Conversion Disorder Functional Neurological Disorder is not a diagnosis of exclusion. It is a diagnosis based on positive clinical features.
Definitions:
- Medically Unexplained Physical Symptoms (MUPS)
- Patient complaints and symptoms for which no medical etiology is identified
- Somatic Symptom Disorder
- Somatic symptoms that are distressing to a patient for at least 6 months
- Importantly, patients have disproportionate and excessive thoughts and behaviours towards these symptoms
- Functional Symptom
- Another term for MUPS commonly used in all specialties
- Every specialty sees patients with functional symptoms, for example:
- Cardiology – non-cardiac chest pain
- Gastroenterology – IBS
- Rheumatology – fibromyalgia
- Functional Neurological Disorder (FND)
- Previously conversion disorder
- Term for a constellation of functional symptoms that are neurologic in nature
- DSM 5 updated criteria will be explained below
Epidemiology:
- Carson, Handb Clin Neuro, 2016
- 60-75% Women to men
- Average age 35-50 years
- Williams, J Psychosom Res, 2001
- 1 in every 25 ED visits is somatization
- Stone, Brain, 2009
- 30% of patients had symptoms not fully explained by their diagnosed condition
- 4% prevalence of functional neurological disorder
- Kroenke, Am J Med, 1989
- Retrospective review of 1,000 patients with new complaints
- Etiology identified in only 16%
- Carson, J Neuro Neurosurg Psych, 2011
- Multi-centre prospective cohort study of 3781 patients showed that compared to patients with an etiology, patients with unexplained symptoms had:
- Worse physical health
- Higher anxiety
- Increased rates of unemployment for health reasons
- Increased rates of receiving disability
- Multi-centre prospective cohort study of 3781 patients showed that compared to patients with an etiology, patients with unexplained symptoms had:
- Reuber, Ann Neurol, 2003
- Prognosis for psychogenic non-epileptic seizure (PNES) is worse than epilepsy
- Higher rates of spells
- More disability
- Longer duration of illness
- Prognosis for psychogenic non-epileptic seizure (PNES) is worse than epilepsy
- Gelauff, J Neuro Neurosurg Psych, 2014
- Systematic review of 9 studies
- Time from symptom onset to receiving diagnosis was a prognostic factor for recovery
- Important to recognize FND early and as ED physicians we are well-positioned to do so given the high presentation rate
Pathophysiology:
- Vuilleumier, Brain, 2001
- 7 patients prospectively enrolled with functional sensorimotor loss
- Vibrostimulatory stimulus applied and SPECT
- Re-scanned 6 months after recovery
