Community-acquired pneumonia is the leading infectious cause of death in Canada, and combined with influenza is the 7th most common cause of death overall.
Diagnosis
The 3 main pillars leading to the diagnosis of pneumonia are as follow:
- Evidence of infection (fever, chills, leukocytosis)
- Signs and symptoms localized to the respiratory system (cough, SOB, sputum, chest pain, crackles on auscultation, elevated RR, low SpO2)
- Chest imaging (new or changed infiltrate)
Although this seems straightforward, pneumonia is often misdiagnosed. According to a 2019 national audit by the British Thoracic Society, the admission diagnosis of pneumonia was accurate only 63.3% of the time.1 Moreover, this only includes cases where pneumonia was over-called, not cases where pneumonia was missed.
Potential Causes of Misdiagnosis
- Atypical presentations
- Patients can present with a host of “non-classic”
- Symptoms including; myalgia, abdominal pain, back pain, headache or dizziness. 2
- Elderly patients are more likely to present atypically and often possess fewer of the cardinal symptoms.2,3
- Confusion may be the only presenting complaint.4
- Patients can present with a host of “non-classic”
- ED time pressure to make a diagnosis
- Administrative pressures for rapid diagnosis challenge physicians faced with atypical presentations.
- There is lack of ability for continuous monitoring and reassessments that would allow a diagnosis to ‘declare itself’.
- Poor predictive value of the history and physical exam
- There are no individual items from the clinical history or physical exam that raise or lower the probability of pneumonia enough to either confirm or exclude the diagnosis.2,5
- Weak positive and negative likelihood ratios for individual clinical signs and symptoms have very little impact on our post-test probability of disease.5
- Even worse, experienced physicians often can’t agree about whether a given clinical finding is even present.5
- Kappa value for tachypnea = 0.25 (poor)
- Kappa value for crackles on auscultation = 0.41 (fair)
- Kappa value for wheezes on auscultation = 0.51 (fair)
- Poor predictive value of the chest x-ray
- In an ED study of 324 patients with suspected CAP that underwent both chest x-ray and CT imaging, CT scan revealed parenchymal infiltrates in 33% of patients that had a normal chest x-ray. Moreover, CT scan excluded pneumonia in 29.8% patients that initially had an infiltrate on x-ray.6 x-ray both over-called and under-called pneumonia in approximately one-third of cases.
- In another ED study comparing CXR to CT, CXR over-called pulmonary opacities compared with CT scan. Of the pulmonary opacities identified on x-ray, only 26.9% were confirmed on CT. Possible explanations for this include poor radiograph quality, poor inspiratory effort, and better delineation of alternative diagnoses such as CHF and atelectasis on CT. In this study, x-ray had a sensitivity of only 43%.7
- In a study that looked at patients admitted with a clinical diagnosis of pneumonia with initial negative radiographs, 50% developed infiltrates within 48 hours on repeat x-ray.8
- In a study that looked at the agreement between staff radiologists on the presence/ absence of an infiltrate on chest x-ray, the overall Kappa coefficient was found to be 0.37 (poor) with disagreement in 1 / 7 cases.9
- Lack of a readily available gold standard
- Given the inherent flaws of both the clinical encounter and chest x-ray at diagnosing pneumonia, the ‘gold standard’ in many studies is either the final discharge diagnosis or a consensus opinion from a panel of experts that takes into account all variables from the entire hospital length of stay.
- Ultrasound has consistently outperformed chest x-ray with sensitivity and specificity for diagnosing pneumonia typically in the 80-90% range depending on the study.2,10 The main drawbacks to ultrasound remain time and operator experience, but can certainly be a useful adjunct in the right hands.
- The sensitivity of CT for identifying pneumonia approaches 100%2,6 although increased ED length of stay, cost, and radiation exposure limit its utility for the routine workup of pneumonia patients.
Approach to Diagnosis
- Largely due to the lack of a readily available alternative, it is my opinion that the gold standard for diagnosing community-acquired pneumonia remains a thorough clinical assessment combined with the presence of an infiltrate on chest x-ray.
- Establishing a pre-test probability based on the clinical encounter before interpreting the chest x-ray becomes fundamentally important.
- Very high pretest probability → treatment without chest x-ray or indeterminate chest x-ray with antibiotics should be considered.
