In this post, we review an approach to antibiotics, bronchodilators, and corticosteroids in the outpatient management for acute exacerbations of COPD.
COPD – The Basics
What is it?
- An incompletely reversible obstructive lung disease due to airway and/or alveolar abnormalities leading to airflow limitation and persistent respiratory symptoms
- Caused by significant exposure to noxious particles or gases
- Most common – cigarette smoking
- Occupational exposures (i.e. fumes, dusts)
- Environmental exposures (i.e. air pollution, indoor solid fuel burning)
- Caused by significant exposure to noxious particles or gases
Pathophysiology simplified
- Inflammation/narrowing of airways – decreased forced expiration (FEV1) leading to hyperinflation
- Parenchymal destruction – decreased gas transfer leading to hypoxia and hypercapnia
- Mucous hypersecretion – chronic cough
- Hypoxic vasoconstriction – pulmonary hypertension
Diagnosis
- Diagnosis made with spirometry in the correct clinical context and FEV1/FVC <0.70
- Baseline severity = GOLD 2019 scoring:
- GOLD 1 (mild): FEV1 ≥ 80% predicted
- GOLD 2 (moderate): 50% ≤ FEV1 < 80% predicted
- GOLD 3 (severe): 30% ≤ FEV1 <50%
- GOLD 4 (very severe): FEV1 ≤ 30% predicted
Acute Exacerbations of COPD (AECOPD)
Definition
- A routinely referred to definition of is from the 1987 Anthonisen trial describing the “cardinal symptoms” that included an increase from baseline of:
- Dyspnea
- Sputum volume
- Sputum purulence
- This same group defined types of exacerbations from 1 to 3:
- Type 1 had all three cardinal symptoms
- Type 2 had two of three symptoms
- Type 3 had one of three symptoms in addition to other upper respiratory infection symptoms
- Over thirty years later, the current GOLD 2019 guidelines provide a less specific, but more universally applicable, definition of an AECOPD: “acute worsening of respiratory symptoms that result in additional therapy.”
Causes & Risks
- Causes of acute exacerbations are split into non-infectious (30%) and infectious (70%)
- Non-infectious causes may include allergic/eosinophilic exacerbations
- Infectious causes are split nearly 50:50 between viral (35-65%) and bacterial (35-60%)
- Sethi (2008) described the bacterial pathogens in COPD exacerbations:
- Bacterial:
- Haemophilus influenzae (20-30%)
- Moraxella catarrhalis (10-15%)
- Streptococcus pneumoniae (10-15%)
- Note: the role of Pseudomonas aeruginosa in exacerbations is low and mainly isolated in more advanced disease. Also, unlike community-acquired pneumonias, atypical bacteria play a very small role in AECOPD (1-5%)
- Bacterial:
- Hurst (2010) found the best predictor of an exacerbation was a history of previous exacerbations, especially if within the previous year
- As the baseline severity of COPD increased so did the likelihood of exacerbations and the severity of exacerbations (i.e.: higher hospitalization rates)
Diagnosis
- It can be difficult to have a solid approach to diagnosing an AECOPD given the original definition and non-specific symptoms (Anthonisen 1987) as well as the even less specific current GOLD 2019 definition
- Dr. Aaron (respirologist at The Ottawa Hospital) advised the following considerations to having an organized approach:
- Patient presents with dyspnea (+/- cough, increased sputum volume, and/or sputum purulence)
- Without dyspnea, a confident diagnosis of AECOPD should not be made; other symptoms may be present; however dyspnea is key
- Consider if this is indeed from COPD? There are 2 key considerations here:
- Is there a formal history of COPD (i.e. PFTs available)?
- If no formal history, are there risk factors present?
- ≥10 pack year smoking history
- Exposure history (e.g. fumes, occupational, environmental)
- A simultaneously important step = Rule out alternative diagnoses
- All patients should have a chest X-ray
- Guide additional investigations by history and physical findings (i.e. chest pain and ACS work-up, orthopnea/edema and CHF work-up, etc.)