- Findings:
- Decreased cerebral blood flow and hypoactivation of thalamus and basal gang.
- Hypoactivation of basal ganglia and thalamus in FND
- Decreased cerebral blood flow and hypoactivation of thalamus and basal gang.
- Burke, Neuroimage, 2014
- 10 patients with functional sensory loss
- Vibrostimulatory stimulus to affected vs. unaffected limb with fMRI done
- Trend towards lower somatosensory cortex activation
- Higher activation in contralateral paralimbic structure (10 of them)
- These are emotional centres that can regulate integration of neural impulses
- Conclusions:
- Emotional suppression of sensory-integration stimuli in FND
- Parees, Brain, 2014
- 14 patients and 14 controls
- Background:
- Sensory attenuation is a phenomenon of experiencing self-generated stimuli with less intensity than externally generated stimuli
- Example: you can’t tickle yourself but someone else can tickle you easily
- Question: do patients with sensory FND have impaired sensory attenuation?
- Sensory attenuation is a phenomenon of experiencing self-generated stimuli with less intensity than externally generated stimuli
- Force-matching experiment
- Classic experiment for this
- Robot puts pressure on participant’s finger. Immediately after patient needs to match the pressure that they felt, on their own finger (a thin pressure probe checks the pressure)
- Expected:
- Due to sensory attenuation, participants apply more force than the robot (they experience their own force as less)
- Results:
- Functional sensory loss patients were more accurate
- Intriguing as it is easier to fake inaccuracy than it is to fake accuracy
- Conclusions:
- Impaired sensory attenuation in patients with FND
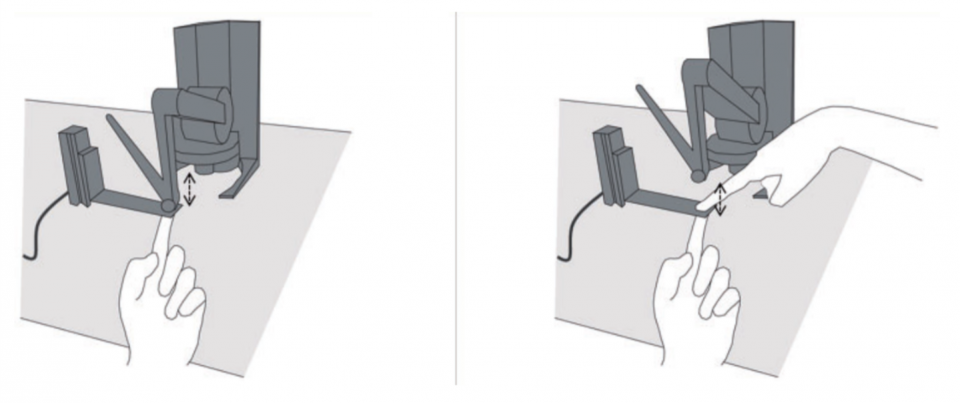
- Parees, Brain, 2012
- 10 patients with functional tremor, 8 patients with Parkinsonian tremor
- Wore actigraph (device that measures amount of time with tremor)
- In spite of knowing that the objective was to see how they could estimate what proportion of time they had a tremor:
- Parkinson’s overestimated by 23%
- Functional overestimated by 80%
- Conclusions:
- Impaired attention in FND
- Whenever they attended or perceived their hand they thought it was tremorous, even if this was a small percentage of the time
- Impaired attention in FND
- Limitations of all studies
- Small case-control studies
- Heterogeneity in types of FND
- Larger studies needed for validation and replication
Conclusions
- Pathophysiological basis of FND likely includes the following:
- Hypoactivation of basal ganglia
- Emotional suppression by paralimbic structures