- High pretest probability + infiltrate on chest x-ray → treat with antibiotics
- Very low pre-test probability → forgoing the chest x-ray may be an appropriate choice.
- In young otherwise healthy patients, a chest x-ray may not be necessary, OR, if completing a chest x-ray, treatment should only be offered when there is a definitive consolidation present.
Pneumonia Prevalence
- It is estimated that the prevalence of pneumonia in ambulatory patients with cough and either sputum, fever, or hemoptysis is approximately 10%.11
- Given this low prevalence, a normal physical examination coupled with the absence of any vital sign abnormalities results in a probability of pneumonia that falls below 5%.
Microbiology
- Viral
- Common pathogens:
- Influenza
- Rhinovirus
- RSV
- Coronaviruses
- In a 2017 systematic review that included both inpatient and outpatient cases of CAP, a pooled 22% had a virus isolated.27
- In more recent studies where the newer more sensitive real-time multiplex polymerase chain reaction (PCR) testing is performed, this number is even higher, approaching 30%.28
- In a study where patients with CAP were compared with asymptomatic controls, the rate of viral detection was 24.5% vs 2.1% respectively providing evidence that viruses likely play a true pathologic role.29
- In up to 2/3 of cases, >1 pathogen was identified, often a combination of a virus and bacteria.30 Viral infections can therefore lead to a break-down of host defences predisposing to a subsequent bacterial pneumonia.
- Due to the high rate of co-infection, the identification of a virus is not enough to discontinue antibiotic therapy.
- Common pathogens:
- Bacterial
- Common pathogens:
- Streptococcus pneumoniae (most common)
- Annual incidence has decreased 9.6- 8.9 cases / 100,000 population since implementation of routine immunization programs for streptococcus in 2010.
- Mycoplasma pneumonia
- Haemophilus influenzae
- Moraxella catarrhalis
- Klebsiella pneumoniae
- Streptococcus pneumoniae (most common)
- Gram-negatives appear to be much less common in the outpatient setting (11-24%).
- In a recent study, viruses and bacteria were identified in 87% of cases.
- Common pathogens:
- MRSA / Pseudomonas
- Healthcare-associated pneumonia (HCAP) has been removed in the 2019 IDSA/ATS guidelines as it was found to be poorly predictive of antibiotic-resistant pathogens.20 This categorization led to a significant increase in the use of broad-spectrum antibiotics without any demonstrated improvement in patient outcomes.20
- The 2 strongest individual predictors for possible MRSA or Pseudomonas pneumonia include:
- Prior isolation of these organisms, particularly from the respiratory tract
- Common in patients with cystic fibrosis, bronchiectasis, or severe COPD
- Empiric treatment, sputum and blood cultures are recommended
- Hospitalization in the previous 90 days with receipt of IV antibiotics
- Sputum and blood cultures are recommended, but not empiric coverage, unless of course the patient is really sick.
- Prior isolation of these organisms, particularly from the respiratory tract
Investigations
- Blood cultures: not recommended for regular CAP patients seen in the ED as the rate of false positives equals and often exceeds the rate of true positives.48 The latter has the propensity to significantly increase hospital length of stay and duration of antibiotic therapy.20 In CAP patients with truly positive blood cultures (2-9%)20, management is rarely changed. In the setting of severe CAP, sepsis, or when a resistant organism is suspected, blood cultures are indicated as they have the potential to help tailor, and potential narrow, antibiotic therapy down the line leading to improved anti-microbial stewardship.
- Sputum Cultures: not recommended for regular CAP patients seen in the ED as it will rarely lead to a change in management. Similar to above, for patients with severe CAP or sepsis, sputum cultures are indicated as more virulent pathogens such as Legionella, Staph Aureus, and Pseudomonas become more common in these subgroups.20
- Legionella Urinary Antigen: not recommended for regular CAP patients seen in the ED as it remains a relatively rare cause of CAP. Often found in clusters, it is not associated with person-person transmission but rather exposure to a contaminated water source.49 Clinical features that would raise the possibility of Legionella pneumonia include an ongoing outbreak in the area, recent travel history, or extrapulmonary symptoms including diarrhea.49 Urinary antigen testing for legionella would be indicated if there is a high suspicion based on the presence of the above features, or in the setting of severe pneumonia/sepsis.