- Beghe (2013) described thinking about not labeling patients as having an AECOPD but rather “exacerbations of respiratory symptoms in patients with COPD.” This worsening may be from airway inflammation from COPD, but it may also be from alternative respiratory and non-respiratory conditions and changing the way we label these patients may improve our awareness of possible alternative diagnoses that may be present
- If worsening dyspnea is present in a patient with COPD (or known risk factors are present), and alternative diagnoses have been ruled out, a diagnosis of an acute exacerbation can be supported
- Patient presents with dyspnea (+/- cough, increased sputum volume, and/or sputum purulence)
Severity
- The GOLD 2019 guidelines grade exacerbation severities in an “event-based” format (i.e. depending on the management provided and treatment setting):
- Mild – treat with short acting bronchodilators (SABD) (home)
- Moderate – treat with SABDs + corticosteroids +/- antibiotics (home)
- Severe – above treatment + hospitalization
- There are no routinely described patient features that are used to classify exacerbations into the various severities. However GOLD (2019) list indications for hospitalization; and since such a disposition is considered to be a “severe” exacerbation, these features can be used to help identify the severe events.
- Indications for hospitalizations (GOLD 2019) include:
- Severe symptoms: tachypnea, hypoxia, confusion
- Acute respiratory failure
- New physical signs (i.e. edema, cyanosis)
- Serious comorbidities (i.e. cardiac)
- Failure of initial treatment
- Lack of home support
- Indications for hospitalizations (GOLD 2019) include:
- Important note: >80% of AECOPD are managed at home (i.e. mild-moderate)
- The treatment considerations for acute exacerbations can be described as the “ABCs”
- Antibiotics
- Bronchodilators
- Corticosteroids
- The treatment considerations for acute exacerbations can be described as the “ABCs”
Treatment of AECOPD
The following information provides an overview of important literature behind each of the above treatment modalities to assist physicians in making evidence-based decisions and are arranged in order of increasing controversy.
Bronchodilators
- The use of short acting bronchodilators (short acting beta-agonists – SABAs; and short acting anticholinergics – SAACs) are known fundamental components to AECOPD
- A Cochrane Review (McCrory 2003) looked at the difference between effectiveness of SABAs (Salbutamol) vs. SAACs (Ipratroprium bromide) in AECOPD and found no difference in the degree of bronchodilation at 1.5h
- There was also no evidence that the combo of SABA+SAAC was more effective than the individual use of either formulation alone
- Another Cochrane Review (Van Geffen 2016) analysed the effectiveness of bronchodilators delivered through nebuliser vs. MDI+spacer and found no difference between these modalities on bronchodilation at 1 hour
- GOLD 2019 recommends using a SABA +/- SAAC
Bottom Line
- For slightly quicker onset consider SABA (5min vs. 10-15min), but no difference in bronchodilation vs. SAAC
- If history of intolerance to one type due to side effects, then use the other
- For more severe exacerbations can consider combination (however no evidence for this!)
- Choose delivery method based on severity of symptoms:
- MDI+spacer for those not severely tachypneic
- Nebulizer if unable to take solid breath due to tachypnea
- But switch to MDI+spacer ASAP
Discharge Instructions
- This is VERY often overlooked in the ED
- We often forget to educate patients to increase SABA+/-SAAC use during exacerbations despite this being a foundational component to improve dyspnea
- Instruct patients to use 2-4 inh q4h straight x 24-48h
PEARL! What about the use of long-acting bronchodilators?
- No evidence these are effective in treating ACUTE exacerbations
- But there is STRONG evidence they help prevent future exacerbations when used at baseline! (GOLD 2019)
- If the patient is not on a long-acting bronchodilator (i.e. Tiotropium – long acting anticholinergic, LAAC) we should be giving them a prescription for this in the ER
- An example would be: Tiotropium handihaler 18mcg 1 inh daily
- Note: either long acting anticholingerics (LAACs) or long acting beta-agonists (LABAs) can be considered, but there is slightly more evidence behind the use of LAACs
Corticosteroids
- The use of corticosteroids to treat AECOPD comes up very frequently. But what does the evidence show?