- Loss of sensory attenuation
- Alterations of attention and perception
Differences with malingerers
- Prevalence of malingering in MUPS is around 1%
- Studies show different brain activation in FND vs. feigners
- FND: less activation of left dorso-lateral prefrontal cortex which is involved in the generation of action, choice or planning of movement
- Evidence that FND is different from malingerers:
- Homogeneity of symptoms
- Persistence over time
- High frequency during video EEG even though told diagnosis is functional
- Persistence after shown maneuvers
- Historical consistency
- Red flags for malingering in the ED:
- Inconsistency in patient’s history
- Inconsistency from patient’s history to collateral history
- Inconsistency between patient’s history and medical records
- Inconsistency in patient throughout multiple encounters
- Spectrum:
- Low intent of symptoms in FND
- Malingering = external gain
- Factitious = internal gain
Diagnosis in the ED:
- DSM 5 criteria:
- One or more symptoms of altered motor or sensory function
- Clinical findings provide evidence of incompatibility between the symptom and recognized neurological or medical conditions
- The symptom or deficit is not better explained by another medical or mental disorder
- The symptom or deficit causes clinically significant distress or impairment
- Malingering is not a criterion
- No requirement to identify a psychosocial stressor
- Key for us in the ED as in our brief encounters we might not identify a stressor
Clinical signs
- Motor
- Hoover
- Sens 63, Spec 100
- Applies only to proximal leg weakness
- Hoover
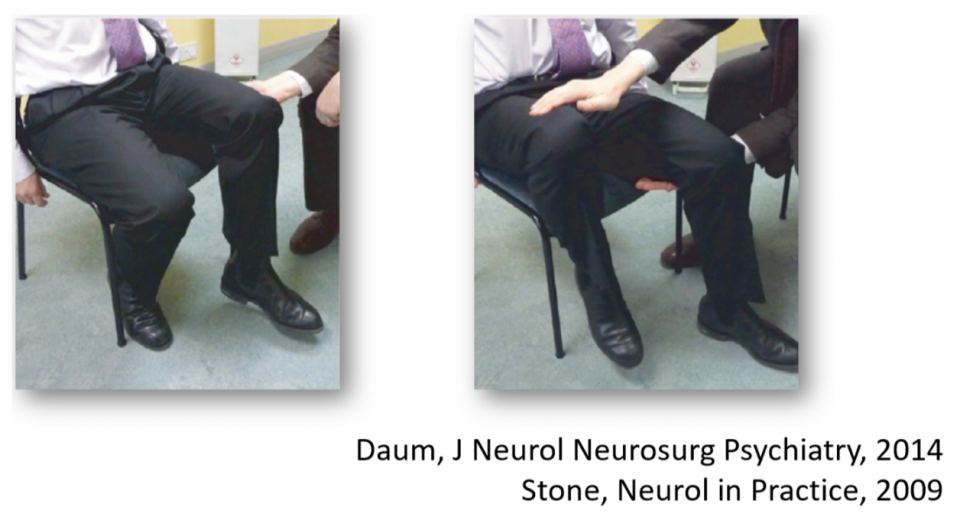
- Give way weakness
- Sens 69, Spec 98
- Caution in:
- Pain
- Language barrier
- Cognitive barriers for patient not able to follow commands
- Neglect (eg. Stroke)
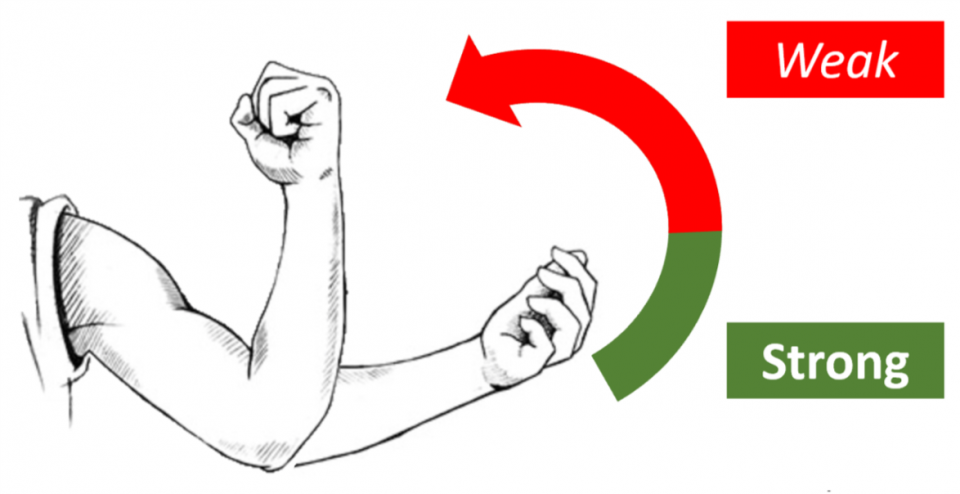
- Motor inconsistencies
- Sens 13, Spec 98
- Example:
- Weakness of plantar flexion at bedside, but able to ambulate on tip-toes
- Pathology that can rarely mimic it:
- Apraxias
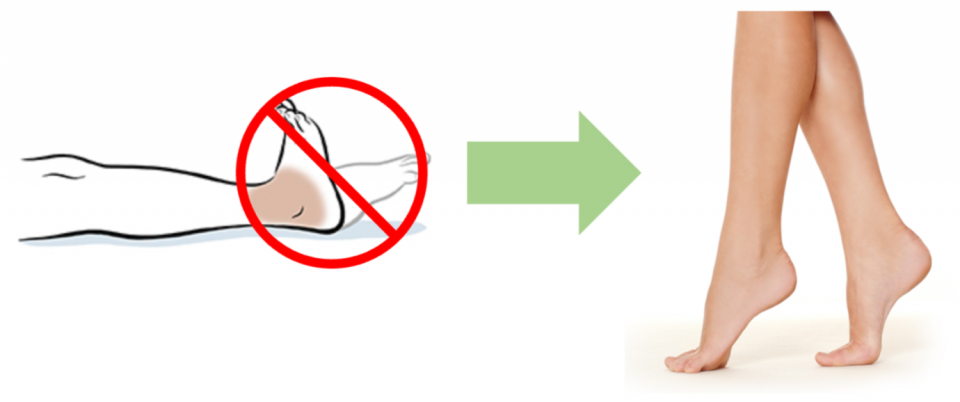
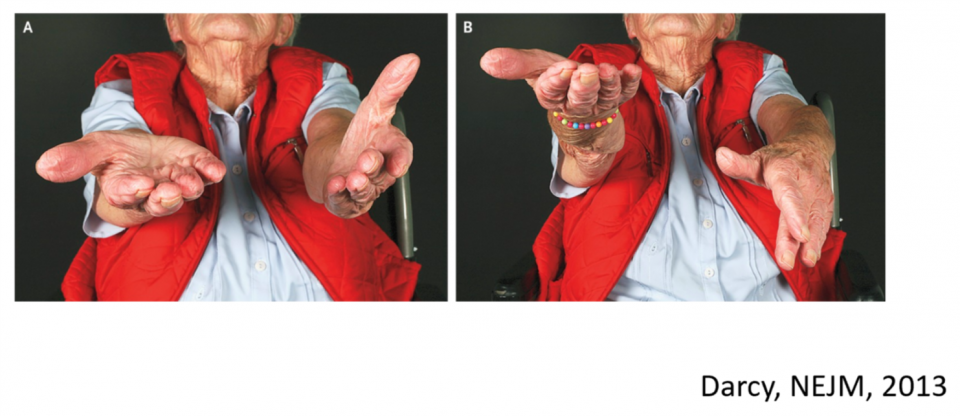
- Pronator drift
- Used by expert consensus (no sens/spec available for FND)
- Expected in upper motor weakness: drift of limb and pronation
- Observations in functional weakness: isolated drift with no pronation
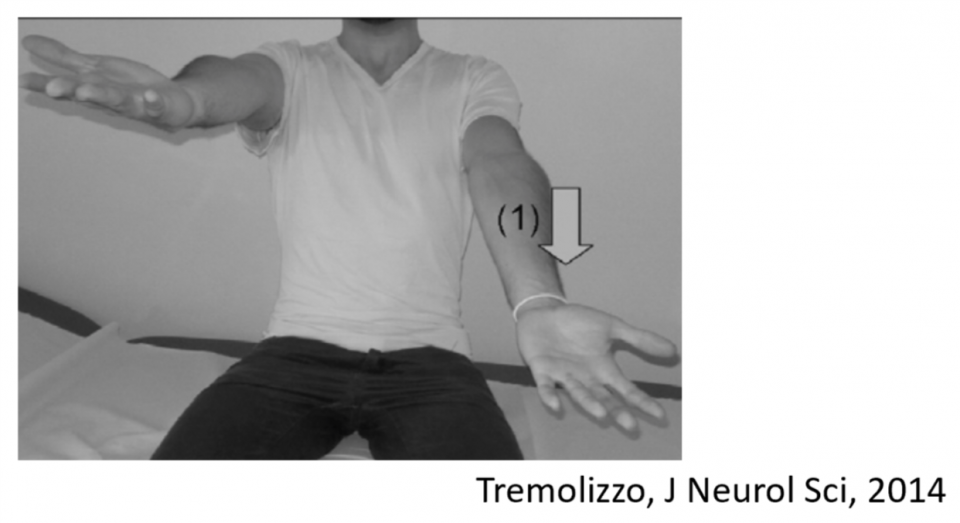
- Tremor entrainment
- Used by expert consensus (no sens/spec available for FND)
- Historical clues for functional tremor:
- Abrupt onset
- Associated with psychosocial stressor
- Onset after injury
