Epidemiology of pulmonary embolism in pregnancy
- The rate of pulmonary embolism (PE) in pregnancy is 5.4 per 10 000 pregnancies.
- This is 5x the risk of the general non-pregnant female population
- The rates of venous thromboembolism change throughout pregnancy:
- Antepartum: 5.4 per 10 000
- Peripartum: 7.2 per 10 000
- Postpartum: 4.3 per 10 000
- This leads to 0.96 deaths per 100 000 pregnant patients, which accounts for 10% of maternal deaths
- PE is 1 of the 3 leading causes of direct maternal deaths in western countries, the others being postpartum hemorrhage and amniotic fluid embolism.
- In some developed countries, notably European countries, PE is the number one leading cause of maternal death.
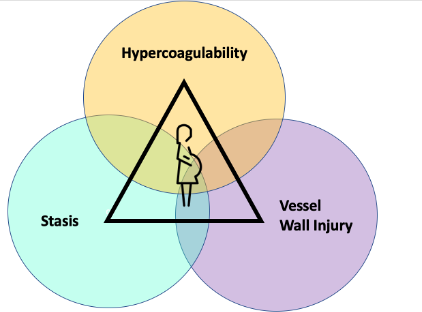
Pathophysiology and diagnostic challenges in pregnancy
- Using the principles of Virchow’s triad, pregnant patients have multiple risks factors for developing VTE:
- Increased venous stasis from venous dilatation and obstruction from the gravid uterus
- Increased procoagulant factors to protect from hemorrhage at the time of childbirth or miscarriage
- Vessel wall injury during labour and following caesarian section
- The symptoms of PE have a significant clinical overlap with normal changes seen in pregnancy.
- Tachycardia, shortness of breath, leg edema and GERD which are normal signs and symptoms in pregnancy mimic a DVT/PE.
- This overlap creates a diagnostic dilemma.
Clinical decision rules
- Guidelines regarding the use of clinical decision rules vary widely between societies with inconsistent recommendations.
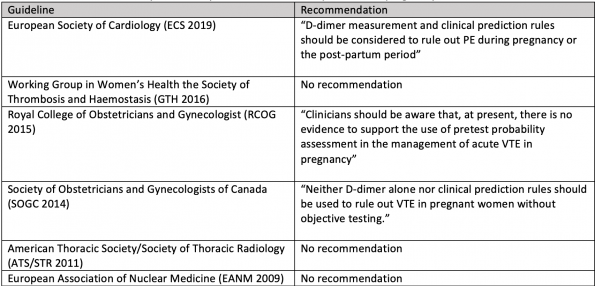 (Table adapted from Wan T et al, Thromb Res 2017)
(Table adapted from Wan T et al, Thromb Res 2017)
- Most societies either have no recommendation regarding the use of clinical decision rules or recommend against its use with the exception of the European Cardiology Society (ECS).
- The ECS have the most recently published guidelines (2019) supporting the use of clinical prediction rules and d-dimer based on 2 recent studies:
- CT-PE Pregnancy Group study (2018)
- Righini M, Robert-Ebadi H, Elias A, et al. Diagnosis of pulmonary embolism during pregnancy: a multicenter prospective management outcome study. Ann Intern Med 2018; 169: 766–773.
- Pregnancy-Adapted YEARS study (2019)
- Van der Pol LM, Tromeur C, Bistervels IM, et al. Pregnancy-adapted YEARS algorithm for diagnosis of suspected pulmonary embolism. N Engl J Med 2019; 380: 1139–1149.
- CT-PE Pregnancy Group study (2018)
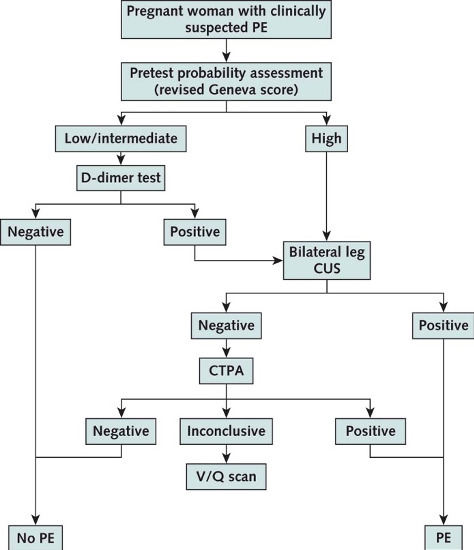
1. CT-PE Pregnancy Group Study (2018)
- A multicenter prospective management outcome study published in Annals of Internal Medicine in 2018. Their goal was to prospectively validate a new diagnostic algorithm in pregnant women with suspected PE. Their algorithm was compromised of a pretest clinical probability assessment (revised Geneva score), d-dimer testing, bilateral lower limb compression ultrasonography and CTPA. They included 395 pregnant patients in 11 hospitals in France and Switzerland who presented to the ED with suspected PE.
