You may have seen the headlines; E-cigarette and Vaping Associated Lung Injury (EVALI) represent a new disease process that is potentially life-threatening. Emergency Physicians will inevitably be exposed to EVALI as front line health care providers. Here, we will outline the definition, treatment, and counseling for patients with EVALI in the ED and explore the Public Health concerns related to Vaping and E-cigarette use in North America.
Vaping and Electronic Nicotine Dispensing Systems (ENDS)
- E-Cigarettes are a part of a group of devices known as Electronic Nicotine Dispensing Systems (ENDS).
- ENDS were originally developed in 2003 in China by pharmacist Hon Lik who was a long-term smoker seeking a safer alternative to smoking after his father passed away from lung cancer caused by cigarette smoking. (3)
- Many different versions of ENDS have entered the market as Nicotine Replacement Therapy (NRT).
- E-Cigarettes are cigarette-shaped device containing a nicotine-based liquid that is vaporized and inhaled, used to simulate the experience of smoking tobacco (1).
- Vaping is the action or practice of inhaling and exhaling the vapor produced by an electronic cigarette or similar device. (1)
E-Cigarette as Nicotine Replacement Therapy
- E-Cigarettes are not currently approved by the FDA for NRT (2).
- There is insufficient evidence to recommend e-cigarettes as a smoking cessation aide (5).
- A significant number of people are using both cigarettes and ENDS simultaneously instead of breaking their nicotine addiction (6).
- ENDS appears to be more effective than placebo for NRT, but there is no evidence to support ENDS’ superiority to the nicotine patch alone for NRT (7).
A Public Health Crisis
There has been a decline of cigarette use in the U.S. over the past 40 years, most notably in the adolescent population. This decline in smoking is a major public health victory through the implementation of significant public health awareness campaigns and restrictions.
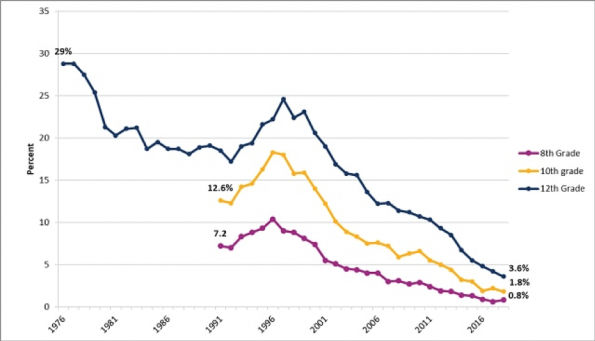
Unfortunately, the use of ENDS has largely replaced the use of cigarettes with increased popularity which is at least partially associated with a lack of market regulation. Companies such as Juul, IQOS and Blu have been able to capitalize on a market of young individuals who are not addicted to nicotine.
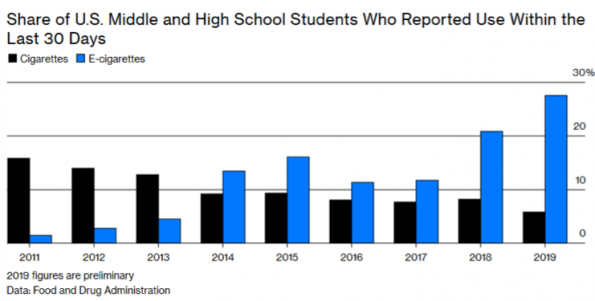
These companies have been successful by rehashing advertising strategies that have long since been restricted in the tobacco industry. These strategies are similar to those used by the tobacco industry earlier in the 20th Century and are being clearly directed at youth rather than adults with smoking cessation goals. The tobacco industry has potentialized the ENDS market with large investments; replacing the loss of the cigarette smoking clientele. Altria, the largest American cigarette company, recently purchased 35% of Juul for 12.8 Billion dollars, indicating how valuable they believe ENDS to be. (8)
E-cigarette and Vaping Associated Lung Injury (EVALI)
Epidemiology
- There has been 2,290 confirmed cases of EVALI and 47 deaths in the United States.
- There are fewer reported cases in Canada although the total number of cases is unclear as per Health Canada.