Disposition
- Mortality rates for community-acquired pneumonia vary drastically based on the severity of disease(5-8%).2,12–15
- Most of these deaths occur in patients over the age of 65.16
- Physicians tend to overestimate the probability of poor outcomes for patients diagnosed with CAP.13,17 This can lead to potentially unnecessary admissions and increased healthcare costs. A recent briefing on the economic burden of pneumonia in Canada revealed that the average direct cost per case to be over $8,000.18 Inpatient services account for an estimated 90% of these costs.19
- Due in part to the significant costs associated with inpatient treatment, several national organizations (IDSA, ATS, BTS, NICE) have strongly recommended the use of a risk assessment tool to help guide disposition decisions for patients presenting to hospital with community-acquired pneumonia.20–22
Risk Assessment Tool
-
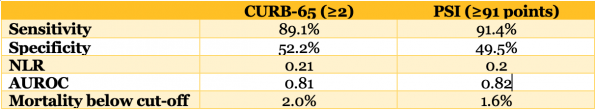
- The Pneumonia Severity Index (PSI) and the CURB-65 rule are the 2 tools that have been most robustly studied.
- Each tool risk stratifies patients based on 30-day mortality.
- A 2010 systematic review and meta-analysis that compared the CURB-65 with the PSI for predicting mortality in hospitalized patients with radiographically confirmed CAP, the sensitivity, specificity, negative likelihood ratios and ROC curves were all very comparable.23
-
- Pneumonia Severity Index (PSI)
- An issue arises with these risk assessment tools; they are being used for safe discharge rather than mortality risk. A tool aiding in safe discharge is needed – only the PSI has been validated for this purpose.13,17,19,2
- PSI compared with standard care lead to an 18% absolute reduction in the admission rate of low-risk patients and a 10% decrease in admissions overall.19
- Multiple other studies have confirmed decreased admission rates for low-risk patients ranging from 15-37%.13,17,24
- CURB-65 Score
- Has never been prospectively studied as a disposition tool.
- In a multi-centre retrospective ED study of 21,000 patients looking at 30-day mortality, the CURB-65 score again performed well with a sensitivity of 92.8%, negative predictive value of 99%, and a negative likelihood ratio of 0.2.12
- If a CURB-65 score of ≤1 had been used as a strict discharge cut-off in this trial, the rate of admissions would have decreased by 26%. With this said, 2.1% of patients discharged would have died compared with the 0.7% observed in the actual study.
- Due to the retrospective nature, we have no insights into the rate of ED bounce backs or other adverse events aside from mortality. In another retrospective study, a panel consisting of one ID physician, one internist, and 3 respirologists reviewed all admitted CAP patients that were considered low risk based on the CURB-65 score. They found that in 83% of cases, admission was reasonable even in this low-risk cohort.
- The most common reasons for admission despite a low CURB score were hypoxia, failure of outpatient therapy, and cardiovascular events.25
- In my opinion, the CURB-65 Score is most useful where the mortality rate overall is quite low – the NPV in high 90s of the CURB-65 can support a safe discharge home.
- There are a host of other factors to consider (home supports, access to follow-up, etc) but I certainly believe there can be a role for this tool as an adjunct to physician gestalt. It could lead to fewer admissions.
- Pneumonia Severity Index (PSI)
Approach to Outpatient Treatment
-
- Antibiotic Selection
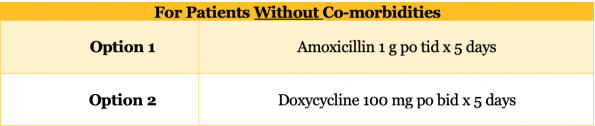
- Patient WITHOUT co-morbidities
- Antibiotic Selection
-
-
-
- The big change from the 2007 IDSA guidelines is the absence of a strong recommendation for macrolide monotherapy for regions where Strep pneumoniae resistance exceeds 25%. In Ottawa, based on the 2019 antibiogram, our resistance rate is 30%.
- Amoxicillin and Doxycycline both demonstrate excellent Strep pneumoniae coverage and have a more favourable side effect profile than fluoroquinolones.