- An RCT by Aaron (2003) looked at the use of Prednisone 30mg PO daily for 10 days versus placebo and found:
- By day 10 the Prednisone group had significant improvement in bronchodilation (FEV1) and dyspnea scores
- At day 30 the Prednisone group had decreased rates of relapse with a NNT of 6
- Note: both groups received 10d of antibiotics
- A Cochrane Review (Walters 2014) analyzed the evidence behind corticosteroid use in AECOPD involving 16 RCTs (however, only 3 were outpatients) and found the use steroids resulted in:
- Improved FEV1 at day 3 and end-of-treatment (EOT), improved dyspnea at day 3, and decreased relapse at day 30 (NNT of 9)
- There were increased events of hyperglycemia in the steroid groups (NNH 7)
- No mention on the degree of hyperglycemia or which patient population. Therefore, one should ensure consideration is made in those with poorly controlled diabetes
- This paper also looked at the effectiveness of IV vs. PO formulation of steroids and found NO difference in any outcomes of effectiveness. But increased hyperglycemia was noted in the IV groups
- Overall evidence for steroid use:
- Studies are limited for outpatient use (the study with the strongest methodology = Aaron 2003)
- Strongest benefit in in those with subjective dyspnea
- Aaron 2003 = ~100% in both treatment and control groups presented with dyspnea and this is the population where benefit was seen
Steroid Dosing
- Arcos (2016) performed a systematic review attempting to compare high- versus low-dose systemic steroids in AECOPD. They were unable to find any RCTs comparing different dosing on clinical outcomes (either primary outcomes or subgroup analysis)
- Bottom Line – who knows? It’s controversial
- Most studies use prednisone 30-50mg PO daily, and any dose between this is OK
- PEARL! Consider patient reliability and Rx convenience!
- In Canada oral prednisone for AECOPD dosing = 5mg or 50mg tabs
- Therefore, 30mg = 6tabs, 40mg = 8tabs
- Use lower doses in reliable patients without high baseline pill burdens & bite-the-bullet for simplicity on the 50mg dose in the less reliable patients with multiple daily pills
- Bottom Line – who knows? It’s controversial
How about Duration of treatment?
- A non-inferiority RCT (the REDUCE trial – Leuppi 2013) looked at short (Prednisone 40mg PO x 5d) vs. long (Prednisone 40mg PO x 14d) duration of steroids in AECOPD and found
- No differences in FEV1, dyspnea scores, or relapse at 180d between the two groups
- No differences in adverse events
- Note: 92% of these patients were admitted for ~8-9d (therefore sicker exacerbations)
- However, these results can very likely apply to the less severe outpatients with AECOPD
- A recent Cochrane Review (Walters 2018) analyzed the bigger picture of RCTs comparing short vs. long duration of steroids and found no difference in relapse, time to next exacerbation, length of hospitalization, or adverse events between the groups
- They conclude a 5 day treatment is sufficient
- Is is important to note again that all patients were admitted (severe to very severe exacerbations), but applicability still very likely exists to the less severe outpatient population of interest
Bottom Line
- Use corticosteroids in AECOPD with DYSPNEA (remember, we really shouldn’t be making the diagnosis of AECOPD without dyspnea anyways!)
- Give Prednisone 30-50mg PO daily x 5 days (consider patient reliability and convenience)
- GOLD 2019 uses 40mg PO daily x 5 days (references to the REDUCE trial mentioned above)
- Important considerations = those with history of needing long tapers (unlikely to now benefit from 5d course)
- What about Dexamethasone?
- Unfortunately no evidence yet for AECOPD
- Give Prednisone 30-50mg PO daily x 5 days (consider patient reliability and convenience)
Antibiotics
- Lets recap: ~70% of exacerbations are infectious
- 30-65% viral
- 35-50% bacterial
- Are there any clinical features that could point towards a bacterial cause? There really isn’t much out there, but from what there is, it seems sputum purulence is likely the strongest predictor in the COPD population
- A study by Soler (2007) compared bronchoscopic sputum culture results of those hospitalized with AECOPD to the colour of sputum at presentation, using this diagram:
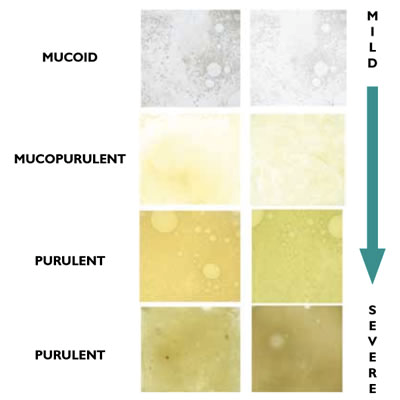
- They found purulent sputum had a sensitivity of 89.5% and specificity of 76.2% for positive bacterial cultures
- Another study by Stockley (2000) looked at a similar question but in outpatients with AECOPD using expectorated sputum samples and found sputum purulence to be 94.4% sensitive and 77.0% specific for positive bacterial cultures
- Taking these studies into consideration, there was a review article (Dickson 2014) that compared studies of exacerbations of COPD and stable COPD where sputum cultures were obtained (all were bronchoscopic sampling). Interestingly they found:
- Bacteria with pathological potential were in cultures:
- 51-70% during AECOPD, and
- 25-48% during stability
- This raises the question of whether the bacteria are acute pathogens or colonizers? This is still in question throughout the COPD community
- Bacteria with pathological potential were in cultures:
- Overall – based on the above evidence:
- Absence of purulence can be helpful to rule out bacterial cause (decent sensitivities)
- Presence of purulence requires the use of the clinical context and severity of the patient, as the presence of this alone is not good enough to predict bacterial infection
Antibiotic use is the most controversial component of AECOPD management
- The current available guidelines recommend antibiotic use in the following patients:
- GOLD 2019:
- 3/3 cardinal symptoms (dyspnea, sputum purulence, sputum volume)
- 2/3 cardinal symptoms (if sputum purulence present)
- Those mechanically ventilated
- 3/3 cardinal symptoms (dyspnea, sputum purulence, sputum volume)
- ERS/ATS 2017 (Europe & US)
- Simply states those with “exacerbations”; however, increased benefit with sputum purulence
- CTS 2008 (Canada)
- Exacerbations with purulent sputum
- Unfortunately, there hasn’t been a Canadian update for the past 10 years
- GOLD 2019:
- What is evident with these guidelines is the presence of inconsistency!