- Sign:
- Patient to copy movement generated by examiner with ONE hand (both arms extended)
- Important for patient to copy movement rather than generate it on his/her own
- Contralateral hand (ie. Hand that is NOT copying movement) shows one of:
- Acquires frequency/movement of movement patient is copying and performing with other hand
- Tremor stops
- Unable to perform test
- Patient to copy movement generated by examiner with ONE hand (both arms extended)
- Sensory
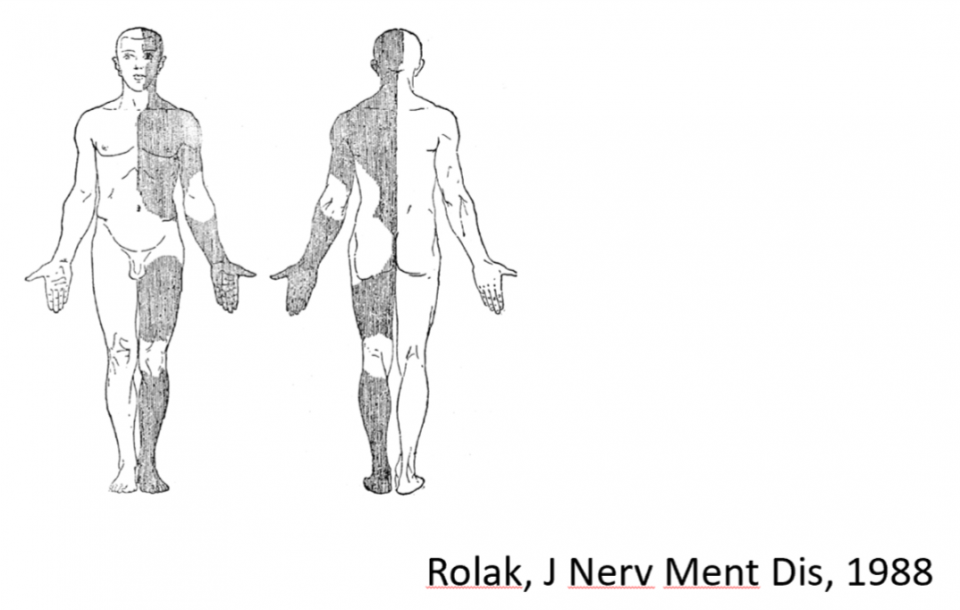
- Non-anatomical sensory loss
- Sens 70, Spec 100
- Caution is required as this can be mimicked by multiple concurrent processes or MS
- Non-anatomical sensory loss
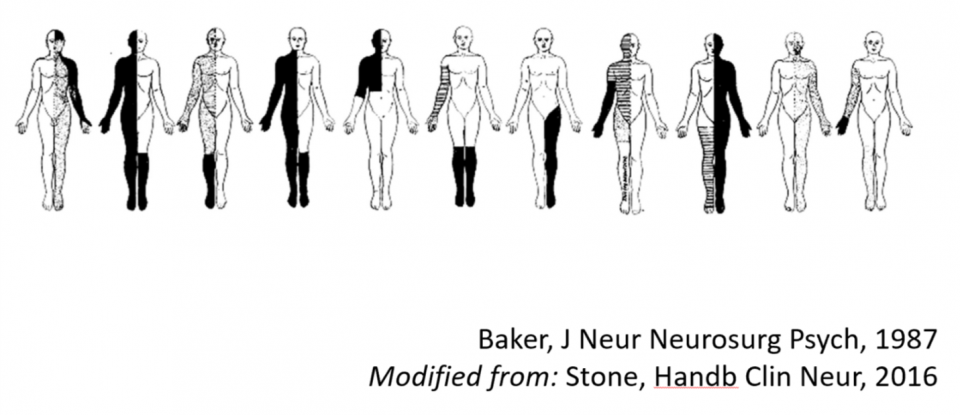
- Midline splitting = sensory defects that stop at midline
- Sense 20, Spec 93
- Caution as this can be mimicked by lacunar strokes
2. Gait
- Dragging monoplegic leg
- Sens 8.4, Spec 100
- Physiologic hemiplegic gait = circumduction of hip
- Functional monoplegic gait = dragging leg behind body
- Huffing and puffing sign
- Sens 8.4, Spec 100
- Patients seen “huffing and puffing” and over-exerting in attempt to generate movement. Rare in physiologic weakness as patients are just unable to move
3. La Belle Indifference – is it useful?
- Stone, Br J Psychiatry, 2006
- Systematic review of 11 studies (356 functional patients, 157 organic patients)
- Prevalence the same in both groups (21 vs. 29%)
- No discriminatory value
Limitations
- No high-quality studies
- Selection bias, non-consecutive sampling, high heterogeneity
- Lack of blinding
- Use of clinical sign as component of diagnosis
Summary for clinical signs used to raise suspicion of FND
- Motor:
- Hoover sign
- Give-way weakness
- Motor inconsistencies
- Drift without pronation
- Tremor entrainment
- Sensory – caution if in isolation:
- Non-anatomical sensory loss
- Gait:
- Dragging monoplegic leg
- “Huffing and Puffing”
Treatment and Management in the ED:
Summary
- Disclose the diagnosis
- Avoid investigations and admissions if possible
- Refer to outpatient neurology
Disclosure of diagnosis
- Elements of an evidence-based therapeutic disclosure:
- Explain:
- What they do have
- “You have functional weakness,” or “this is functional sensory loss that you are experiencing”
- The mechanism of the symptoms
- Use analogies:
- “Think of your brain like a computer. The hardware is intact and not damaged, the problem lies in its software”
- Use analogies:
- What they do have
- Explain:

- How you made the diagnosis
- Show them the signs (eg. Hoover sign, give-way weakness, etc.)
