Status epilepticus (SE) is considered one of the most common neurological emergencies with an estimated incidence of 20 cases/100,000 people worldwide.
Just the facts
- The distribution is bimodal, peaking in patients <1 and >60 years old.
- In children, the cause is predominantly from febrile illnesses, with or without infection, and in adults it is largely secondary to acute damage from strokes or anoxic brain injuries.
- The mortality rate is extremely high (30% in adults and 3% in children).
- Prognosis is largely affected by:
- Etiology
- Duration of seizure
- Older age
Pathophysiology
- Seizures occur when neurons in the brain synchronously fire when they are not supposed to, a process driven by either an excess of excitatory signals or by a lack of inhibitory signals.
- Glutamine binds NMDA receptors more than GABA binds to GABA receptors in the synapse
- Neurons firing in this manner can manifest as either motor symptoms (tonic, clonic or myoclonic movements) or non-motor symptoms (behaviour arrest or emotional outbursts).
- When the spread of signals reaches the reticular activating system (RAS) in the brain, there is loss of consciousness which characterizes a generalized seizure.
Classification
The old classification
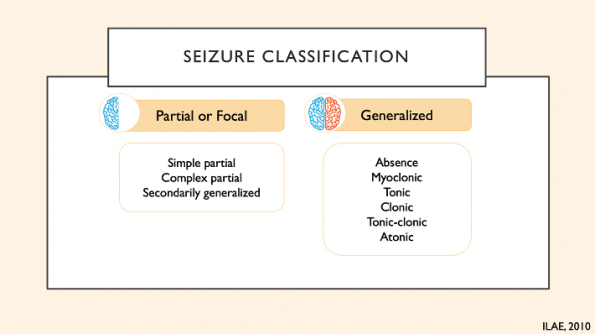
Prior to 2017, types of seizure were divided into 2 major categories:
- Partial of focal onset – one hemisphere is affected
- Generalized onset – both hemispheres are affected with involved loss of consciousness
Focal onset seizures were subdivided into:
- Simple partial seizures – without loss of awareness
- Complex partial seizures – with loss of awareness
Where awareness was defined as knowledge and understanding or surrounding events and nay not necessarily by synonymous with consciousness, although at times used interchangeably.
Generalized onset seizures included the following categories:
- Absence seizures
- Myoclonic seizures
- Tonic-clonic seizures
- Tonic seizures
- Clonic seizures
- Atonic seizures
The new classification
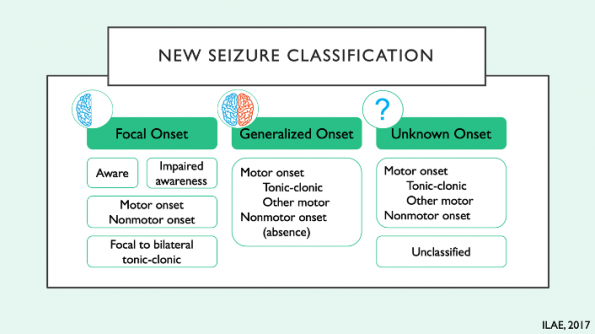
In 2017, the international league against epilepsy (ILAE) published the most recent seizure classification.
There are 3 categories:
- Focal seizures – with or without impaired awareness, having motor or non-motor features. Seizures that begin with a focus and progress to generalized are now categorized as focal to bilateral tonic-clonic seizures.
- Generalized seizures – motor onset (eg. tonic-clonic) or non-motor onset (absent)
- Unknown onset seizures – third category reserved for seizures that have unknown onset, with motor or non-motor features.
Defining status epilepticus

The duration of 5 minutes was chosen arbitrarily, selected primarily based on animal studies that showed prolonged seizures led to irreversible neuronal damage and therefore urgency is required in status epilepticus treatment.
Now, it’s all about time
In 2015, the definition of status epilepticus (SE) was modified. The ILAE introduced the idea of times T1 and T2 to the definition
- Time T1 is the time point at which the seizures becomes “abnormally prolonged”.
- Time T2 is the time point beyond which there is high risk for long term sequelae
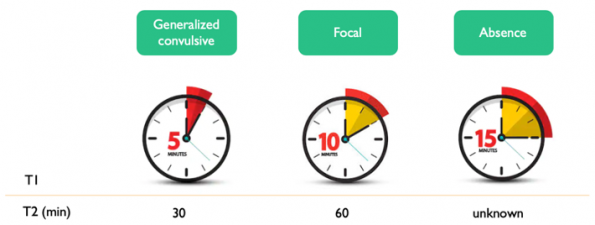
According to this method of defining status epilepticus, the new definitions add the following:
- Generalized convulsive status epilepticus (GCSE) = seizures lasting >5 minutes. Permanent neuronal damage expected at 30 minutes.
- Focal SE = seizures lasting>10 minutes. Permanent neuronal damage expected at 60 minutes.
