What is pulseless electric activity (PEA)?
Pulseless electrical activity is diagnosed based on a patient having an organized, non-shockable rhythm and no palpable pulse. However, PEA is not always a cardiac arrest state. In many cases, patients with PEA have underlying cardiac activity and detectable arterial blood pressure.1
PEA is a spectrum of disease
Imagine a spectrum, where on one end is a dead patient, and on the other end is a patient who is alive. Everything in between can be considered PEA. There are many different terminologies that are used to describe the spectrum of PEA, many of these referring to the same thing, and they all describe different states on the spectrum of PEA.
- True electromechanical dissociation (EMD) – or ‘true PEA’ – is electrical activity with no cardiac activity, so that’s closer to the ‘dead patient’ end of the spectrum.
- On the other end is just hypotension, in a patient who is very much alive, and you may or may not be able to palpate a pulse.
- In the middle, we have what’s now commonly known as pseudo-EMD, or pseudo-PEA, where the patient looks like they might be dead, but there’s organized cardiac activity, a decent end-tidal CO2, and there’s probably a blood pressure that you can detect with an arterial line.
- This is why some will say pseudo-PEA should be considered as ‘profound shock’ or ‘end-stage hypotension.’
- Scott Weingart has also coined the phrase ‘pulseless rhythm with echocardiographic standstill’ or PRES, and ‘pulseless rhythm with echocardiographic motion’ or PREM.2

Figure 1: clinical states as they lie on the PEA spectrum of disease. EMD: electro-mechanical dissociation; PEA: pulseless electrical activity; PRES: pulseless rhythm with echocardiographic standstill; PREM: pulseless rhythm with echocardiographic motion.
Our job is to determine where the patient is on this spectrum. The diagnostic modalities that can help us manage PEA include manual pulse check, POCUS pulse check, cardiac POCUS, and an arterial line.
POCUS pulse check
- We are bad at manual pulse checks.3–5
- POCUS pulse check is a simple technique that involves placing the ultrasound probe over the carotid or femoral artery and checking to see if it’s pulsatile.6,7 It’s been shown to be feasible and change management in cardiac arrest management.6–8
- While POCUS pulse checks do not completely replace manual pulse checks, it is a useful adjunct to determine where the patient is on the PEA spectrum.
Cardiac POCUS
- Cardiac POCUS is an essential initial diagnostic test for managing PEA.
- Cardiac activity has been shown to be a strong predictor of ROSC and survival in cardiac arrest.9–11
- It’s possible for patients with cardiac standstill to survive, but chances are very small.9,11
- Patients with cardiac standstill are closer to the ‘dead patient’ end of the spectrum while patients with good cardiac activity closer to the ‘alive’ end.
Arterial lines
- Invasive blood pressure monitoring in the form of an arterial line should be a top priority for the management of PEA. It is the ultimate diagnostic tool to determine where the patient is on the spectrum of PEA.