- Several studies have looked at high dose Amoxicillin monotherapy for both inpatient and outpatient CAP and demonstrated excellent efficacy despite its relative lack of atypical coverage.36,37
- In a study that comparing Doxycycline vs Levofloxacin for inpatient CAP, Doxycycline demonstrated comparably low clinical failure rates and a lower hospital length of stay.38
-
-
-
-
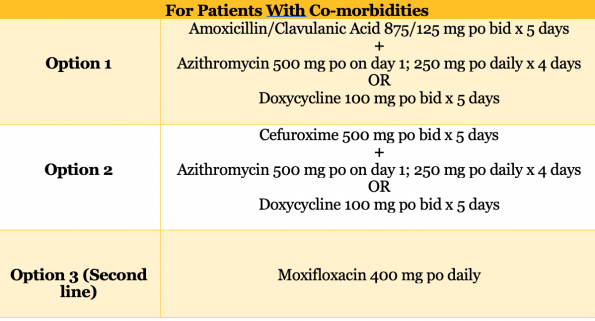
- Patient WITH co-morbidities
- Co-morbidities including: chronic heart, lung, liver or renal disease, diabetes, alcoholism, malignancy and asplenia.
- Patient WITH co-morbidities
-
-
-
-
- A 2014 Cochrane review, as well as 16 RCTs and meta-analysis reviewed in the 2019 IDSA guidelines, revealed no significant differences in relevant patient outcomes when any of the above antibiotic regimens were compared for outpatient CAP.20,39
- Despite this paucity of evidence, the expanded coverage for patients with co-morbidities was recommended for two main reasons.
- Increased vulnerability to poor outcomes if the initial antibiotic regimen fails.
- More likely to have a history of contact with the hospital system.
-
-
2. Duration of Antibiotics
-
-
- The recommended duration for outpatient antibiotic therapy for community-acquired pneumonia is now 5 days representing a significant change compared with previous practice patterns.
- In a multicenter RCT published in JAMA in 2016, a 5 day course of antibiotics was found to be non-inferior to a 10 day course in terms of clinical success at 10 and 30 days.40 There was also no significant difference in mortality or other clinically important adverse events. Multiple other studies have replicated these findings41,42 providing us with good evidence that shorter courses of antibiotics are both safe and probably the favoured approach.
-
3. Treatment Failure
-
-
- IDSA guidelines from 2007 provide two broad definitions:
-
-
-
-
- Progression of pneumonia with evidence of clinical deterioration
- Persistent or non-responding pneumonia
-
-
-
-
- The first steps to ensure patients returning to the ED are on appropriate antibiotics. As the updated IDSA guidelines are recent, macrolide monotherapy may still be seen which may be inadequate for Strepcoccus Pneumoniae coverage.
- If they are on appropriate antibiotics and you believe they are truly worsening clinically, this is probably an indication for admission to hospital. Concerning non-responding pneumonia, this is defined by the IDSA as absence or delay in reaching clinical stability.43
-
-
-
- Median time to clinical stability of 3 days, with 25% of patients requiring up to 6 days.43 Therefore, a patient returning to the ED before day 3, may not have reached clinical stability yet.
- As such, prior to day 3, assuming appropriate antibiotic treatment, we should not be switching a patient’s antibiotics unless we feel they are clinically worsening.
- Moreover, we don’t expect patients to feel much better from their pneumonia until approximately day 12. 40
- After day 3, if they still have not met all of the above signs of clinical stability, then admission may be reasonable as they are not responding to our treatment to the extent that we would expect. They may need expanded antibiotic coverage and closer inpatient monitoring.
-
Approach to Inpatient Treatment
1. Non-Severe Community Acquired Pneumonia

-
-
- Gram negatives such as H. influenzae and M. catarrhalis, which often produce beta-lactamase, are more common in the inpatient setting.20
- The evidence for beta-lactam monotherapy in the setting of CAP patients being admitted to hospital is mixed. A 2015 RCT in the New England Journal showed no difference in 90-day mortality when comparing beta-lactam monotherapy vs beta-lactam + macrolide combination therapy vs fluoroquinolone monotherapy.44
- Conversely, another well-done RCT published in JAMA could not demonstrate non-inferiority of beta-lactam monotherapy when compared with beta-lactam plus macrolide combination therapy when looking at the time to clinical stability.45
- A systematic review looking at the totality of the evidence which includes these two trials in addition to a series of lower quality studies concluded that combination therapy was superior to beta-lactam monotherapy.46 For this reason, beta-lactam monotherapy is not included as a treatment option in the inpatient setting. With this said, the benefit to combination therapy seems to be greater in more severe cases of pneumonia where the rate of legionella is known to be higher.