- EVALI is affecting a younger population (77% of patients under the age of 35 years old and a median age of 24).
- THC containing products have been associated with 83% of EVALI cases.
- Many of the cases have been associated with the purchase of altered ENDS products purchased from friends, families or off the street. (9)
Clinical Presentation
The presentation of EVALI is quite variable. Typically, patients with EVALI will have symptoms presenting in a subacute course over weeks. There are case reports of more fulminant courses over days, but these cases appear to be outliers. (11) EVALI symptoms are predominantly respiratory and gastrointestinal in manifestation.
Respiratory Symptoms:
-
- Cough
- Shortness of breath
- Chest pain
- Hemoptysis
Gastrointestinal Symptoms:
-
- Abdominal pain
- Nausea
- Vomiting
- Diarrhea
Physical Exam:
- Fever
- Tachycardia
- Hypoxia
- Tachypnea
Most will have findings on respiratory exam in keeping with increased work of breathing and crackles on examination of lung fields. Approximately 25% of patients will have a normal exam, especially early in the disease process, which can contribute to the difficulty in diagnosing EVALI. (11)
Investigations
- Blood work
- Elevated WBC with neutrophil predominance
- Elevated ESR and CRP
- Transaminitis
- Chest x-ray
- diffuse bilateral interstitial opacifications
- focal consolidations
- Normal chest X-ray is uncommon
- CT Thorax
- Diffuse ground-glass opacification with mild septal thickening
Diagnostic Criteria
The Centre of Disease Control and Prevention has outlined a diagnostic criteria for both confirmed and probable cases of EVALI.


Etiology
- Substances including additives, heavy metals, adulterants, and flavourings were suspected as the causative agent.
- Recently, the CDC released results from the analysis of bronchoalveolar lavage samples from 29 confirmed cases of EVALI in 10 different states identifying Vitamin E Acetate as the likely substance responsible for the disease (9).
- Vitamin E Acetate is an additive utilized as a thickening agent in e-cigarette liquid, most commonly with THC containing e-liquids.
- Vitamin E Acetate is found in many types of foods and even cosmetic products and is known to be safe when ingested. When vaporized and inhaled it appears to be toxic to the lungs (9).
Pathophysiology
- The exact pathophysiology of how EVALI arises from exposure to Vitamin E Acetate is unclear.
- It is known that EVALI is not an infection mediated process, even though damage to the lungs from EVALI may make a patient prone to a bacterial superinfection.
- Early suspicions believed that EVALI was a form of lipoid pneumonia (13-14) however, a recent article by Butt et al. in NEJM assessed histopathology from 17 identified cases of EVALI and found no evidence of lipoid pneumonia.
- Evidence of acute lung injury and ARDS were found, as well as, hypersensitivity pneumonitis, organizing pneumonia, diffuse alveolar hemorrhage (15).
- At this time EVALI likely represents a spectrum of acute lung injury responses to exposure to vaping as opposed to a single disease process.
Treatment
There are 3 mainstay of treatment of EVALI:
- Empiric antibiotics
- Due the difficulty in differentiating EVALI from bacterial pneumonia, it is reasonable to empirically treat all patients with suspected EVALI with antibiotics (9,11)
- Systemic steroids
- An observational case series of 46 patients receiving systemic steroids revealed a clinical improvement(13).
- The efficacy of steroids in EVALI is variable and likely represents the spectrum of responses to acute lung injury that has been described in EVALI.
- The decision to initiate steroid therapy should be individualized to each clinical presentation.
- In mild to moderate cases of EVALI, the decision to initiate steroids may be delayed until results of infectious testing is complete.
- In severe cases, including when mechanical ventilation is initiated, it is advisable to initiate systemic steroids given the life-threatening nature of severe EVALI. (11)
- Supportive care
- General supportive care is recommended in all cases of EVALI.
- ARDS has been seen in severe cases of EVALI requiring mechanical ventilation. In these cases, a lung protective strategy based off ARDSnet protocol or in keeping with evidence-based ventilator strategies is suggested to help reduce morbidity and mortality.