- Arterial lines can help with hemodynamic-directed CPR: they help us detect ROSC, monitor the quality of chest compressions, and response to vasopressors.12,13
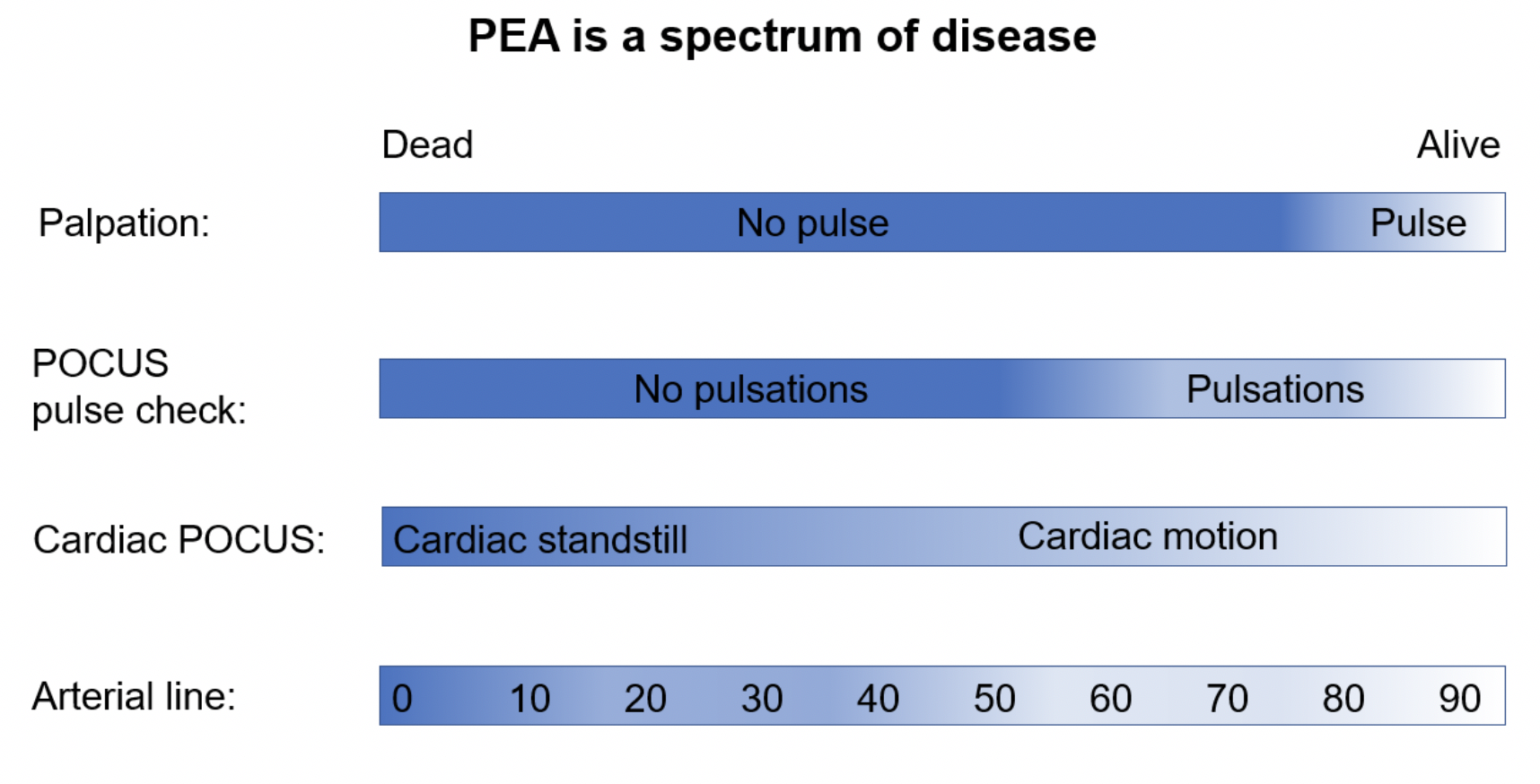
Figure 2: modalities used to classify and/or quantify a patient’s status on the PEA disease spectrum. POCUS: point-of-care ultrasound; PEA: pulseless electrical activity.
Consider using alternatives to 1mg (or 1000mcg) epinephrine (epi) boluses in PEA
- Current AHA and ILCOR guidelines for the management of PEA recommend giving 1mg of epinephrine every 3-5 minutes.14,15 However, it is unclear whether this is really the best vasopressor choice for PEA with cardiac activity.
How did 1mg epinephrine become standard practice?