- Absence SE = seizures lasting >15 minutes. There is no recommendation to when permanent neuronal damage is expected for this type of seizure.
GCSE has the poorest outcomes while focal and absence SE are not as life threatening.
Refractory SE
Defined as SE that is refractory to both 1st and 2nd line anti-epileptics. This is not to be confused with benzodiazepine-refractory SE, which as stated is only refractory to benzodiazepines.
Super-refractory SE
Defined as SE that is refractory to 1st, 2nd and 3rd line agents for 24 hours.
Treating status epilepticus
The most recent evidence-based guidelines were published by the Neurocritical Care Society in 2012 and by the American Epilepsy Society (AES) in 2016.
In brief, as adapted from the AES guideline, there are 4 phases:
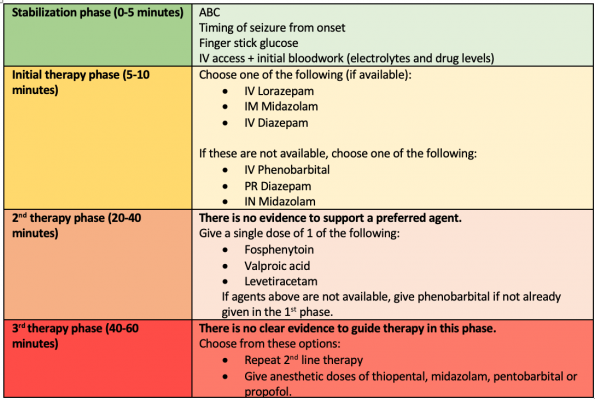
An important disclaimer from the AES guidelines
“Depending on the etiology or severity of the seizures, patients may go through the phases faster or even skip the second phase and move rapidly to the third phase, especially in sick or ICU patients”.
There is evidence to suggest that this statement is more often true than not which explains why there is great variation in how physicians manage SE.
Benzodiazepines
A randomized trial by Alldredge et al (2001), provided evidence to support that giving benzodiazepines is important. Results showed termination of 60% of seizures with the administration of lorazepam or diazepam compared to giving placebo alone.
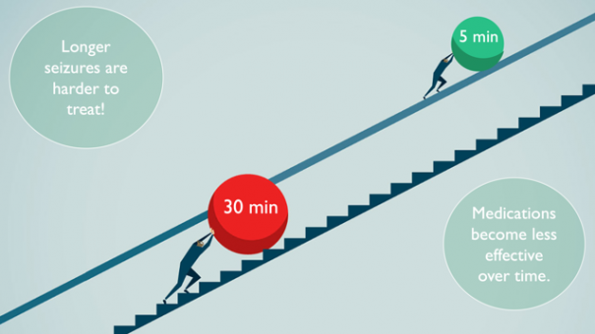
The longer patients are left to seize, the more refractory seizures become – therefore harder to treat. In was found in multiple studies that GABA receptors were quickly internalized after an initial massive surge of GABA at the start of the seizure. This can occur as quickly as < 5 minutes of seizure duration. For this reason, many of the available antiepileptic drugs become less effective with time.
Delayed benzodiazepines = longer seizures and poorer outcomes
A multicenter observation prospective study of 218 patients (Gainza-Lein 2018) demonstrated that delayed benzodiazepine administration by > 10 minutes from seizure onset was associated with more deaths (66 vs 34%, with an adjusted odds ratio of 11), a higher need for continuous infusion of IV anesthetics and a longer convulsion duration (49 minutes longer in the delayed group)4.
This study was limited by the biases of an observational study design. However, it was the first study to identify an association between delayed benzodiazepine administration and poor outcomes including death. This paper may change our view SE as an extremely time-sensitive emergency similar to strokes and cardiovascular events.
Which benzodiazepine is preferred?
The RAMPART trial (2012), a randomized, noninferiority trial of 893 children and adults demonstrated that IM midazolam is not inferior to IV lorazepam. In cases where an IV line cannot be easily established, IM midazolam is the medication of choice. When access is available, IV lorazepam is preferred instead. Midazolam was easier and faster to administer (1.2 mins vs 4.8 mins to give lorazepam), which translated to a shorter time to seizure termination. However, if an IV was present, giving IV lorazepam led to earlier seizure termination (1.6 vs 3.3 mins). Adverse event rates, including endotracheal intubation, hospitalization, recurrent seizures, and hypotension were not statistically different between groups5.
There is no difference in efficacy between IV lorazepam and IV diazepam6,7 and there are no direct comparisons between IV midazolam and IV lorazepam8.
Dosing of benzodiazepines
Benzodiazepines tend to be dosed below guideline recommendations.
-
- Patterns of benzodiazepine dosing by paramedics and ED physicians were explored in a brief analysis of the pre-enrollment population of the ESETT trial9.