- “Can you see how when you are not attending to your leg it’s working fine? This tells me that the your nervous system is intact and there’s no damage – but your body is having difficulty sending messages to your leg and improves with distraction”
- “Can you see how when you are distracted your tremor disappears?”
- You believe them
- Reversibility
- “Because your nervous system is not damaged, the problem is entirely reversible”
- Show them the signs (eg. Hoover sign, give-way weakness, etc.)
- Common pitfalls in disclosing the diagnosis:
- Telling them what they don’t have and expecting them to be satisfied
- They will be wondering what they do have
- They came to the ED seeking a diagnosis because of the distress their problem is causing them
- Preemptively attributing their symptoms to psychological problems
- While stress is a major contributor to most patients with FND, they might be unaware of stressors or psychological burden
- Without having the time and relationship to effectively tease this out, you risk losing their trust in your diagnosis
- Using the “negative workup” as the evidence for their diagnosis
- As Dr. Stone put it in his interview from 2015:
- “It’s much more persuasive for patients to know that they have a typical feature of a common condition, than to be told ‘well all the tests are normal’. That explanation usually is greeted with a sort of ‘well, perhaps they just haven’t done all the tests.”
- As Dr. Stone put it in his interview from 2015:
- Telling them what they don’t have and expecting them to be satisfied
- Resources after disclosing:
- Provide patients with neurosymptoms.org
- Written by a functional specialist
- Patient-centered language
- Covers almost all functional disorders for more patient info
- Provide patients with neurosymptoms.org
Avoiding investigations:
- Patients with FND are typically over investigated (Hatcher, BMJ, 2008)
- Example:
- Wei, J, Epilepsy Behav, 2018:
- Retrospective review of 224 patients with PNES
- 66% received brain imaging before diagnosis
- 1 in 5 received at least 4 CT or MRIs before receiving diagnosis
- Wei, J, Epilepsy Behav, 2018:
- Example:
- Investigations are generally not anxiolytic
- Example:
- Howard, J Neurol Neurosurg Psych, 2005:
- 150 patients with chronic daily headache
- Stratified by HADS + vs. –, ITT analysis
- Minimal crossover (93% vs 4%)
- No difference in anxiety at 1 year
- Only difference at 1 year: fewer referrals if MRI offered
- Howard, J Neurol Neurosurg Psych, 2005:
- Example:
Referral to neurologist:
- Local opinion:
- “Every [patient with FND] should be referred to a neurologist. However, the timing of the consult does not have to be in the ED and most often is best for it not to be in the ED.” C. De Meulemeester, The Ottawa Hospital
- Goals of referral:
- Confirm diagnosis
- Arrange therapy
- Refer to psychiatry
- Identify concurrent neurological disorders
- Importance of referral:
- High degree of organic disease in patients with FND
- FND is a red flag for underlying neurologic disease
- Conversely, organic neurologic disease is a risk factor for FND
- Stone, J, Neurol, 2012
- Scottish study of 2467 outpatient neurology patients
- Proven neurological disease
- Concomitant functional symptom in 1 in 8 of these patients
- Stone, J, Neurol, 2012
- Examples of therapies supported by RCTs that neurologists can help arrange:
- Physiotherapy
- CBT
+/- Referral to ED psychiatry if:
- Multiple recurrent visits
- Need to establish care-plan
- Suspect factitious disorder or malingering
Psychogenic Non-Epileptic Seizures:
- Last year 905 ED visits to TOH for seizures
- More than 2/3 had diagnosis of “unspecified convulsions” – term used also when suspicion for PNES but no confirmation of diagnosis