Since 1906, the importance of aortic diastolic pressure during cardiac resuscitation was known, and epinephrine was shown to increase aortic diastolic pressure.16 In the 1960s, experiments with asphyxiated dogs showed epi given at a dose of 1mg increased ROSC. Survival past ROSC was not studied.17,18
Based on these experiments, epi started to show up in guidelines. It was unclear, however, whether the dose of 1mg, which was used in dogs, should be changed to allow for the difference in weight between dogs and humans.19 Many trials in the 1990s studied high-dose epinephrine and found no difference in survival compared to the standard 1mg dose.19,20 There was even perhaps a signal towards harm for high-dose epi.20
Since then, many more observational studies have consistently shown epi improves ROSC and survival to hospital admission, but there is no clear improvement in survival to hospital discharge or survival with good neurologic outcome.20 There are two major placebo-controlled RCTs on which the current 2019 AHA guidelines are based.21,22
An RCT by Jacobs et al. from Australia showed increased ROSC and survival to hospital admission in favour of epi, with no difference in survival to hospital discharge or neurologic outcomes. Despite approval by ethics boards and “clearly demonstrated clinical equipoise for adrenaline in cardiac arrest,” the trial was stopped early due to ambulance services refusing to participate, adverse press coverage and involvement of politicians questioning the ethics of withholding epi.21 Therefore, the trial was underpowered to detect any differences in long-term survival or neurologic outcome.
The PARAMEDIC-2 trial from the UK showed increased ROSC, survival to hospital admission, and survival to hospital discharge in favour of epi. The trial’s primary outcome of survival at 30 days was also positive in favour of epi (NNT=125). However, the epi group had a higher proportion of survivors at hospital discharge and 3 months with unfavourable neurologic outcome.22 Believers of epi hailed this as a landmark trial that finally demonstrates survival benefit in an RCT for epi in cardiac arrest. Skeptics saw this as further evidence to show that epi has never been demonstrated to improve neurologically favourable survival, and perhaps argues against the use of epi.
Currently, this is what we know and what we don’t know about vasopressors in cardiac arrest:

Epinephrine dose
- We know that the 1mg dose comes from animal studies in the 1960s.17,18
- High-dose is not recommended because of all the negative high-dose trials.20
- Low-dose epi is not well studied, although a new RCT is currently being planned called the CanROC EpiDOSE trial (NCT03826524).
- Recently, there has only been this one non-randomized before-and-after study that compared 1mg to 0.5mg.23 They stratified based on shockable and non-shockable rhythms and there was no difference in survival outcomes.23
- Most guidelines still recommend 1mg, but the European Resuscitation Council guidelines on intra-operative PEA actually suggests using a lower dose.24
Epinephrine timing
- Epi may have the most benefit in the first 10 minutes of cardiac arrest.20
- This theory is based on the idea that cardiac arrest can be split into three phases:25
- The first few minutes of cardiac arrest is called the electrical phase, where shockable rhythms should be shocked immediately to have the best outcomes.
- After about 4 minutes, if there is no CPR, a shock alone will be less successful than a shock with CPR—this is the circulatory phase. The idea is that you need to generate blood flow using CPR and epi during the circulatory phase to have the best outcomes, shockable rhythm or not. This is probably where epi is the most effective.
- After 10 minutes, you reach the metabolic phase, where all the adverse effects of tissue ischemia begin. Epi after 10 minutes may simply worsen all of the tissue ischemia by causing increased peripheral and cerebral vasoconstriction.25
- A large observational study found that chances of survival decreased if you received more epi, or if you got epi later into the resuscitation.26
- Therefore: earlier is better, less is better, and likely not beneficial after the first 10 minutes of cardiac arrest.20,25,26
Vasopressin
- 2nd most well-studied vasopressor for cardiac arrest.
- Causes vasoconstriction without a lot of the harmful side effects of epi, such as increased myocardial oxygen consumption, ventricular arrhythmias, and myocardial dysfunction after resuscitation.27
- Lab studies have shown that vasopressin was associated with better blood flow to vital organs, delivery of cerebral oxygen, chances of resuscitation, and better neurologic outcome compared to epi.27 Also, vasopressin levels were found to be higher in successfully resuscitated patients. Therefore, it was postulated that it might be beneficial to give vasopressin during CPR.27
- Unfortunately, the theoretical benefits of vasopressin have not been borne out in RCTs and the International Liaison Committee on Resuscitation (ILCOR) currently suggests against the use of vasopressin.15
- However, in a small prehospital study from Slovenia, vasopressin was shown to result in significantly better outcomes for PEA with cardiac activity.28
Vasopressor infusions
While many advocate for PEA to be managed as profound shock using an infusion of vasopressors, there is, unfortunately, not much evidence to support this practice. A recent prospective multi-centre observational study including 20 sites in the US and Canada studied patients specifically in PEA with cardiac activity.29 They classified the cardiac activity on POCUS as organized and disorganized. Then they compared standard ACLS medications versus a continuous infusion of adrenergic agents, which included epinephrine, norepinephrine, dopamine, dobutamine, or phenylephrine, and looked at survival outcomes. Among the patients with organized cardiac activity, the rate of ROSC was significantly higher in the continuous vasopressors group. The rate of survival to hospital and discharge were also higher but these differences were not statistically significant.29
While it’s difficult to draw any practice-changing conclusions from this study, the results support the idea of PEA being a spectrum and shows that cardiac POCUS is helpful for stratifying and prognosticating patients on that spectrum. It also shows that an infusion of vasopressors in someone who has organized cardiac activity (closer to the alive end of the PEA spectrum) is probably a reasonable idea and may be better than a 1mg bolus of epi.