Evidence behind the guidelines:
- The most fundamental and frequently referenced paper for the use of antibiotics in AECOPD is the 1987 Anthonisen trial
- RCT (n=173) of outpatients given 10 days of antibiotics vs. placebo in the setting of an exacerbation
- This is where the “Types” of exacerbations were defined:
- Type 1 = 3/3 cardinal symptoms
- Type 2 = 2/3 cardinal symptoms
- Type 3 = 1/3 cardinal symptoms with other URTI symptoms
- It is very important to note that these patients had severe (bordering very severe) baseline COPD (mean FEV1 33%), had daily dyspnea and sputum production, overall a quite severe baseline
- Also, no CXRs were performed, there was no intention to treat analysis of the results, and it was heavily industry funded by companies producing antibiotics
- What were the results?
- Note: this study included patients with multiple exacerbations; therefore the results are broken down into “1st” and “all” exacerbations
- The results have been summarized in the table below.
Treatment success at day 21
| Placebo | Antibiotic | p-value | |
| 1st exacerbation | 52.3% | 66.8% | 0.06 |
| All exacerbations | 55.0% | 68.1% | <0.01 |
| Type 1 exacerbations | 43.0% | 62.9% | Not listed |
| Type 2 exacerbations | 60.0% | 70.1% | Not listed |
| Type 3 exacerbations | 69.7% | 74.2% | Not listed |
- The statistical benefit was only seen in those with multiple exacerbations (no statistical difference between treatment groups for 1st exacerbations)
- Also – there are no p-values listed by the authors for results of the various “types” of exacerbations, and therefore one can imply no significant difference exists
- The days to resolution between groups: Placebo (15.5d) vs. Antibiotic (14.1d)
- Again, no p-value is listed for the unlikely clinically significant difference of 1.4 days beyond the 2-week mark of illness, implying no difference
- The bottom line of this article is that despite heavy referencing within guidelines, and methodological concerns listed above, it really should only be generalized to those with severe to very severe baseline symptoms and ultimately doesn’t support routine antibiotic use that well at all!