- The primary outcome was the rate of thromboembolic events during the 3-month follow-up period.
- Findings: Pulmonary embolism was diagnosed in 28 of 395 women. This represented a prevalence of 7.1%. CTPA was avoided in 11.7% of patients using the algorithm without any VTE in the 3-month follow-up period. When compression leg ultrasound was indicated, 1.8% of these were positive for DVT.
- Limitations:
- The Geneva score is not ideal to use in a pregnant population as some of the criteria are not always applicable (ie. age over 60).
- The yield from bilateral compression ultrasound was very low (1.8% had DVT detected by ultrasound).
- There was also a high rate of prophylactic anticoagulation in the follow-up period.
- Interpretation: Safely ruling-out PE in pregnant patients who are low/intermediate risk with a negative d-dimer can be done using this algorithm while also avoiding imaging in some patients (~10%).
2. Pregnancy Adapted YEARS Study (2019)
- A prospective multicenter, observational study in the Netherlands published in the New England Journal of Medicine in 2019. They assessed 510 pregnant women presenting to the ED or obstetrical ward for suspected PE.
- Their goal was to validate the pregnancy-adapted YEARS algorithm by assessing the incidence of VTE during a 3-month follow-up period and the rate of avoided imaging studies.
- Findings: PE was diagnosed in 4% of patients using this algorithm. The only VTE diagnosed in the 3-month follow-up period was a single DVT. CTPA was avoided in 32-65% of women depending on trimester.
- Limitations:
- May have been bias due to clinicians being aware of the d-dimer results prior to applying the algorithm.
- One of the YEARS criteria “PE the most likely diagnosis” is quite subjective and can independently alter the d-dimer threshold.
- Low incidence rate of pulmonary embolism suggests that they may have been too inclusive in terms of patients enrolled in the study.
- Interpretation: The pregnancy-adapted YEARS algorithm can be safely used to rule-out PE in pregnant patients by using adapted d-dimer thresholds while avoiding large numbers of imaging studies.
Of note the Pregnancy-Adapted YEARS algorithm has since been externally validated by applying the algorithm to the CT-PE Pregnancy Group population. However, when applied to this patient population CTPA was avoided in only 21% of patients (versus 39% in the original study). (Langlois E, et al. J Thromb Haemost. 2019)
Imaging studies
1. Leg ultrasound
- Compression ultrasound is the test of choice to diagnose DVT in pregnant women.
- The rationale for performing a leg ultrasound in a pregnant woman with suspected DVT is that if DVT is confirmed, this leads to the same therapy as PE, and consequently chest imaging can be avoided altogether. This has the benefit of avoiding unnecessary radiation.
- The yield of a lower limb ultrasound has been shown to be quite low in the absence of of DVT symptoms and potentially add unnecessary cost to the healthcare system.
- In a study looking at 158 pregnant women with suspected PE, 136 of those patients had no DVT symptoms and DVT was not detected in any of these patients. (Cooper et al. J Thorac imaging, 2012)
- Even in pregnant patients with suspected PE and the presence of DVT symptoms, the yield of leg ultrasound is lower than the non-pregnant population. There is a high incidence of isolated DVTs in the iliac veins in pregnancy, which are more difficult to detect than more distal thrombi by ultrasound.
- Although society guidelines have varying recommendations regarding the use of leg ultrasound in pregnant patients with suspected PE, it should only be performed in patients with DVT symptoms.
2. CTPA versus V/Q scan
- There has been a substantial increase in the incidence rate of VTE over the last decade.
- This is largely due to the increased use of advanced imaging studies. In the last 17 years, the use of CTPA has steadily increased.
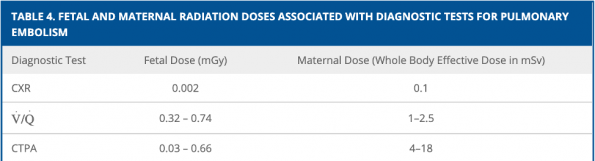
- The National Council of Radiation Protection and Measurements states that CTPA exposes the fetus to 50mGy or less which is considered a negligible risk compared to other adverse events of pregnancy. Using this threshold, chest radiography, V/Q scans and CTPA are all safely below this threshold. Although V/Q scans has slightly lower radiation to the fetus than CTPA, this difference is negligible and should not influence your imaging choice.