Priorities in PEA
Based on the evidence reviewed here, the following would be the authors’ management algorithm for PEA, with the priorities numbered in the red boxes. Please note that ceasing chest compressions upon discovery of a POCUS pulse in the absence of a manually-detectable pulse remains
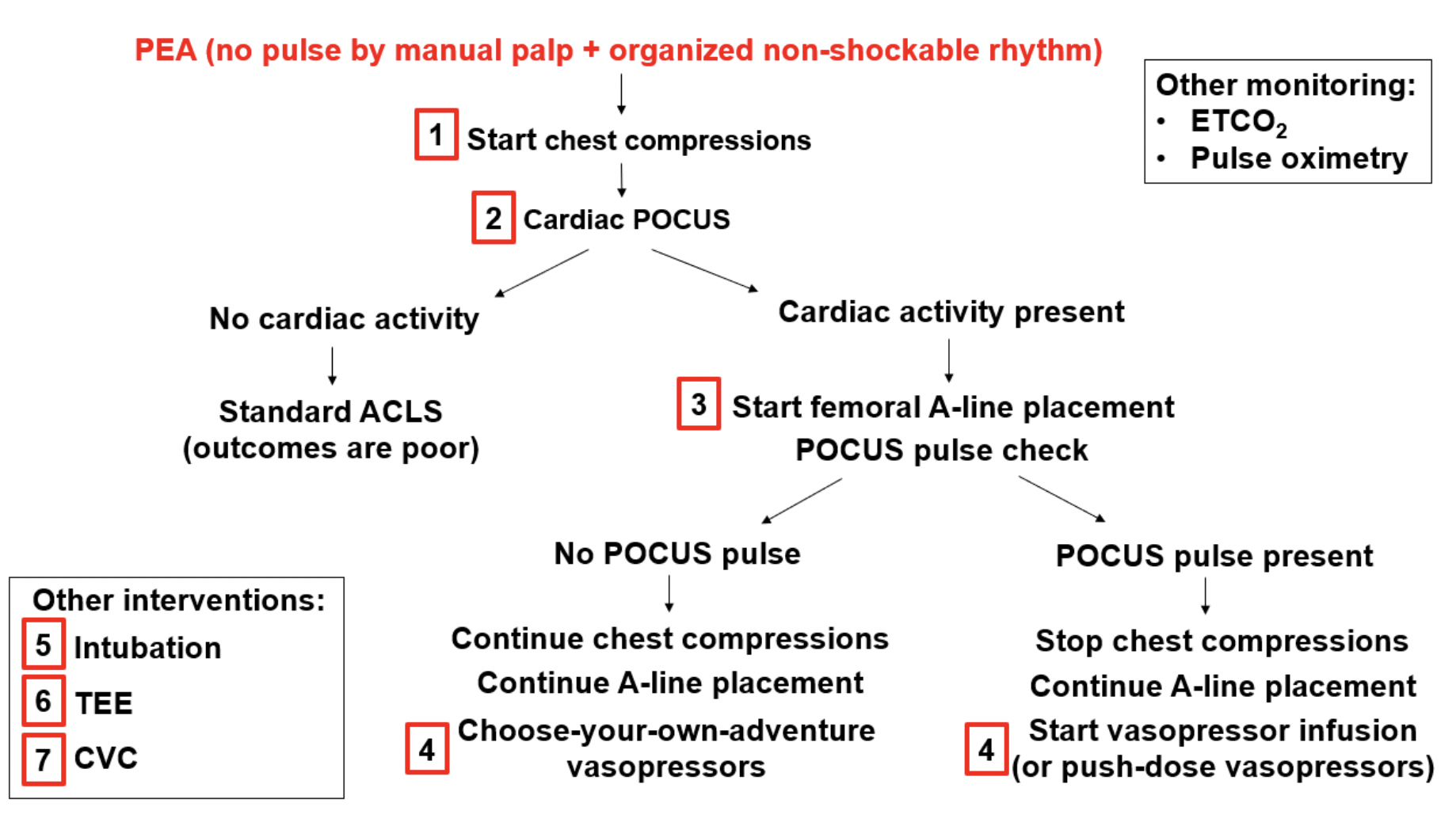
Figure 3: priority-based management algorithm for the undifferentiated patient in PEA arrest, derived by Dr. Bo Zheng. PEA: pulseless electrical activity; ETCO2: end-tidal CO2 monitoring; ACLS: advanced cardiac life support; POCUS: point-of-care ultrasound; TEE: trans-esophageal echocardiography; CVC: central venous catheter.
References
- Paradis NA, Martin GB, Goetting MG, Rivers EP, Feingold M, Nowak RM. Aortic pressure during human cardiac arrest; Identification of pseudo- electromechanical dissociation. Chest. 1992;101(1):123-128. doi:10.1378/chest.101.1.123
- Helman, A. Simard, R. Weingart, S. PEA Arrest, PseudoPEA and PREM. Emergency Medicine Cases. October, 2019. https://emergencymedicinecases.com/pea-arrest-pseudopea-prem. Accessed [November 28, 2019].
- Dick WF, Eberle B, Wisser G, Schneider T. The carotid pulse check revisited: What if there is no pulse? Crit Care Med. 2000;28(11 SUPPL.):183-185.
- Lapostolle F, Le Toumelin P, Agostinucci JM, Catineau J, Adnet F. Basic cardiac life support providers checking the carotid pulse: Performance, degree of conviction, and influencing factors. Acad Emerg Med. 2004;11(8):878-880.