-
2. Severe Community Acquired Pneumonia

-
-
- In the setting of severe CAP, observational data suggests that macrolide containing therapies have a mortality benefit (3% ARR).47 With this said, the quality of the studies was low, and the definitions for severe CAP were variable.
-
References
- Lim WS, Lawrence H. Adult Community Acquired Pneumonia Audit 2018-2019. Br Thorac Soceity Reports. 2019;10(4):1-13.
- Long B, Long D, Koyfman A. Emergency Medicine Evaluation of Community-Acquired Pneumonia: History, Examination, Imaging and Laboratory Assessment, and Risk Scores. J Emerg Med. 2017;53(5):642-652. doi:10.1016/j.jemermed.2017.05.035
- Metlay JP, Schulz R, Li Y, et al. Influence of age on symptoms at presentation in community- acquired pneumonia. Arch Intern Med. 1997;157:1453-1459.
- Waterer GW, Kessler LA, Wunderink RG. Delayed Administration of Antibiotics Community-Acquired Pneumonia. Chest. 2006;130(1):11-15. doi:10.1378/chest.130.1.11
- Metlay J, Kapoor W, Fine Mi. Does This Patient Have Community-Acquired Pneumonia ? JAMA. 1997;278(17):1440-1445.
- Claessens Y-E, Debray M-P, Tubach F, et al. Early Chest Computed Tomography Scan to Assist Diagnosis and Guide Treatment Decision for Suspected Community-acquired Pneumonia. Am J Respir Crit Care Med. 2015;192(8):974-982. doi:10.1164/rccm.201501-0017OC
- Self WH, Courtney DM, Mcnaughton CD, Wunderink RG, Kline JA. High discordance of chest x-ray and computed tomography for detection of pulmonary opacities in ED patients : implications for diagnosing pneumonia ☆. Am J Emerg Med. 2013;31(2):401-405. doi:10.1016/j.ajem.2012.08.041
- Hagaman JT, Panos RJ, Rouan GW, Shipley RT. Admission Chest Radiograph Lacks Sensitivity in the Diagnosis of Community-Acquired Pneumonia. Am J Med Sci. 2009;337(4):236-240.
- Albaum MN, Hill LC, Murphy M, et al. lnterobserver Reliability of the Chest Radiograph in Community-Acquired Pneumonia. Chest. 1996;110(2):343-350. doi:10.1378/chest.110.2.343
- Llamas-álvarez AM, Tenza-lozano EM, Latour-pérez J. Accuracy of Lung Ultrasonography in the Diagnosis of Pneumonia in Adults Systematic Review and Meta-Analysis. Chest. 2017;151(2):374-382. doi:10.1016/j.chest.2016.10.039
- Emerman CL, Dawson N, Speroff T, et al. Comparison of Physician Judgment and Decision Aids for Ordering Chest Radiographs for Pneumonia in Outpatients. Ann Emerg Med. 1991;20(11):1215-1219.
- Sharp AL, Jones JP, Wu I, et al. CURB-65 Performance Among Admitted and Discharged Emergency Department Patients With Community-acquired Pneumonia. Acad Emerg Med. 2016:400-405. doi:10.1111/acem.12929
- Yealy D, Auble T, Stone R, et al. Effect of Increasing the Intensity of Implementing Pneumonia Guidelines. Ann Intern Med. 2005.
- Loke YK, Kwok CS, Niruban A, Myint PK. Value of severity scales in predicting mortality from community-acquired pneumonia : systematic review and meta-analysis. Thorax. 2010;65:884-890. doi:10.1136/thx.2009.134072
- Aujesky D, Auble TE, Yealy DM, et al. Prospective comparison of three validated prediction rules for prognosis in community-acquired pneumonia. Am J Med. 2005;118:384-392. doi:10.1016/j.amjmed.2005.01.006
- Williams S, Gousen S, DeFrances C. National Hospital Care Survey Demonstration Projects: Pneumonia Inpatient Hospitalizations and Emergency Department Visits.; 2018.