- A Cochrane Review (Vollenweider 2012) looked at RCTs (n=16 with 9 outpatient studies) of antibiotic use in AECOPD vs. placebo and split the analysis into mild-moderate (outpatient), severe (inpatient), very severe (ICU) exacerbations:
- For mild-moderate exacerbations (when restricting to antibiotics used today – Clavulin, Septra, Doxycycline, PCN):
- No difference in treatment failure was found (defined as no resolution/deterioration of symptoms at 7-30d)
- For mild-moderate exacerbations (when restricting to antibiotics used today – Clavulin, Septra, Doxycycline, PCN):
- A recent multicenter RCT (van Velzen 2017) looked at Doxycycline (7days) vs. Placebo for outpatient AECOPD, where both groups received Prednisone x 10d, and they found:
- No difference in treatment non-response at day 21 for:
- All patients: Doxy (21%) vs Placebo (31%) – p=0.07
- “Type 1” Exacerbations: p=0.61
- Purulent sputum: p=0.54
- No difference in time to next exacerbation: Doxy (148d) vs Placebo (161d) – p=0.91
- The conclusion was they did not recommend routine use of antibiotics for outpatient management of AECOPD
- No difference in treatment non-response at day 21 for:
- Lastly, and even more recently, another Cochrane Review on this matter was published (Vollenweider 2018) again looking at RCTs of antibiotics versus placebo (n=19 with 11 outpatient studies) and found:
- For mild-moderate (outpatient) exacerbations:
- No good evidence to support difference in dyspnea, re-exacerbation, or time to next exacerbation
- There was a slight, statistically significant, decrease in treatment failure with antibiotics
- Note: this was listed as “Low Grade” evidence due to large variations in the study protocol and baseline severities (i.e. Anthonisen 1987) of the studies involved
- The authors actually concluded that, despite this slight benefit seen, uncertainty continues and, “current data are not conclusive enough to show whether antibiotics should generally be used, because results among outpatients are heterogeneous and are associated with risk of bias, and because evidence on patient-important outcomes such as health-related quality of life is not available”
- For mild-moderate (outpatient) exacerbations:
- Jumping back to the GOLD 2019 guidelines for antibiotic use listed above, what references are these guidelines based off of? Well, there’s only two listed:
- 1st is the Anthonisen 1987 study we just spent time analyzing
- 2nd is a Cochrane Review (Ram 2006) that was withdrawn many years ago for methodological flaws (didn’t separate severity of exacerbations or those in the ICU vs. outpatient management) and for the availability of new Cochrane Reviews on the same topic = Vollenweider 2012, 2018. Despite these both being available GOLD has yet to update their 2006 reference
Overall – Evidence for Antibiotic Use
- Severe AECOPD (inpatient/ICU) = Use Antibiotics
- Mild-Moderate AECOPD (outpatient) = uncertain benefit, low grade evidence at best, large percentage placebo improvement in trials
- If no sputum purulence = no antibiotics
- But caution with purulence alone, use clinical context
- Future directions?
- How about considering a delayed prescription for the patient to allow time for the other fundamental treatments to work? If not better in 2-3days then fill the prescription
- If no sputum purulence = no antibiotics
Antibiotic Choice
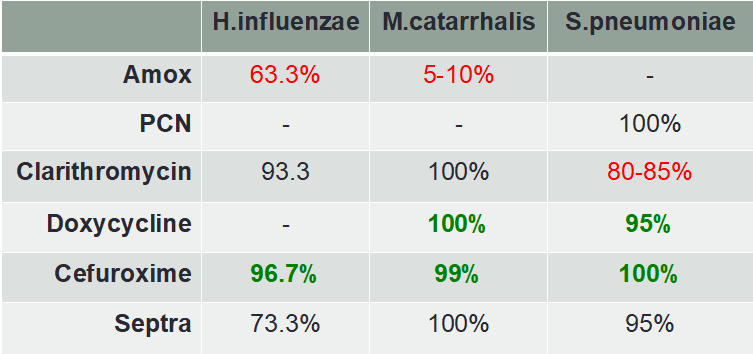
- The main bacteria involved are H.influenzae, M.catarrhalis, and S.Pneumoniae. Pseudomonas is minimally involved and really only in those with severe baseline disease
- 2 key considerations in choosing antibiotics:
- 1. Baseline risk of resistant organisms
- 2. Local resistance patterns
- Risk stratification for resistance:
- Previous Canadian Thoracic Society guidelines on the management of AECOPD (Balter 2003, O’Donnell 2008) and a detailed expert review article (Santos 2016) highlight the key patient features that increase the likelihood of resistant bacteria being present. The following table is a summary of these features dividing risks of resistance into low, high, and risk of pseudomonas presence
- 2 key considerations in choosing antibiotics:

- Avoid repeat usage of the same class
- As with all other antibiotic prescribing practices, it is important to consider the use of recent antibiotics (i.e. past 3 months) as well as if the patient is on baseline prophylactic antibiotics to avoid repeat usage of the same class
- Local resistance patterns:
- The main causative organisms listed above for bacterial AECOPD are not the common bugs that cause severe illness throughout medicine, so information on true “local” resistance is limited