- Zengin S, Gümüşboğa H, Sabak M, Eren ŞH, Altunbas G, Al B. Comparison of manual pulse palpation, cardiac ultrasonography and Doppler ultrasonography to check the pulse in cardiopulmonary arrest patients. Resuscitation. 2018;133(September):59-64.
- Simard RD, Unger AG, Betz M, Wu A, Chenkin J. The POCUS Pulse Check: A Case Series on a Novel Method for Determining the Presence of a Pulse Using Point-of-Care Ultrasound. J Emerg Med. 2019;56(6):674-679.
- Badra K, Coutin A, Simard R, Pinto R, Lee JS, Chenkin J. The POCUS pulse check: A randomized controlled crossover study comparing pulse detection by palpation versus by point-of-care ultrasound. Resuscitation. 2019;139(November 2018):17-23.
- Schonberger RB, Lampert RJ, Mandel EI, Feinleib J, Gong Z, Honiden S. Handheld doppler to improve pulse checks during resuscitation of putative pulseless electrical activity arrest. Anesthesiology. 2014;120(4):1042-1045.
- Gaspari R, Weekes A, Adhikari S, et al. Emergency department point-of-care ultrasound in out-of-hospital and in-ED cardiac arrest. Resuscitation. 2016;109:33-39.
- Wu C, Zheng Z, Jiang L, et al. The predictive value of bedside ultrasound to restore spontaneous circulation in patients with pulseless electrical activity: A systematic review and meta-analysis. PLoS One. 2018;13(1):1-13.
- Flato UAP, Paiva EF, Carballo MT, Buehler AM, Marco R, Timerman A. Echocardiography for prognostication during the resuscitation of intensive care unit patients with non-shockable rhythm cardiac arrest. Resuscitation. 2015;92:1-6.