- Local experts suggest 30% of our outpatient elective epilepsy monitoring unit admissions have diagnosis of PNES
- Consistent with literature
- ED accuracy in recognizing epileptic vs. non-epileptic seizures
- Wasserman, Epilepsy Behav, 2017
- 44% accuracy
- Important to be precise as disclosing PNES diagnosis carries benefits:
- 40% cessation of seizures immediately after receiving diagnosis (Aboukasm, Epilepsia, 1998)
- 39% fewer ED visits (Jorsch, Epilepsy & Behav, 2011)
- Wasserman, Epilepsy Behav, 2017
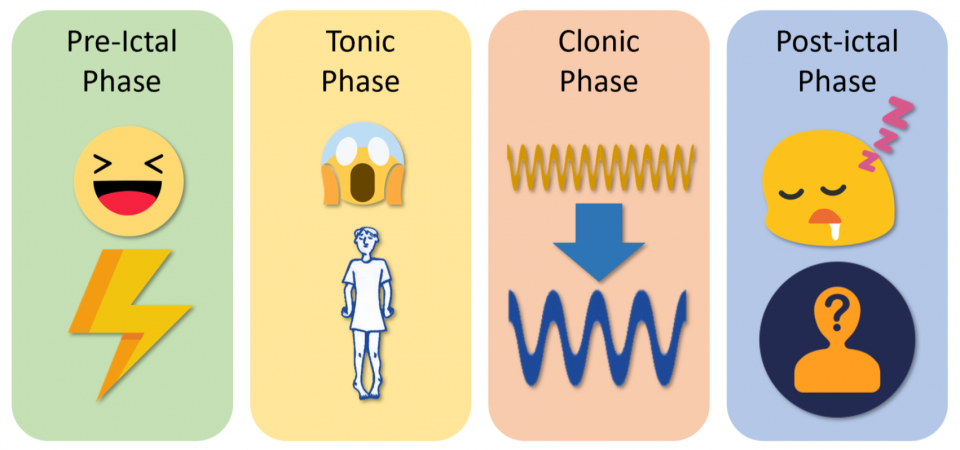
- Recognizing PNES
- Most PNES mimic generalized tonic-clonic (GTC) seizures
- Recognize PNES by understanding all features and nuances of prototypical GTC seizures and identifying deviations
- Prototypical GTC seizure:
- Pre-ictal phase
- Patient normal
- Tonic phase
- Onset is abrupt
- Epileptic cry
- Posturing – predominantly extensors over flexors
- Clonic phase
- Gradual transition to clonic phase
- Initially high frequency, low amplitude convulsions
- Gradual transition to low frequency, high amplitude convulsions
- Post-ictal phase
- Stertorous breathing
- Confusion
- Amnesia
- Pre-ictal phase
- Prototypical GTC seizure:
- Findings (LaFrance, Epilepsia, 2013; Erba, Seizure, 2017):
- Validated, favouring epileptic seizures:
- Stertorous breathing
- Sens 61-91, Spec 100
- Post-ictal confusion
- Sens 67, Spec 84
- Stertorous breathing
- Validated, favouring PNES:
- Asynchronous movements
- Sens 44-96, Spec 93-100
- Pelvic thrusting
- Sens 1-44, Spec 92-100
- Side-to-side movements
- Sens 15-63, Spec 92-100
- Closed eyes
- Sens 34-96, Spec 74-100
- Ictal crying
- Sens 13-37, Spec 100
- Asynchronous movements
- Validated, favouring epileptic seizures:
- Other findings used by expert consensus (Chen, Continuum, 2016):
- Favouring epileptic seizures:
- Impaired corneal reflex
- Extensor plantar reflexes
- Gaze deviation
- Away from epileptic focus during ictus
- Towards epileptic focus during post-ictal phase (due to hypoactivation)
- Favouring PNES:
- Self-protection or guarding with arm drop over face
- Visual fixation (can be tested with mirror)
- Increases or suppresses with suggestibility
- Fluctuating course (e.g. fluctuation in clonus frequency and amplitudes)
- Absence of tonic phase
- Responding to examiner
- Favouring epileptic seizures:
- Caveats:
- Large spectrum of how PNES and epileptic seizures can present
- Eg:
- Frontal seizures can present with bizarre behaviour
- Focal seizures can retain consciousness
- PNES subtypes include focal mimickers
- Eg:
- Gold-standard diagnosis and confirmation is with video EEG
- Large spectrum of how PNES and epileptic seizures can present
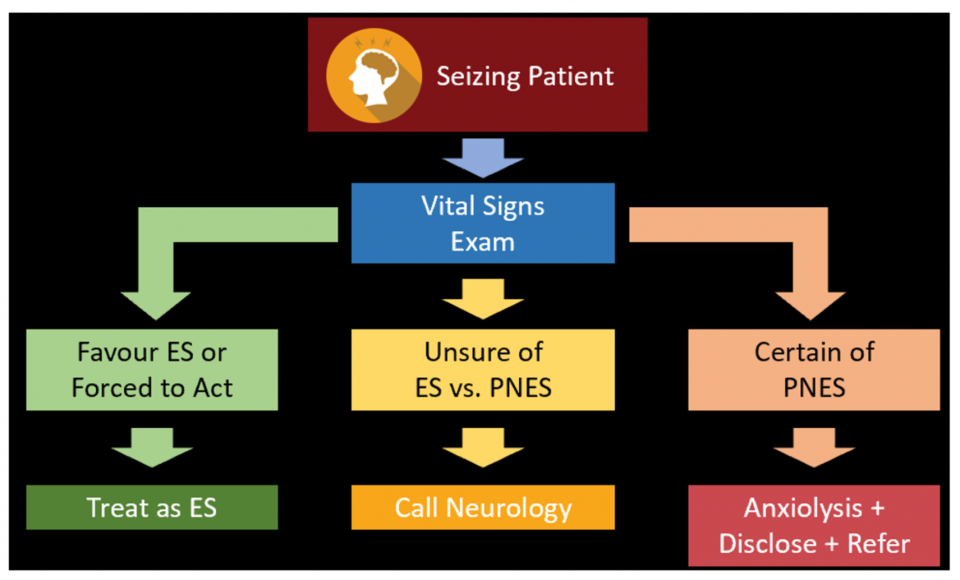
- Approach to patient with prolonged seizure:
- If VS abnormal (i.e. hypoxia) or clearly epileptic, then treat as epileptic
- If VS normal and clearly PNES: then reassure, disclose diagnosis, and refer for gold standard video EEG
- If VS normal and unsure as to whether epileptic or PNES: then call neurology for opinion +/- ED EEG
References
1. Winter A. Somatoform Disorders. Rosen’s Emergency Medicine: Concepts and Clinical Practice, 9th ed. 2017; Ch. 103: 1359.
2. Sharpe M, Mayou R, Seagroatt V et al. Why do doctors find some patients difficult to help? Quarterly journal of medicine. 1994; 87.
3. Carson A, and Lehn A. Epidemiology. Handbook of clinical neurology. 2016; 139-3rd series. Ch. 5: 48-51.
4. Kroenke K, and Mangelsdorff A. Common symptoms in ambulatory care: incidence, evaluation, therapy, and outcome. American journal of medicine. 1989; 86(3).
5. Williams E, Guthrie E, Mackway-Jones K et al. Psychiatric status, somatisation, and health care utilization of frequent attenders at the emergency department – a comparison with routine attenders. Journal of psychosomatic research. 2001; 50.