- Fortunately, using the Canadian Antimicrobial Resistance Alliance (CARA) database for Ontario (2015, 2016), the majority of this information can be obtained. The Ottawa Hospital 2018 local susceptibilities are available for some S.pneumoniae results and information had to be gathered from a B.C. report for M.catarrhalis resistance patterns (Bandet 2014)
- A summary of these local resistance patterns relating to the possible causative organisms determined by the risk stratification previously listed is available in these tables:
Low Risk
High risk
Pseudomonas Risk
- Important considerations reflected in the tables:
- High levels of beta-lactamase producing H.influenzae and M.catarrhalis organisms (increasing amoxicillin resistance)
- Increasing macrolide resistance with S.pneumoniae
- Fluoroquinolone patterns are NOT listed in the “low-risk” table because of FDA Black Box warnings (2016, 2018) contraindicating their use in this group. This is related to CNS effects (i.e. delirium, agitation), hypoglycemia, peripheral neuropathies, and tendinopathies
- These should be reserved only for those considered “high-risk” for resistance
- Clavulin should also be reserved for “high-risk” patients given the strong susceptibilities of less aggressive alternatives (i.e. doxycycline, cefuroxime)
Bottom Line – Antibiotic Choice and Duration
- Antibiotic Choice:
- Consider both baseline risk of resistant organisms and local resistance patterns
- Using the tables above, consider:
- Low risk: doxycycline or cefuroxime
- Can still use amox or macrolides; but if no improvement then switch to a different class with better coverage
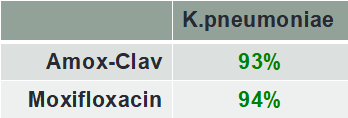
- High risk: 1st line Clavulin, if contraindications then a fluoroquinolone
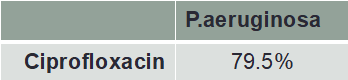
- Pseudomonas risk: can still be affected by the “typical” organisms therefore it is okay to treat with “high-risk” choice above and if no improvement then consider ciprofloxacin
- Low risk: doxycycline or cefuroxime
- Using the tables above, consider:
- Consider both baseline risk of resistant organisms and local resistance patterns
- Antibiotic Duration:
- Several systematic reviews and meta-analyses have compared short (5d) versus long (>7d) courses of antibiotics in AECOPD (Falagas 2008, Moussaoui 2008, Stolbrink 2017). The common conclusion throughout found no difference in effectiveness between 5d vs. longer courses, however, more adverse events occurred in the longer courses
- The above resources looked at macrolides, 2nd/3rd gen cephalosporins, clavulin, and fluoroquinolones
- Overall:
- If antibiotic given – use 5d course
- Exception – use 7-10d as clinically indicated for amoxicillin and doxycycline (no available studies found on these)
- Several systematic reviews and meta-analyses have compared short (5d) versus long (>7d) courses of antibiotics in AECOPD (Falagas 2008, Moussaoui 2008, Stolbrink 2017). The common conclusion throughout found no difference in effectiveness between 5d vs. longer courses, however, more adverse events occurred in the longer courses
Bottom Line for Antibiotics in AECOPD
- Uncertain evidence for true benefit in mild-moderate (outpatient) exacerbations
- Consider using with purulent sputum and concerning clinical severity
- Choice:
- Low-risk: doxycycline, cefuroxime
- High-risk: clavulin, moxifloxacin/levofloxacin (only if cannot use clavulin)
- Duration:
- All above antibiotics = 5days
- Exception: Doxycycline and amoxicillin = 7days
- All above antibiotics = 5days
- Note: consider giving the patient a delayed prescription to fill if symptoms are not improving in 48h with optimization of other treatments (i.e. bronchodilators and steroids)!
Disposition considerations
Pulmonary function testing
- A diagnosis of COPD cannot be made clinically; it requires formal PFTs
- Indications for ordering from the ED:
- Unclear diagnosis:
- Possible first AECOPD in a patient who meets the risk factors mentioned above in the diagnostic approach and has no previous PFTs
- Progression of disease:
- Subacute/progressive worsening in symptoms
- Unclear diagnosis:
- Easy to complete referral form in ED
- Copy their family physician to follow-up on results
- Indications for ordering from the ED:
Smoking cessation
- Very often overlooked in the ED
- Can decrease further decline in lung function and decrease all-cause mortality (Anthonisen 2005)
- What can we do in the ED?
- Provide local resources to assist with quitting:
- In Ottawa – myQuit (myquit.ca) and the Quit Smoking Program (at the Heart Institute)
- Both are free programs, self-referral, and can coach patient’s through initiating their quitting process
- 5 weeks of free nicotine patches or gum provided
- Prescribe Dual Nicotine Replacement Therapy (NRT):
- Varenicline (Champix):
- We SHOULD be prescribing this in the ED!
