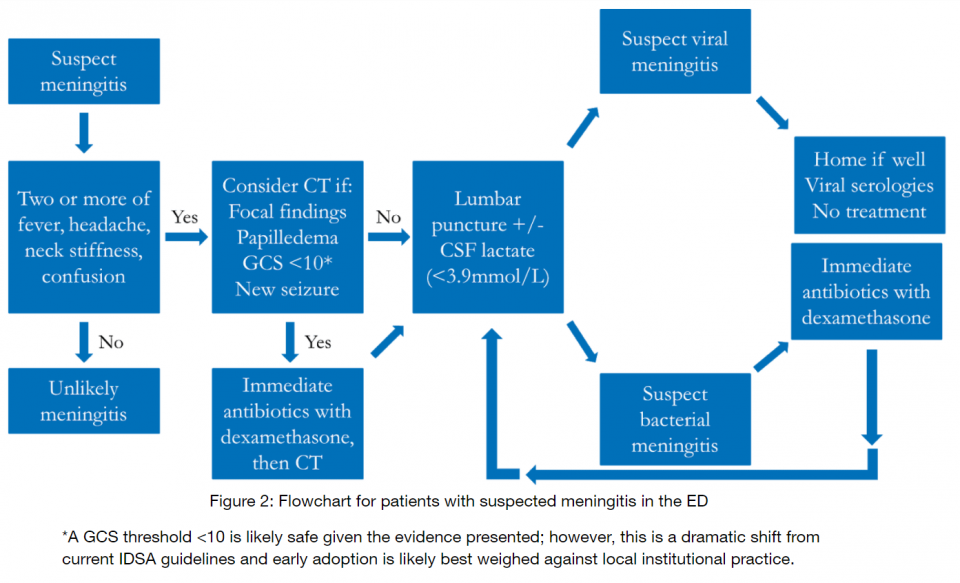
The rational use of CT prior to lumbar puncture in meningitis
We have historically been preoccupied with identifying which patients with suspected meningitis may have an intracranial mass lesion, thus putting them at risk for post-lumbar puncture herniation. This has largely been driven by several case reports finding a temporal link between LP and herniation12. The current IDSA guidelines were based on a 2001 study which derived clinical criteria associated with any CT head abnormality13.
The concern with over-reliance on CT is that patients often do not receive antibiotics prior to their CT scan. This practice has been associated with delays in therapy and an associated increase in mortality14.
Acknowledging that a CT head abnormality alone does not reliably predict herniation and the known effect on antimicrobial treatment delays, Swedish guidelines were changed in 20091. New onset seizure, immunosuppression, and moderate-severely impaired mental status (i.e. not actively herniating) were removed as indications for CT prior to LP. Following this intervention, Glimåker et al. found that antibiotics were given 1.18 hrs earlier, fewer patients suffered long-term neurological sequelae, and mortality dropped from 11.7% to 6.9%, although the latter effect was no longer significant when accounting for confounders1.
In a subsequent study, the same group reviewed the outcomes of patients with bacterial meningitis who had undergone a CT prior to LP with adherence to the Swedish guidelines versus both European and IDSA guidelines. Those who were worked up with adherence to the Swedish guidelines had a lower mortality (aOR 0.48) and more favourable neurological outcome (aOR 1.52) than the remainder of the cohort15. Undergoing an LP prior to CT was associated with the greatest mortality benefit (aOR 0.38); this benefit persisted regardless of the state of immunosuppression or level of consciousness.
Take home points
-
- CT delays antimicrobial therapy and worsens outcomes
- Give antibiotics prior to CT when imaging before LP
- Consider prompt LP for immunocompromised patients and those with a decreased level of consciousness
A brief review of the best-supported treatment in bacterial meningitis
Once the diagnosis of bacterial meningitis is established, or if suspected and the patient is sent first to CT, antibiotics must be administered as soon as possible. Early antimicrobial therapy is associated with a mortality benefit and reduced incidence of unfavourable outcomes14,16. Current North American adult guidelines recommend a 3rd generation cephalosporin with vancomycin; the addition of ampicillin is indicated for patients who are aged >50, who are immunocompromised, and for alcoholics12.
The use of dexamethasone (10mg IV) given concurrently with antibiotics is supported by a 2015 Cochrane review which demonstrated a lower incidence of hearing loss (ARR 5.2%), short-term neurological sequelae (ARR 3.7%), mortality in patients with S.pneumoniae (ARR 6.1%), and hearing loss in children with H.influenzae (ARR 8%). These benefits were present only in high-income countries.