- There is however a significant difference in radiation to maternal breast tissue. CTPA can lead to more maternal radiation by almost 7-fold when compared to V/Q scans. Both imaging modalities have similar rates of inconclusive studies in pregnant patients.
- A single CT with this dose of radiation would increase a woman’s lifetime risk of cancer by 1.5%.
3. Chest radiography
- A reasonable initial investigation in any pregnant patient with shortness of breath and chest pain
- The fetal radiation dose associated with chest radiography is extremely low (0.002 mGy), less than 100 fold of the toxic fetal dose and therefore should not deter you from performing a chest X-ray.
- Chest radiographs should be performed in all pregnant who require advanced chest imaging.
- In patients with abnormal chest radiographs, studies have shown a rate of inconclusive V/Q scans up to 40%, in comparison to an inconclusive rate of 6% in patients with normal chest X-rays. (Cahill et al. Obstet Gynecol, 2009).
- Utilize chest radiographs to triage patients to the test that is most likely to give a useful diagnostic answer. If the chest radiograph is normal, perform a V/Q scan. If the chest X-ray is abnormal, strongly consider doing a CTPA.
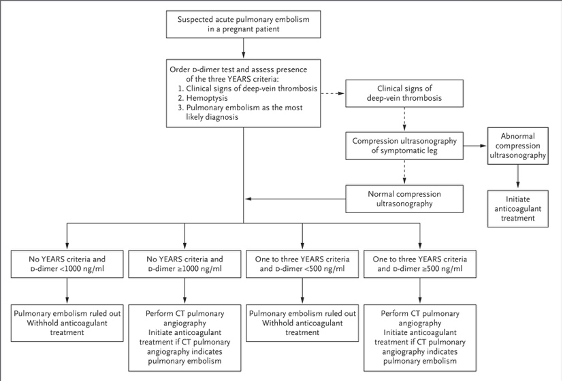
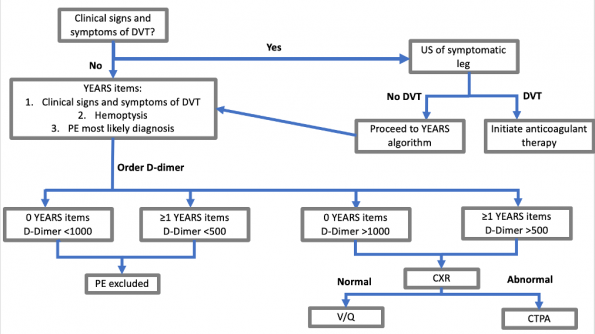
Diagnostic algorithm for PE in pregnancy
Based on the evidence reviewed here, consider using the following algorithm on the pregnancy-adapted YEARS study to work-up pregnant patients with suspected pulmonary embolism:
Take home points
- Perform leg ultrasounds in patients with DVT symptoms.
- If PE cannot be ruled-out using the pregnancy-adapted YEARS algorithm, chest radiography should be used to triage patients to the appropriate invasive chest imaging study.
- In patients with normal chest radiographs, V/Q scans should be preferentially ordered. Otherwise, patients with abnormal chest X-rays should have a CTPA study done to optimize the likelihood of obtaining a conclusive study.
References
- A. Cutts, H.A. Tran, E. Merriman, Nandurkar, G. Soo, D. DasGupta, N. Prassannan, B.J. Hunt. The utility of the wells clinical prediction model and ventilation-perfusion scanning for pulmonary embolism diagnosis in pregnancy. Blood Coagul. Fibrinolysis, 25 (4) (Jun 2014), pp. 375-378
- O’Connor, J. Moriarty, J. Walsh, J. Murray, S. Coulter-Smith, W. Boyd. The application of a clinical risk stratification score may reduce unnecessary investigations for pulmonary embolism in pregnancy. J. Matern. Fetal Neonatal Med., 24 (12) (Dec 2011), pp. 1461-1464.
- Cahill AG, Stout MJ, Macones GA, Bhalla S. Diagnosing pulmonary embolism in pregnancy using computed tomographic angiography or ventilation-perfusion. Obstet gynecol 2009;114:124-129.
- Chan WS, Lee A, Spencer FA, Chunilal S, Crowther M, Wu W, Johnston M, Rodger M, Ginsberg JS. D-dimer testing in pregnant patients: towards determining the next ‘level’ in the diagnosis of deep vein thrombosis. J Thromb Haemost2010;8:1004–1011.