- Champix 0.5mg/1mg starter kit as directed, then Champix 1mg PO BID x 11 weeks
- LU Code 423 (one per year)
- Very few contraindications:
- Known allergy, CKD (dose adjust for CrCl <30), pregnancy
- Side effects to discuss:
- Nausea, insomnia, abnormal dreams
- Instruct patient to start ~8-10d prior to planned quit date
- Nicotine patches:
- Can be used in conjunction with Varenicline if cravings continue when quitting
- Over-the-counter but less expensive than smoking
- General dosing recommendations:
- If ≥10 cigarettes/day = 21mg patch
- If <10 cigarettes/day = 14mg patch
- Varenicline (Champix):
- Provide local resources to assist with quitting:
To wrap it all up
- An approach to diagnosing an AECOPD was discussed
- Remember that dyspnea is needed for a true exacerbation
- Remember to rule out other causes and think about risk factors if no previous documentation of COPD
- Causes of exacerbations are 30:70 for non-infectious to infectious with ~50:50 split between viral and bacterial
- Treatments:
- Bronchodilators: use short acting (Ventolin +/- Atrovent) in the ED and ensure patient will optimize their use on discharge. If not already on a long-acting anticholinergic (i.e. Tiotropium – Spiriva) we SHOULD be providing a prescription for daily use to prevent FUTURE exacerbations
- Corticosteroids: best evidence for efficacy is for those who are dyspneic. Give 5 days of prednisone anywhere between 30-50mg daily. Consider patient reliability for compliance and number of pills with each dose
- Antibiotics: remains the most controversial topic. Consider use in appropriate clinical context with purulent sputum. Base choice of antibiotics on baseline resistance likelihood and local resistance patterns while limiting fluoroquinolones as much as possible. A 5 day prescription is sufficient, and you can consider giving a delayed prescription
- Disposition considerations:
- PFTs: order if no previous, unclear diagnosis, and/or progressive symptoms
- STOP SMOKING! By far the biggest benefit we can offer our patients. Recommend local quit smoking programs and provide prescription for Varenicline (Champix) +/- use of nicotine patches for continued cravings
References
- Global Initiative for Chronic Obstructive Lung Disease. Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Pulmonary Disease. Updated 2019. Global Initiative for Chronic Obstructive Lung Disease, 2019.
- Anthonisen NR, Manfreda J, Warren CP, Hershfield ES, Harding GK, Nelson NA. Antibiotic therapy in exacerbations of chronic obstructive pulmonary disease. Ann Intern Med. 1987;106(2):196-204.
- Sethi S, Murphy TF. Infection in the pathogenesis and course of chronic obstructive pulmonary disease. N Engl J Med. 2008;359(22):2355-2365.
- Hurst JR, Vestbo J, Anzueto A, et al. Susceptibility to exacerbation in chronic obstructive pulmonary disease. N Engl J Med. 2010;363(12):1128-1138.
- Beghe B, Verduri A, Roca M, Fabbri LM. Exacerbation of respiratory symptoms in COPD patients may not be exacerbations of COPD. Eur Respir J. 2013;41(4):993-995.
- McCrory DC, Brown CD. Anti-cholinergic bronchodilators versus beta2-sympathomimetic agents for acute exacerbations of chronic obstructive pulmonary disease. Cochrane database Syst Rev. 2002;(4):CD003900.
- van Geffen WH, Douma WR, Slebos DJ, Kerstjens HAM. Bronchodilators delivered by nebuliser versus pMDI with spacer or DPI for exacerbations of COPD. Cochrane database Syst Rev. 2016;(8):CD011826.
- Aaron SD, Vandemheen KL, Hebert P, et al. Outpatient oral prednisone after emergency treatment of chronic obstructive pulmonary disease. N Engl J Med. 2003;348(26):2618-2625.
- Walters JAE, Tan DJ, White CJ, Gibson PG, Wood-Baker R, Walters EH. Systemic corticosteroids for acute exacerbations of chronic obstructive pulmonary disease. Cochrane database Syst Rev. 2014;(9):CD001288.
- Bonilla Arcos D, Krishnan JA, Vandivier RW, et al. High-Dose Versus Low-Dose Systemic Steroids in the Treatment of Acute Exacerbations of Chronic Obstructive Pulmonary Disease: Systematic Review. Chronic Obstr Pulm Dis (Miami, Fla). 2016;3(2):580-588.
- Leuppi JD, Schuetz P, Bingisser R, et al. Short-term vs conventional glucocorticoid therapy in acute exacerbations of chronic obstructive pulmonary disease: the REDUCE randomized clinical trial. JAMA. 2013;309(21):2223-2231.