- Marquez AM, Morgan RW, Ross CE, Berg RA, Sutton RM. Physiology-directed cardiopulmonary resuscitation: Advances in precision monitoring during cardiac arrest. Curr Opin Crit Care. 2018;24(3):143-150.
- Link MS, Berkow LC, Kudenchuk PJ, et al. Part 7: Adult advanced cardiovascular life support: 2015 American Heart Association guidelines update for cardiopulmonary resuscitation and emergency cardiovascular care. Circulation. 2015;132(18):S444-S464.
- Panchal AR, Berg KM, Hirsch KG, et al. 2019 American Heart Association Focused Update on Advanced Cardiovascular Life Support: Use of Advanced Airways, Vasopressors, and Extracorporeal Cardiopulmonary Resuscitation During Cardiac Arrest: An Update to the American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation. 2019.
- Soar J. 2019 International Consensus on Cardiopulmonary Resuscitation and Emergency Cardiovascular Care Science with Treatment Recommendations.; 2019.
- Attaran RR, Ewy GA. Epinephrine in Resuscitation: Curse or Cure? Future Cardiol. 2010;6(4):473-482.
- Redding J, Pearson J. Evaluation of Drugs for Cardiac Resuscitation. Anesthesiology. 1963;24(2):203-207.
- Pearson J, Redding J. Epinephrine in cardiac resuscitation. Am Heart J. 1963;66(2):210-214.
- Stiell I, Hebert P, Weitzman B, et al. High-dose epinephrine in adult cardiac arrest. N Engl J Med. 1992;327(15):1045-1050.
- Long B, Koyfman A. Emergency Medicine Myths: Epinephrine in Cardiac Arrest. J Emerg Med. 2017;52(6):809-814.
- Jacobs IG, Finn JC, Jelinek GA, Oxer HF, Thompson PL. Effect of adrenaline on survival in out-of-hospital cardiac arrest: A randomised double-blind placebo-controlled trial. Resuscitation. 2011;82(9):1138-1143.
- Perkins GD, Ji C, Deakin CD, et al. A Randomized Trial of Epinephrine in Out-of-Hospital Cardiac Arrest. N Engl J Med. 2018;2018:NEJMoa1806842.
- Fisk CA, Olsufka M, Yin L, et al. Lower-dose epinephrine administration and out-of-hospital cardiac arrest outcomes. Resuscitation. 2018;124:43-48.
- Truhlář A, Deakin CD, Soar J, et al. Cardiac arrest in special circumstances: Section 4 of European Resuscitation Council Guidelines for Resuscitation 2015. Notfall und Rettungsmedizin. 2015;18(8):833-903.
- Weisfeldt ML, Becker LB. Resuscitation After Cardiac Arrest. Jama. 2002;288(23):3035.
- Dumas F, Bougouin W, Geri G, et al. Is epinephrine during cardiac arrest associated with worse outcomes in resuscitated patients? J Am Coll Cardiol. 2014;64(22):2360-2367.
- Wenzel V, Krismer A, Arntz R, Sitter H, Stadlbauer K, Lindner K. A comparison of vasopressin and epinephrine for out-of-hospital cardiopulmonary resuscitation. N Engl J Med. 2004;350(2):105-113.
- Prosen G, Križmarić M, Zavrašnik J, Grmec Š. Impact of modified treatment in echocardiographically confirmed pseudopulseless electrical activity in out-ofhospital cardiac arrest patients with constant end-tidal carbon dioxide pressure during compression pauses. J Int Med Res. 2010;38(4):1458-1467.
- Gaspari R, Weekes A, Adhikari S, et al. A retrospective study of pulseless electrical activity, bedside ultrasound identifies interventions during resuscitation associated with improved survival to hospital admission. A REASON Study. Resuscitation. 2017;120:103-107.




During CPR on traumatic arrest, with a LUCAS device, is a bloop pressure accurate for the movement from the compressions? Can I have a BP on the monitors if no pulses are felt in PEA?