6. Stone J, Carson A, Duncan R et al. Symptoms ‘unexplained by organic disease’ in 1144 new neurology out-patients: how often does the diagnosis change at follow-up? Brain. 2009; 132.
7. Wei D, Garlinghouse M, Li W, Swingle N, Samson K, and Taraschenko O. Utilization of brain imaging in evaluating patients with psychogenic nonepileptic spells. Epilepsy and Behavior. 2018; 85.
8. Gelauff J, Stone J, Edwards M, and Carson A. The prognosis of functional (psychogenic) motor symptoms: a systematic review. Journal of neurology, neurosurgery, and psychiatry. 2014; 85.
9. Carson A, Stone J, Hibberd C et al. Disability, distress and unemployment in neurology outpatients with symptoms ‘unexplained by organic disease’. Journal of neurology, neurosurgery, and psychiatry. 2011; 82(7).
10. Rueber M, Pukrop R, Bauer J et al. Outcome in psychogenic nonepileptic seizures: 1 to 10-year follow-up in 164 patients. Annals of Neurology. 2003; 53.
11. Burke M, Ghaffar O, Staines W et al. Functional neuroimaging of conversion disorder: the role of ancillary activation. Neuroimage: Clinical. 2014.
12. Vuilleumier P, Chicherio C, Assal F et al. Functional neuroanatomical correlates of hysterical sensorimotor loss. Brain. 2001; 124.
13. Parees I, Brown H, Nuruki A et al. loss of sensory attenuation in patients with functional (psychogenic) movement disorders. Brain. 2014; 137.
14. Parees I, Saifee T, Kassavetis P et al. Believing is perceiving: mismatch between self-report and actigraphy in psychogenic tremor. Brain. 2012; 135.
15. Baas C, and Halligan P. Factitious disorders and malingering: challenges for clinical assessment and management. The Lancet. 2014; 383.
16. Bass C, and Halligan P. Factitious disorders and malingering in relation to functional neurologic disorders. Handbook of clinical neurology. 2016; 139-3rd series. Ch. 42: 513-514.
17. Spence S, Crimslisk H, Cope H, Ron M, and Grasby P. Discrete neurophysiological correlates in prefrontal cortex during hysterical and feigned disorder of movement. The Lancet. 2000; 355(9211).
18. Nicholson T, Stone J, and Kanaan R. Conversion disorder: a problematic diagnosis. Journal of Neurology, Neurosurgery, and Psychiatry. 2011; 82.
19. Paget J. Lecture I: Nervous mimicry of organic diseases. The Lancet. 1873; 102(615): 511-513.
20. Feinstein A. Conversion disorder. Continuum. 2018; 24(3).
21. Stone J, and Carson A. Functional neurologic disorders. Continuum. 2015; 21(3).
22. American Psychiatric Association. Diagnostic and statistical manual of mental disorders, 5th ed. (DSM-5). Washington, DC: American Psychiatric Association, 2013.
23. Daum C, Hubschmid M, and Aybek S. The value of ‘positive’ clinical signs for weakness, sensory and gait disorders in conversion disorder: a systematic and narrative review. Journal of Neurology, Neurosurgery and Psychiatry. 2014; 85.
24. Stone J. The bare essentials: functional symptoms in neurology. Neurology in Practice. 2009; 9.
25. McWhirter L, Stone J, Sandercock P, and Whiteley W. Hoover’s sign for the diagnosis of functional weakness: a prospective unblinded cohort study in patients with suspected stroke. Journal of Psychosomatic Research. 2011; 71.
26. Soonoo M. Abductor sign: a reliable new sign to detect unilateral non-organic paresis of the lower limb. Journal of Neurology, Neurosurgery and Psychiatry. 2004; 75.
27. Stone J, Warlow C, and Sharpe M. The symptom of functional weakness: a controlled study of 107 patients. Brain. 2010; 133.
28. Baker J, and Silver J. Hysterical paraplegia. Journal of neurology, neurosurgery and psychiatry. 1987; 50.
29. Darcy P, and Moughty. Images in clinical medicine – pronator drift. NEJM. 2013; 369.
30. Tremolizzo L, Susani E, Riva M et al. Positive signs of functional weakness. Journal of the neurological sciences. 2014; 340.
31. Roper L, Saifee T, Parees I, Rickards H, and Edwards M. How to use the entrainment test in the diagnosis of functional tremor. Practical Neurology. 2013; 13.
32. Stone J, and Vermeulen M. Functional sensory symptoms. Handbook of clinical neurology. 2016; 139-3rd series. 271-281.
33. Rolak L. Psychogenic sensory loss. The journal of nervous and mental disease. 1988; 176(11).
34. Laub H, Dwivedi A, Revilla F et al. Diagnostic performance of the “Huffing and Puffing” sign in psychogenic (functional) movement disorders. Movement disorders clinical practice. 2014; 2(1).
35. Carson A, and Lehn A. The assessment of patients with functional neurologic disorders. Handbook of clinical neurology. 2016; 139-3rd series. Ch. 15: 169-188.
36. Gould R, Miller B, Goldberg M, and Benson F. The validity of hysterical signs and symptoms. The journal of nervous and mental disease. 1986; 174(10).
37. Chabrol H, Peresson G, and Clanet M. Lack of specificity of the traditional criteria for conversion disorders. European Psychiatry. 1995; 10.
38. Stone J, Smyth R, Carson A et al. Systematic review of misdiagnosis of conversion symptoms and “hysteria”. BMJ. 2005; 331(7523).