- The analysis consisted of 207 patients with benzodiazepine-refractory status epilepticus and included 511 administrations of benzodiazepines. Overall, <30% met minimum dose recommendation per current guidelines10.
- Since the ESETT trial enrolled status epilepticus patients, it was likely selecting for patients who had a higher likelihood of having factors associated with prolonged seizures such as benzodiazepine underdosing. However, accepting this limitation, this article still demonstrates that there is room for improvement in how we dose benzodiazepine for treatment of SE.
Benzodiazepine doses per current guidelines:
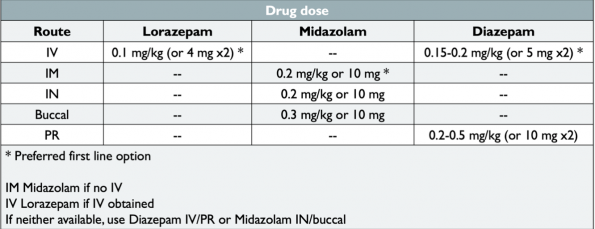
Second-line antiepileptics
The emerging role of Levetiracetam
ConSEPT and EcLiPSE were two large RCTs designed to prove that levetiracetam is better than phenytoin.
- Both involved >200 pediatric patients with refractory convulsive SE.
- The primary outcome in ConSEPT was clinical termination of seizure 5 minutes after infusion of the study drug11.
- EcLiPSE instead looked at how long it took, from time of randomization, for the drug in question to stop all signs of convulsion12.
- In the end, both studies were negative. Levetiracetam was non-superior to phenytoin.
Second-line anti epileptics as equals
A large multicenter RCT called the ESETT trial further confirmed that second line antiepileptics are not different in their efficacy and safety9.
- ESETT compared fosphenytoin, valproic acid and levetiracetam, for treatment of benzodiazepine-refractory GCSE in 384 children and adults.
- They found that termination of seizure occurred in approximately 50% of patients in all treatment arms.
- There were no differences in efficacy between age groups in the secondary analysis and there were no significant differences in adverse outcomes13.
Okay great, which second-line agent should be used?
The decision should be individualized and patient-specific. A few drug complications can be factored in to help avoid using them in a way that will worsen the patient’s condition.

Neurolytic intubation
Early intubation and sedation is an attractive option to terminate seizures in a timely manner. Unfortunately, the evidence is still sparse. The literature on using IV anesthetic agents to treat seizure stems from observational studies looking at managing refractory status in the ICU. Current guidelines recommend using either propofol, midazolam or phenobarbital, often causing a therapeutic coma in order to take control of persistent seizures evidenced on EEG14,15.
Many physicians will reach for propofol as the agent of choice because of its ultra-fast onset and high lipid solubility. However, propofol needs to be used with caution due to its propensity to cause hypotension and respiratory depression in addition to propofol infusion syndrome. For this reason, guidelines advise that midazolam can be an alternative. Midazolam may cause less hypotension because it is not diluted in propylene glycol and a lower dose of 0.2 mg/kg IV is often given to avoid causing significant hypotension15.
Phenobarbital is rarely used due to possible serious adverse effects but some evidence does suggest barbiturates are associated with lower frequency of treatment failure and seizure recurrence14.
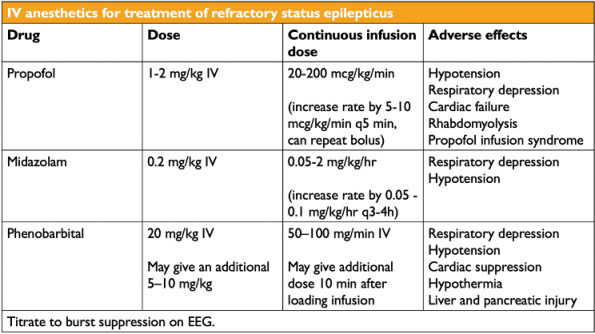
Ketamine
Propofol and midazolam work as strong GABA agonists but, as seizures become increasingly more refractory, the number of GABA receptors will decrease while NMDA receptors will increase. Thus, adding a strong NMDA antagonist like ketamine should theoretically help.
Similar to other IV anesthetics, there is limited data looking at ketamine for the treatment of SE. Several case reports and case series demonstrated the efficacy for ketamine at 57% overall when administered as an add-on agent to other conventional IV anesthetics, which themselves have an efficacy anywhere between 42-66%14,16. This medication has a relatively favourable side effect profile. Specifically, it is not associated with cardiac depression and hypotension like other IV anesthetics. The recommended dose is 1-2 mg/kg bolus followed by 1-10 mg/kg/hour infusion16.