Part 4: Take home points
-
- Administer early empiric antibiotics
- Immediately in patients requiring resuscitation; LP when stable
- Concurrent dexamethasone 10mg IV
- Administer early empiric antibiotics
Summary:
References:
- Glimaker M, Johansson B, Grindborg Ö, Bottai M, Lindquist L, Sjölin J. Adult bacterial meningitis: Earlier treatment and improved outcome following guideline revision promoting prompt lumbar puncture. Clin Infect Dis. 2015;60(8):1162-1169. doi:10.1093/cid/civ011
- van de Beek D, de Gans J, Spanjaard L, Weisfelt M, Reitsma JB, Vermeulen M. Clinical features and prognostic factors in adults with bacterial meningitis. N Engl J Med. 2004;351(18):1849-1859. doi:10.1056/NEJMoa040845
- Attia J, Hatala R, Cook DJ, Wong JG. The rational clinical examination. Does this adult patient have acute meningitis? Jama. 1999;282(2):175-181. doi:10.1001/jama.282.2.175
- Nakao JH, Jafri FN, Shah K, Newman DH. Jolt accentuation of headache and other clinical signs: Poor predictors of meningitis in adults. Am J Emerg Med. 2014;32(1):24-28. doi:10.1016/j.ajem.2013.09.012
- Tamune H, Takeya H, Suzuki W, Tagashira Y, Kuki T, Nakamura M. Absence of jolt accentuation of headache cannot accurately rule out meningitis in adults. Am J Emerg Med. 2013;31(11):1601-1604. doi:10.1016/j.ajem.2013.08.028
- Sato R, Kuriyama A, Luthe SK. Can We Rule Out Meningitis from Negative Jolt Accentuation? A Retrospective Cohort Study. Headache. 2017;57(4):586-592. doi:10.1111/head.13022
- McGill F, Griffiths MJ, Solomon T. Viral meningitis. Curr Opin Infect Dis. 2017;30(2):248-256. doi:10.1097/QCO.0000000000000355
- Kupila L, Vuorinen T, Vainionpää R, Hukkanen V, Marttila RJ, Kotilainen P. Etiology of aseptic meningitis and encephalitis in an adult population. Neurology. 2006;66(1):75-80. doi:10.1212/01.wnl.0000191407.81333.00
- McGill F, Heyderman RS, Michael BD, et al. The UK joint specialist societies guideline on the diagnosis and management of acute meningitis and meningococcal sepsis in immunocompetent adults. J Infect. 2016;72(4):405-438. doi:10.1016/j.jinf.2016.01.007
- Sakushima K, Hayashino Y, Kawaguchi T, Jackson JL, Fukuhara S. Diagnostic accuracy of cerebrospinal fluid lactate for differentiating bacterial meningitis from aseptic meningitis: A meta-analysis. J Infect. 2011;62(4):255-262. doi:10.1016/j.jinf.2011.02.010
- Kaewpoowat Q, Salazar L, Aguilera E, Wootton SH, Hasbun R. Herpes simplex and varicella zoster CNS infections: clinical presentations, treatments and outcomes. Infection. 2016;44(3):337-345. doi:10.1007/s15010-015-0867-6
- Tunkel AR, Hartman BJ, Kaplan SL, et al. Practice guidelines for the management of bacterial meningitis. Clin Infect Dis. 2004;39(9):1267-1284. doi:10.1086/425368
- Hasbun R. Computed tomography of the head before a lumbar puncture in suspected meningitis — is it helpful? N Engl J Med. 2001;345(24):1768-1770. doi:10.1056/NEJM200112133452410
- Proulx N, Fréchette D, Toye B, Chan J, Kravcik S. Delays in the administration of antibiotics are associated with mortality from adult acute bacterial meningitis. QJM – Mon J Assoc Physicians. 2005;98(4):291-298. doi:10.1093/qjmed/hci047
- Glimåker M, Sjölin J, Åkesson S, Naucler P. Lumbar Puncture Performed Promptly or After Neuroimaging in Acute Bacterial Meningitis in Adults: A Prospective National Cohort Study Evaluating Different Guidelines. Clin Infect Dis. 2017;66(January):321-328. doi:10.1093/cid/cix806
- Bodilsen J, Dalager-Pedersen M, Schønheyder HC, Nielsen H. Time to antibiotic therapy and outcome in bacterial meningitis: A Danish population-based cohort study. BMC Infect Dis. 2016;16(1):1-7. doi:10.1186/s12879-016-1711-z




Trackbacks/Pingbacks