- There have also be documented cases of EVALI that require ECMO. (9-12, 14)
Take Home Points
- Ask about E-cigarettes and Vaping during your next patient encounter.
- Include vaping along with questions about smoking, EtOH and drug use while gathering information about past medical history.
- Any cases of EVALI should be reported to your local public health agency.
- Counsel patients on the risks of ENDS and vaping.
- Provide clear return to ED instructions if you are discharge patients with suspected EVALI as this disease has a subacute course.
- Treat with empiric antibiotics as bacterial pneumonia cannot be reliably ruled out.
- Consider steroids for more severe cases of EVALI.
References
- Oxford Dictionary
- CDC: About Electronic Cigarettes (E-Cigarettes). (2019, November 8). Retrieved from https://www.cdc.gov/tobacco/basic_information/e-cigarettes/about-e-cigarettes.html. LA Times (https://www.latimes.com/archives/la-xpm-2009-apr-25-fg-china-cigarettes25-story.html)
- Historical Timeline of Electronic Cigarettes. (2019, November 18). Retrieved from http://www.casaa.org/historical-timeline-of-electronic-cigarettes/.
- S. Preventive Services Task Force Final Recommendation Statement. (n.d.). Retrieved November 8, 2019, from https://www.uspreventiveservicestaskforce.org/Page/Document/RecommendationStatementFinal/tobacco-use-in-adults-and-pregnant-women-counseling-and-interventions1.
- Caraballo RS, Shafer PR,Patel D, Davis KC, McAfee TA. Quit Methods Used by US Adult Cigarette Smokers, 2014–2016. Prev Chronic Dis 2017;14:160600. DOI: https://doi.org/10.5888/pcd14.160600
- Hartmann-Boyce J, McRobbie H, Bullen C, Begh R, Stead LF, Hajek P. Electronic cigarettes for smoking cessation. Cochrane Database of Systematic Reviews 2016, Issue 9. Art. No.: CD010216. DOI: 10.1002/14651858.CD010216.pub3
- Johnston, L. D., Miech, R. A., O’Malley, P. M., Bachman, J. G., Schulenberg, J. E., & Patrick, M. E. (2019). Monitoring the Future national survey results on drug use 1975-2018: Overview, key findings on adolescent drug use. Ann Arbor: Institute for Social Research, University of Michigan
- Monica, P. R. L. (2019, October 31). Vaping mistake: Altria to take $4.5 billion writedown on Juul. Retrieved from https://www.cnn.com/2019/10/31/investing/altria-juul-writedown/index.html.
- CDC; Outbreak of Lung Injury Associated with the Use of E-Cigarette, or Vaping, Products. (2019, November 21). Retrieved from https://www.cdc.gov/tobacco/basic_information/e-cigarettes/severe-lung-disease.html#latest-outbreak-information.
- Henry, T., Kligerman, S., Raptis, C., Mann, H., Sechrist, J., & Kanne, J. (2019). Imaging Finding of Vaping-Associated Lung Injury. American Journam of Roentgenology: 1-8. 10.2214/AJR.19.22251
- Electronic Cigarette and Vaping Associated Lung Injury
- Dicpinigaitis, P. V., Trachuk, P., Fakier, F., Teka, M., & Suhrland, M. J. (2019). Vaping-Associated Acute Respiratory Failure Due to Acute Lipoid Pneumonia. Lung. doi: 10.1007/s00408-019-00277-6
- Viswam, D., Trotter, S., Burge, P. S., & Walters, G. I. (2018). Respiratory failure caused by lipoid pneumonia from vaping e-cigarettes. BMJ Case Reports. doi: 10.1136/bcr-2018-224350
- Butt, Y. M., Smith, M. L., Tazelaar, H. D., Vaszar, L. T., Swanson, K. L., Cecchini, M. J., … Larsen, B. T. (2019). Pathology of Vaping-Associated Lung Injury. New England Journal of Medicine, 381(18), 1780–1781. doi: 10.1056/nejmc1913069
- Layden JE, Ghinai I, Pray I, et al. Pulmonary Illness Related to E-Cigarette Use in Illinois and Wisconsin – Preliminary Report. N Engl J Med 2019.