- Walters JA, Tan DJ, White CJ, Wood-Baker R. Different durations of corticosteroid therapy for exacerbations of chronic obstructive pulmonary disease. Cochrane database Syst Rev. 2018;3:CD006897.
- Soler N, Agusti C, Angrill J, Puig De la Bellacasa J, Torres A. Bronchoscopic validation of the significance of sputum purulence in severe exacerbations of chronic obstructive pulmonary disease. Thorax. 2007;62(1):29-35.
- Stockley RA, O’Brien C, Pye A, Hill SL. Relationship of sputum color to nature and outpatient management of acute exacerbations of COPD. Chest. 2000;117(6):1638-1645.
- Dickson RP, Martinez FJ, Huffnagle GB. The role of the microbiome in exacerbations of chronic lung diseases. Lancet (London, England). 2014;384(9944):691-702.
- Wedzicha JAEC-C, Miravitlles M, Hurst JR, et al. Management of COPD exacerbations: a European Respiratory Society/American Thoracic Society guideline. Eur Respir J. 2017;49(3).
- O’Donnell DE, Hernandez P, Kaplan A, et al. Canadian Thoracic Society recommendations for management of chronic obstructive pulmonary disease – 2008 update – highlights for primary care. Can Respir J. 2008;15 Suppl A(Suppl A):1A-8A.
- Vollenweider DJ, Jarrett H, Steurer-Stey CA, Garcia-Aymerich J, Puhan MA. Antibiotics for exacerbations of chronic obstructive pulmonary disease. Cochrane database Syst Rev. 2012;12:CD010257.
- van Velzen P, Ter Riet G, Bresser P, et al. Doxycycline for outpatient-treated acute exacerbations of COPD: a randomised double-blind placebo-controlled trial. Lancet Respir Med. 2017;5(6):492-499.
- Vollenweider DJ, Frei A, Steurer-Stey CA, Garcia-Aymerich J, Puhan MA. Antibiotics for exacerbations of chronic obstructive pulmonary disease. Cochrane database Syst Rev. 2018;10:CD010257.
- Ram FSF, Rodriguez-Roisin R, Granados-Navarrete A, Garcia-Aymerich J, Barnes NC. Antibiotics for exacerbations of chronic obstructive pulmonary disease. Cochrane database Syst Rev. 2006;(2):CD004403.
- Balter MS, La Forge J, Low DE, Mandell L, Grossman RF. Canadian guidelines for the management of acute exacerbations of chronic bronchitis. Can Respir J. 2003;10 Suppl B:3B-32B.
- Santos S, Marin A, Serra-Batlles J, et al. Treatment of patients with COPD and recurrent exacerbations: the role of infection and inflammation. Int J Chron Obstruct Pulmon Dis. 2016;11:515-525.
- Bandet T, Whitehead S, Blondel-Hill E, Wagner K, Cheeptham N. Susceptibility of clinical Moraxella catarrhalis isolates in British Columbia to six empirically prescribed antibiotic agents. Can J Infect Dis Med Microbiol 2014;25(3):155-158.
- Falagas ME, Avgeri SG, Matthaiou DK, Dimopoulos G, Siempos II. Short- versus long-duration antimicrobial treatment for exacerbations of chronic bronchitis: a meta-analysis. J Antimicrob Chemother. 2008;62(3):442-450.
- El Moussaoui R, Roede BM, Speelman P, Bresser P, Prins JM, Bossuyt PMM. Short-course antibiotic treatment in acute exacerbations of chronic bronchitis and COPD: a meta-analysis of double-blind studies. Thorax. 2008;63(5):415-422.
- Stolbrink M, Amiry J, Blakey JD. Does antibiotic treatment duration affect the outcomes of exacerbations of asthma and COPD? A systematic review. Chron Respir Dis. 2018;15(3):225-240.
- Anthonisen NR, Skeans MA, Wise RA, Manfreda J, Kanner RE, Connett JE. The effects of a smoking cessation intervention on 14.5-year mortality: a randomized clinical trial. Ann Intern Med. 2005;142(4):233-239.





I’m curious why you reommend Varenicline + NRT when the Baker study shows no difference between Varenicline, NRT and Varenicline + NRT. I am a fan of Dual NRT, using patches + lozenges (due to the frequent incorrect use of gum). However Hajek suggests that ecigarettes may be superior to even Dual NRT.