So far, it appears the best predictor of efficacy for ketamine is the time of administration. The systematic review by Rosati et al (2018) found that ketamine was 2 times more effective if given early (on day 3 vs day 27) for refractory and super-refractory SE17. In another study, a retrospective review of 60 refractory status episodes, response rate was highest at 60% when ketamine was given as a 3rd or 4th line agent in combination with other IV anesthetics. Given later than that, ketamine became increasingly less effective14.
Take home points
Instead of a stepwise, watch-and-wait approach, perhaps multiple management steps should happen in parallel. The proposed algorithm here is simpler, one that prioritizes early seizure termination. In the first 5 minutes, the focus should be on the ABCs and testing for reversible causes. The first dose of benzodiazepine should be given within this time frame. If the patient is still seizing, a second dose of benzodiazepine and a second-line antiepileptic drug should be available to be given immediately between 5 and 10 minutes. At the same time, the patient should be prepared for intubation. At 10 to 20 minutes, if the seizure persists, the patient needs to be rapidly intubated using IV anesthetic for airway protection and seizure control.

References
- Alldredge BK, Gelb AM, Isaacs SM, et al. A comparison of lorazepam, diazepam, and placebo for the treatment of out-of-hospital status epilepticus [published correction appears in N Engl J Med 2001 Dec 20;345(25):1860]. N Engl J Med. 2001;345(9):631-637.
- Naylor DE, Wasterlain CG. GABA synapses and the rapid loss of inhibition to dentate gyrus granule cells after brief perforant-path stimulation. Epilepsia. 2005;46 Suppl 5:142-147.
- Niquet J, Baldwin R, Suchomelova L, et al. Benzodiazepine-refractory status epilepticus: pathophysiology and principles of treatment. Ann N Y Acad Sci. 2016;1378(1):166-173.
- Gaínza-Lein M, Sánchez Fernández I, Jackson M, et al. Association of Time to Treatment With Short-term Outcomes for Pediatric Patients With Refractory Convulsive Status Epilepticus. JAMA Neurol. 2018;75(4):410-418.
- Silbergleit R, Durkalski V, Lowenstein D, et al. Intramuscular versus intravenous therapy for prehospital status epilepticus. N Engl J Med. 2012;366(7):591-600.
- Leppik IE, Derivan AT, Homan RW, Walker J, Ramsay RE, Patrick B. Double-blind study of lorazepam and diazepam in status epilepticus. JAMA. 1983;249(11):1452-1454.
- Chamberlain JM, Okada P, Holsti M, et al. Lorazepam vs diazepam for pediatric status epilepticus: a randomized clinical trial. JAMA. 2014;311(16):1652-1660.
- Glauser T, Shinnar S, Gloss D, et al. Evidence-Based Guideline: Treatment of Convulsive Status Epilepticus in Children and Adults: Report of the Guideline Committee of the American Epilepsy Society. Epilepsy Curr. 2016;16(1):48-61.
- Kapur J, Elm J, Chamberlain JM, et al. Randomized Trial of Three Anticonvulsant Medications for Status Epilepticus. N Engl J Med. 2019;381(22):2103-2113.
- Sathe AG, Tillman H, Coles LD, et al. Underdosing of Benzodiazepines in Patients With Status Epilepticus Enrolled in Established Status Epilepticus Treatment Trial. Acad Emerg Med. 2019;26(8):940-943.
- Dalziel SR, Borland ML, Furyk J, et al. Levetiracetam versus phenytoin for second-line treatment of convulsive status epilepticus in children (ConSEPT): an open-label, multicentre, randomised controlled trial. Lancet. 2019;393(10186):2135-2145.
- Lyttle MD, Rainford NEA, Gamble C, et al. Levetiracetam versus phenytoin for second-line treatment of paediatric convulsive status epilepticus (EcLiPSE): a multicentre, open-label, randomised trial. Lancet. 2019;393(10186):2125-2134.
- Chamberlain JM, Kapur J, Shinnar S, et al. Efficacy of levetiracetam, fosphenytoin, and valproate for established status epilepticus by age group (ESETT): a double-blind, responsive-adaptive, randomised controlled trial. Lancet. 2020;395(10231):1217-1224.
- Reznik ME, Berger K, Claassen J. Comparison of Intravenous Anesthetic Agents for the Treatment of Refractory Status Epilepticus. J Clin Med. 2016;5(5):54. Published 2016 May 19.
- Brophy GM, Bell R, Claassen J, et al. Guidelines for the evaluation and management of status epilepticus. Neurocrit Care. 2012;17(1):3-23.
- Gaspard N, Foreman B, Judd LM, et al. Intravenous ketamine for the treatment of refractory status epilepticus: a retrospective multicenter study. Epilepsia. 2013;54(8):1498-1503.
- Rosati A, De Masi S, Guerrini R. Ketamine for Refractory Status Epilepticus: A Systematic Review. CNS Drugs. 2018;32(11):997-1009




Extremely helpful! I’ll be using your information for teaching my students going forward.