39. Stone J, Smyth R, Carson A, and Warlow C. La belle indifference in conversion symptoms and hysteria. British Journal of Psychiatry. 2006; 188.
40. Espay A, Aybek S, Carson A et al. Current concepts in diagnosis and treatment of functional neurologic disorders. JAMA Neurology. 2018.
41. Hatcher S, and Arroll B. Assessment and management of medically unexplained symptoms. BMJ. 2008; 336.
42. Howard L, Wessely S, Leese M et al. Are investigations anxiolytic or anxiogenic? A randomised controlled trial of neuroimaging to provide reassurance in chronic daily headache. Journal of Neurology, Neurosurgery and Psychiatry. 2005; 76.
43. Stone J, Carson A, Duncan R et al. Which neurological diseases are most likely to be associated with “symptoms unexplained by organic disease”. Journal of neurology. 2012; 259(1).
44. Ding J, and Kanaan R. What should we say to patients with unexplained neurological symptoms? How explanation affects offence. Journal of psychosomatic research. 2016; 91.
45. Stone J. Functional and dissociative neurological symptoms: a patient’s guide. www.neurosymptoms.org Last accessed: August 21, 2018.
46. Nielsen G, Buszewicz M, Stevenson F et al. Randomised feasibility study of physiotherapy for patients with functional motor symptoms. Journal of neurology, neurosurgery, and psychiatry. 2017; 88(6).
47. Dallocchio C, Tinazzi M, Bombieri F, Arno N, and Erro R. Cognitive behavioural therapy and adjunctive physical activity for functional movement disorders (conversion disorder): a pilot study. Psychotherapy and Psychosomatics. 2016; 85.
48. Sharpe M, Walker J, Williams C et al. Guided self-help for functional (psychogenic) symptoms – A randomized controlled efficacy trial. Neurology. 2011; 77(6).
49. Salmon P, Peters S, and Stanley I. Patients’ perceptions of medical explanations for somatisation disorders: qualitative analysis. BMJ. 1999; 318.
50. Dowrick C, Ring A, Humphris G, and Salmon P. Normalisation of unexplained symptoms by general practitioners: a functional typology. British Journal of General Practice. 2004; 54(500).
51. Goldstein L, Chalder T, Chigwedere C et al. Cognitive-behavioural therapy for psychogenic nonepileptic seizures: a pilot RCT. Neurology. 2010; 74(24).
52. Goldstein L, Mellers J, Landau S et al. COgnitive behavioural therapy vs standardised medical care for adults with Dissociative non-Epileptic Seizures (CODES): a multicentre randomised controlled trial protocol. BMC Neurology. 2015; 15.
53. “Amongst Friends”. 24 Hours in A&E. Channel 4. 2014.
54. Wasserman D, and Herskovitz M. Epileptic vs psychogenic nonepileptic seizures: a video-based survey. Epilepsy and Behaviour. 2017; 73.
55. Aboukasm A, Mahr G, Gahry B, Thomas A, and Barkley G. Retrospective analysis of the effects of psychotherapeutic interventions on outcomes of psychogenic nonepileptic seizures. Epilepsia. 1998; 39(5).
56. Jirsch J, Ahmed S, Maximova K, and Gross D. Recognition of psychogenic nonepileptic seizures diminishes acute care utilization. Epilepsy and behavior. 2011.
57. LaFrance W, Baker G, Duncan R, Goldstein L, and Reuber M. Minimum requirements for the diagnosis of psychogenic nonepileptic seizures: a staged approach. Epilepsia. 2013; 54(11).
58. Abou-Khalil B, Gallagher M, and Macdonald R. Epilepsies. Bradley’s Neurology in Clinical Practice, 7th ed. 2016; Ch. 101.
59. White . Epileptic seizure patterns. Department of Neurology, School of Medicine, Indiana University. 1967. Accessed from: https://www.youtube.com/watch?v=NCC2949FozY
60. Erba G, Bianchi E, Giussani G et al. Patients’ and caregivers’ contributions for differentiating epileptic from psychogenic nonepileptic seizures. Value and limitations of self-reporting questionnaires: A pilot study. Seizure. 2017; 53.
61. Chen D, and LaFrance W. Diagnosis and treatment of nonepileptic seizures. Continuum. 2016.
62. Carlson P, and Perry K. Psychological interventions for psychogenic non-epileptic seizures: a meta-analysis. Seizure. 2017; 45.
63. Razvi S, Mulhern S, and Duncan R. Newly diagnosed psychogenic nonepileptic seizures: health care demand prior to and following diagnosis at a first seizure clinic. Epilepsy and behaviour. 2012; 23.
64. Siket M, and Merchant R. Psychogenic seizures: a review and description of pitfalls in their acute diagnosis and management in the emergency department. Emergency Medicine Clinics of North America. 2011; 29.
65. Farias S, Thieman C, and Alsaadi T. Psychogenic nonepileptic seizures: acute change in event frequency after presentation of the diagnosis. Epilepsy & Behavior. 2003.
66. Petrie K, Muller J, Schirmbeck F et al. Effect of providing information about normal test results on patients’ reassurance: randomised controlled trial. BMJ. 2007; 334(7589).
67. Serinken M, Mehmet Z, Ozgur K, Serkan S, and Ibrahim T. Value of the timing of informing the emergency department patients on cardiac test results: a randomized controlled study. European journal of emergency medicine. 2009; 16(2).