- Atlas S, Benzer T, Borowsky L, et al. Safely Increasing the Proportion of Patients With Community-Acquired Pneumonia Treated as Outpatients. Arch Intern Med. 1998;158:1350-1356.
- Canada TCB of. The Economic Burden of Pneumonia in Canada .; 2017.
- Marrie TJ, Lau CY, Wheeler SL, et al. A Controlled Trial of a Critical Pathway for Treatment of Community-Acquired Pneumonia available demonstrating that they either. JAMA. 2000;283(6).
- Metlay JP, Waterer GW, Long AC, et al. Diagnosis and Treatment of Adults with Community-acquired Pneumonia: An Official Clinical Practice Guideline of the American Thoracic Society and Infectious Diseases Society of America. Am J Respir Crit Care Med. 2019;200(7):e45-e67. doi:10.1164/rccm.201908-1581ST
- National Institute for Health and Care Excellence (NICE). Pneumonia in Adults : Diagnosis and Management.; 2014.
- British Thoracic Society. Annotated BTS Guideline for the Management of CAP in Adults.; 2015. doi:10.1136/thx.2009.121434
- Chalmers JD, Singanayagam A, Akram AR, et al. Severity assessment tools for predicting mortality in hospitalised patients with community-acquired pneumonia . Systematic review and meta-analysis. Thorax. 2010;65:878-883. doi:10.1136/thx.2009.133280
- Jo S, Kim K, Jung K, et al. The Effects of Incorporating a Pneumonia Severity Index into the Admission Protocol for Community-Acquired Pneumonia. J Emerg Med. 2012;42(2):133-138. doi:10.1016/j.jemermed.2010.04.018
- Aliberti S, Ramirez J, Cosentini R, et al. Low CURB-65 is of limited value in deciding discharge of patients with community-acquired. Respir Med. 2011;105(11):1732-1738. doi:10.1016/j.rmed.2011.07.006
- Kelly A. Can VBG analysis replace ABG analysis in emergency care ? Emerg Med J. 2016;33:152-154. doi:10.1136/emermed-2014-204326
- Alimi Y, Lim WS, Lansbury L, Leonardi-bee J, Nguyen-van-tam JS. Systematic review of respiratory viral pathogens identi fi ed in adults with community-acquired pneumonia in Europe. J Clin Virol. 2017;95(July):26-35. doi:10.1016/j.jcv.2017.07.019
- Tatarelli P, Magnasco L, Borghesi M, Russo C. Prevalence and clinical impact of VIral Respiratory tract infections in patients hospitalized for Community ‑ Acquired Pneumonia : the VIRCAP study. Intern Emerg Med. 2019. doi:10.1007/s11739-019-02243-9
- Self WH, Williams DJ, Zhu Y, et al. Respiratory Viral Detection in Children and Adults : Comparing Asymptomatic Controls and Patients With Community-Acquired Pneumonia. J Infect Dis. 2016;213. doi:10.1093/infdis/jiv323
- Wunderink RG, Waterer G. Advances in the causes and management of community acquired pneumonia in adults. Br Med J. 2017;358:1-13. doi:10.1136/bmj.j2471
- Gadsby NJ, Russell CD, Mchugh MP, et al. Comprehensive Molecular Testing for Respiratory Pathogens in Community-Acquired Pneumonia. Clin Infect Dis. 2016;62:817-823. doi:10.1093/cid/civ1214
- Torres A, Blasi F, Peetermans WE, Viegi G, Welte T. The aetiology and antibiotic management of community-acquired pneumonia in adults in Europe : a literature review. Eur J Clin Mircobiology Infect Dis. 2014;33:1065-1079. doi:10.1007/s10096-014-2067-1
- Jain S, Self W, Wunderink R, Fakhran R, Balk R. Community-Acquired Pneumonia Requiring Hospitalization among U.S. Adults. N Engl J Med. 2015;373(5):415-427. doi:10.1056/NEJMoa1500245
- Public Health Agency of Canada. Canadian Antimicrobial Resistance Surveillance System Report.; 2016.
