Methodology: 3.5/5
Usefulness: 4/5
Albers GW, et al. N Engl J Med. 2018 Jan 24.
Journal Club Bottom Line
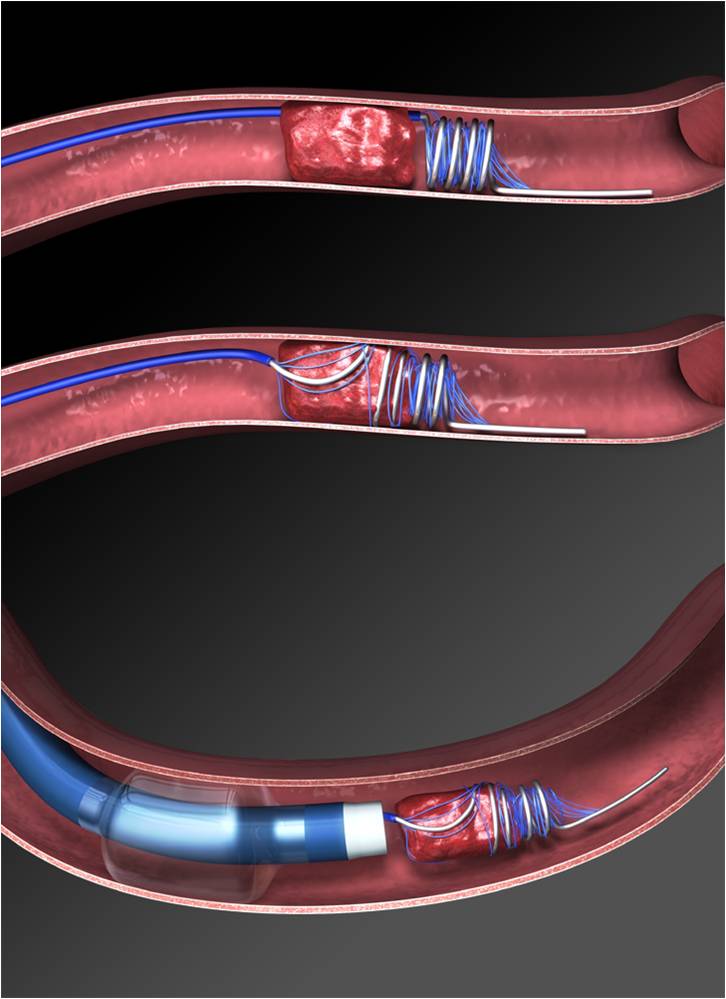
Question/Methods: This was a multicentre, prospective, randomized, open label, blinded outcome trial comparing thrombectomy + medical therapy vs medical therapy alone in patients with delayed presentation (6-16 hours) of proximal MCA & ICA strokes with a small infarct to penumbra ratio on perfusion imaging.
Findings: The endovascular group achieved better functional outcomes evidenced by a favourable shift in median modified Rankin Scale (mRS) scores at 90 days (OR 2.77, P<0.001). Thrombectomy patients were also more likely to achieve functional independence (mRS 0-2) (NNT=3.58); as well as decreased mortality (NNT=8.75) at 90 days.
Limitations: This trial was stopped earlier than the 1st planned interim analysis at the request of an independent review board when the results of the DAWN trial were published. The efficacy boundary used for termination could have been more conservative in order to minimize the chances of type I error. As a result of early cessation, the sample size was also small (N=182).
Interpretation: Patients with proximal MCA and ICA strokes and favourable perfusion imaging have better functional outcomes and decreased mortality at 90 days when treated with thrombectomy + medical therapy (vs medical therapy alone) at an extended treatment window of 6-16 hours. These finding have already influenced the new 2018 AHA stroke guidelines.
Epi lesson
Stopping Clinical Trials after Interim Analyses
Interim analyses are commonly planned in large studies. The overwhelming majority of researchers and clinicians discourage the concept of stopping clinical trial based on interim analysis. Studies that were ‘negative’ during interim analysis have later turned out to be positive and vice-versa. Using interim analysis increases the chance of type-1 error (concluding that there is treatment effect when there is none; false positive) in clinical studies. There are approaches to reduce type 1 error: O’Brien-Fleming, Haybittle-Peto, or Pocock. Essential each one of them increase the threshold of statistical significance (i.e. p-value must be much lower than 0.05) during the interim analysis for stopping the study.