- Chan WS, Rey E, Kent NE, et al. Society of Obstetricians and Gynaecologists of Canada (SOGC) Clinical Practice Guidelines: Venous thromboembolism and antithrombotic therapy in pregnancy. J Obstet Gynaecol Can. 2014; 36 (6): 527–553.
- Cooper M, Matthews S. Imaging pulmonary embolism in pregnancy: what is the value of routine bilateral leg doppler ultrasound in women without symptoms of deep venous thrombosis?. Thorac. Imaging.2012; 27: W155
- Heit JA. Epidemiology of venous thromboembolism. Nat Rev Cadiol. 2015; 12(8): 464-74.
- Hunt BJ, Parmar K, Horspool K, et al. The DiPEP (Diagnosis of PE in Pregnancy) biomarker study: An observational cohort study augmented with additional cases to determine the diagnostic utility of biomarkers for suspected venous thromboembolism during pregnancy and puerperium. Br J Haematol. 2018;180(5):694-704.
- Kline JA, Kabrhel C. Emergency Evaluation for Pulmonary Embolism, Part 2: Diagnostic Approach. J Emerg Med. 2015;49(1):104-17.
- Kline JA, Williams GW, Hernandez-Nino J. D-dimer concentrations in normal pregnancy: new diagnostic thresholds are needed. Clin Chem. 2005;51:825–829.
- Langlois E, et al. Could the YEARS algorithm be used to exclude pulmonary embolism during pregnancy? Data from the CT-PE-pregnancy study. J Thromb Haemost. 2019 Aug; 17 (8): 1329-1334.
- Leung AN, Bull TM, Jaeschke R, et al. American Thoracic Society documents: an official American Thoracic Society/Society of Thoracic Radiology Clinical Practice Guideline–Evaluation of Suspected Pulmonary Embolism in Pregnancy. Radiology. 2012;262(2):635-46.
- Leung AN, Bull TM, Jaeschke R, Lockwood CJ, Boiselle PM, Hurwitx LM, et al. An official American Thoracic Society/Society of Thoracic Radiology clinical practice guideline: evaluation of suspected pulmonary embolism in pregnancy. AM J Respir Crit Care Med 2011; 184: 1200-8.
- M Rossignol, E Moreau, M Dreyfus. Maternal death by venous thromboembolic disease. Gynecol Obstet Fertil Senol. 217 Dec;45 (12S): S31-S37.
- Okonofua F. The DiPEP (diagnosis of pulmonary embolism in pregnany) study and the limited accuracy of clinical decision ruls and d-dimer: what next?. BJOG. 2019 Feb; 126 (3): 393.
- Righini M, Robert-Ebadi H, Elias A, et al. Diagnosis of pulmonary embolism during pregnancy: a multicenter prospective management outcome study. Ann Intern Med 2018; 169: 766–773.
- Righini M, LeGal G, Aujesky D, Roy PM, Sanchez O, Verschuren F, Rutschmann O, Nonent M, Cornuz J, Thys F, et al. Diagnosis of pulmonary embolism by multidetector CT alone or combined with venous ultrasonography of the leg: a randomised non-inferiority trial. Lancet2008;371:1343–1352.
- Royal College of Obstetricians & Gynaecologists. Thromboembolic disease in pregnancy and the puerperium: acute management. Green-top Guideline (April 2015).No. 37b
- Liu, J. Rouleau, K.S. Joseph, R. Sauve, R. Liston, D. Young. Epidemiology of pregnancy associated venous thromboembolism: a population-based study in Canada. J. Obstet. Gynaecol. Can., 31 (2009), pp. 611-620
- Society of Obstetricians and Gynecologist of Canada Guideline: Venous thromboembolism and antithrombotic therapy in pregnancy. J. Obstet. Gynaecol. Can., 36 (6) (Jun 2014), pp. 527-553
- Van der Hulle T et al. Simplified diagnostic management of suspected pulmonary embolism (the YEARS study): a prospective, multicentre, cohort study. 2017; 390:289-297.
- van der Pol LM, Tromeur C, Bistervels IM, et al. Pregnancy-adapted YEARS algorithm for diagnosis of suspected pulmonary embolism. N Engl J Med 2019; 380: 1139–1149.
- Wan T, Skeith L, Karovitch A, Rodger M, Le Gal G. Guidance for the diagnosis of pulmonary embolism during pregnancy: consensus and controversies. Thromb Res. 2017;157:23-28.




Trackbacks/Pingbacks