- Musher DM, Abers MS, Bartlett JG. Evolving Understanding of the Causes of Pneumonia in Adults , With Special Attention to the Role of Pneumococcus. Clin Infect Dis. 2017;65(Table 1). doi:10.1093/cid/cix549
- Hagberg L, Torres A, Rensburg D Van, Leroy B, Rangaraju M, Ruuth E. Efficacy and Tolerability of Once-Daily Telithromycin Compared with High-Dose Amoxicillin for Treatment of Community-Acquired Pneumonia. Infection. 2002;(6):378-386. doi:10.1007/s15010-002-2096-z
- Petitpretz P, Arvis P, Marel M. Oral Moxifloxacin vs High-Dosage Amoxicillin in the Treatment of Mild-to- Suspected Pneumococcal Pneumonia in Adults *. Chest. 2001;119(1):185-195. doi:10.1378/chest.119.1.185
- Mokabberi R, Haftbaradaran A, Ravakhah K. Doxycycline vs . levofloxacin in the treatment of community-acquired pneumonia. J Clin Pharm Ther. 2010;35:195-200. doi:10.1111/j.1365-2710.2009.01073.x
- Maimon N, Nopmaneejumruslers C, Marras TK. Antibacterial class is not obviously important in outpatient pneumonia: a meta-analysis. Eur Respirol J. 2008;31:1068-1076. doi:10.1183/09031936.00109007
- Uranga A, Espana P, Bilboa A, Quintana JM, Arriaga I. Duration of Antibiotic Treatment in Community-Acquired Pneumonia A Multicenter Randomized Clinical Trial. JAMA Intern Med. 2016;176(9):1257-1265. doi:10.1001/jamainternmed.2016.3633
- Tansarli GS, Mylonakis E. Systematic Review and Meta-analysis of the Efficacy of Short- Course Antibiotic Treatments for Community-Acquired Pneumonia in Adults. Antimicrob Agents Chemother. 2018:1-13.
- Moussaoui R, Borgie CAJM De, Broek P Van Den, et al. Effectiveness of discontinuing antibiotic treatment after three days versus eight days in mild to moderate-severe community acquired pneumonia: randomised, double blind study. BMJ.:1-7.
- Mandell LA, Wunderink RG, Anzueto A, et al. Infectious Diseases Society of America/American Thoracic Society Consensus Guidelines on the Management of Community-Acquired Pneumonia in Adults. Clin Infect Dis. 2007;44:S27-S72. doi:10.1086/511159
- Postma D, van Werkhoven C, van Elden L, Thijsen S, Hoepelman A. Antibiotic Treatment Strategies for Community-Acquired Pneumonia in Adults. N Engl J Med. 2015;372(14). doi:10.1056/NEJMoa1406330
- Garin N, Genné D, Carballo S, et al. β -Lactam Monotherapy vs β -Lactam–Macrolide Combination Treatment in Moderately Severe Community-Acquired Pneumonia A Randomized Noninferiority Trial. JAMA Intern Med. 2014;174(12):1894-1901. doi:10.1001/jamainternmed.2014.4887
- Lee J, Giesler D, Gellad W, Fine M. Antibiotic Therapy for Adults Hospitalized With Community-Acquired Pneumonia A Systematic Review. JAMA. 2016;315(6):593-602. doi:10.1001/jama.2016.0115
- Sligl WI, Asadi L, Eurich DT, Tjosvold L, Marrie TJ, Majumdar SR. Macrolides and Mortality in Critically Ill Patients With Community-Acquired Pneumonia: A Systematic Review and Meta-Analysis*. Crit Care Med. 2014;42(2):420-432. doi:10.1097/CCM.0b013e3182a66b9b
- Long B, Koyfman A. Best Clinical Practice: Blood Culture Utility in the Emergency Department. J Emerg Med. 2016;51(5):529-539. doi:10.1016/j.jemermed.2016.07.003
- Sharma L, Losier A, Tolbert T, Dela Cruz CS, Marion CR. Pneumonia Updates on Legionella, Chlamydophila, and Mycoplasma Pneumonia. Clin Chest Med. 2017;38(1):45-58. doi:10.1016/j.ccm.2016.11.011.Pneumonia



Thank you! Great summary of the salient features of approach to diagnosis and management of CAP.