In Ontario, Canada, alcohol-related ED visits have increased 4.4x faster than the overall increase in visits from 2003-2016, and was responsible for more admissions than heart attacks in 2016.
In part 1 of this 2-part series, we will discuss tips for the diagnosis and initial management of alcohol withdrawal. In part 2, we will dive deep into the role of benzodiazepine tapers, how to use gabapentin, naltrexone, and communication strategies to optimize discharge, and help your patient meet their goals.
For quick reference, here is an algorithm for providers, and a handout for patients in Ottawa.
1. PAWSS score
The risk of severe withdrawal can be predicted using the PAWSS score, and previous withdrawal severity. Understanding this risk will help you choose an appropriate strategy to prevent or treat it.
The JAMA Rational Clinical Exam series endorses the PAWSS score to predict “will this hospitalized patient develop severe alcohol withdrawal syndrome?”1
PAWSS score ≥ 4 is predictive of seizure, delirium, or a CIWA ≥ 15 in all patients being admitted to hospital with any alcohol intake in the last 30 days. It had a sensitivity of 93% and specificity of 99%.

Thus far, it has only been validated in patients being admitted, and if trying to specifically predict seizure or delirium, it will be over-inclusive. The best predictors of seizure and delirium are previous withdrawal seizures (Odds Ratio [OR] 2.8) and previous delirium (OR 2.6).2
2. Alcohol withdrawal is diagnosed by the DSM5 criteria not the CIWA-Ar score
The DSM5 criteria are currently the gold standard for withdrawal. The CIWA-Ar score should not be used for diagnosis – the syndrome it describes is non-specific. Inappropriate application of the CIWA-Ar score to a patient not in withdrawal may lead to adverse events including over sedation and misdiagnosis.3
To diagnose withdrawal, the alcohol intake history should also fit – withdrawal is rare if less than 6 drinks a day, unless elderly.4 In one case series, the physical signs of withdrawal were the most prevalent symptoms: tremor (92.6%), sweating (87.1%), tachycardia (79.7%), agitation (79.3%), and hypertension (77.9%).5 Remember, that these can be masked by beta-blockers.
3.SHOT and OAWS scores
The CIWA-Ar score is the most well-validated tool to track withdrawal symptoms, but it has limitations. The SHOT and OAWS scores are reasonable adjuncts.
CIWA-Ar was validated with good inter-rater reliability in 1989.6 However, it was designed for alcohol treatment centers, where super-specialized nurses and physicians could monitor patients for longer periods of time. Variability likely improves with practice.
With the usual endpoint of treatment being CIWA ≤10 for 2 hours, the subjectivity of the CIWA-Ar score can easily make or break the difference between discharge from the ED or ongoing benzodiazepines. For instance, mild to moderate anxiety could be scored anywhere from a 1 to a 6. Also, it cannot be used when the patient is unable to communicate (i.e. delirium, post-ictal, language barrier).
For cases in which most of the CIWA-Ar points are from subjective symptoms or in which the decision to discharge is borderline, use a second score as an adjunct:
- The Sweating Hallucinations Orientation and Tremor (SHOT) score is a simple 4 item score which focuses on the objective signs of withdrawal.7
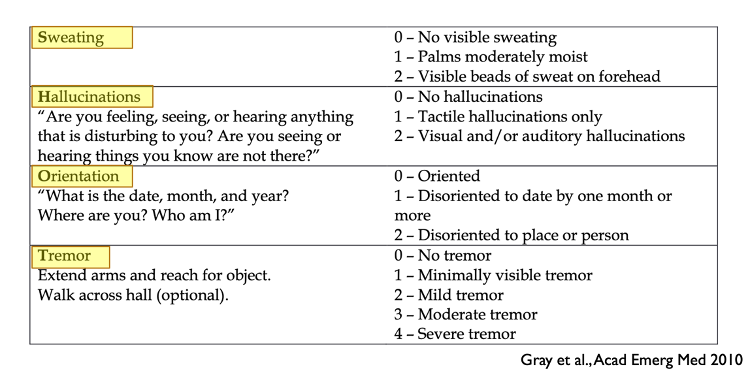
- The Objective Alcohol Withdrawal Score (OAWS) was designed for patients who were unable to communicate due to a language barrier and uses blood pressure, heart rate, tremor, diaphoresis, and agitation.8
4. Treatment
Long-acting benzodiazepines (i.e. diazepam, chloridazepoxide) are first line to treat withdrawal because they prevent severe withdrawal and seizures.
Benzodiazepines prevent seizure (RR 0.16, 95% CI 0.04-0.69) as evidenced in a Cochrane Review.9
Diazepam is ideal because it peaks in ~1h, allowing for frequent re-dosing while monitoring for sedation, and because of the half-life of the drug (50h) and its metabolite (100h). As 90% of seizures occur between 6-48h from the last drink, the half-life of diazepam is well suited for seizure prophylaxis.10
Lorazepam is first line if the patient has advanced cirrhosis/acute hepatitis, elderly, or on respiratory depressants because of its shorter half-life (12-18h).
5. In treating withdrawal, your choice of benzodiazepine strategy should depend on risk of progression to severe withdrawal. Greater risk warrants a more aggressive strategy. Use the PAWSS score, clinical impression, and previous history of seizure or delirium to guide your risk assessment.
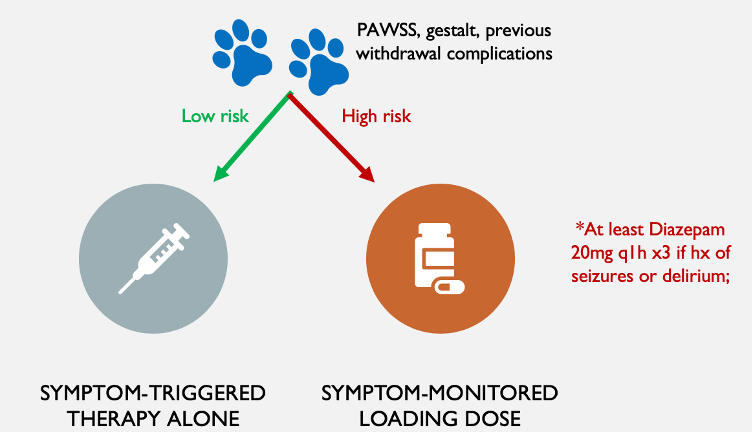
RCTs have proven that CIWA-Ar guided therapy is superior to a fixed-dose taper in terms of safely decreasing duration and dose of therapy.9,11 In an appropriate setting, diazepam can be safely dosed every 1 hour to an endpoint of CIWA ≤10 for 2 consecutive hours. In recent meta-analyses, CIWA-guided therapy resulted in a lower CIWA score at 48h and there was a trend towards lower rates of delirium.9,12 Some of these trials, however, only included low-risk patients without a history of seizure.
In someone at high risk for seizure or delirium, consider a symptom-monitored loading dose of diazepam first, before continuing with usual CIWA-guided therapy. A minimum loading dose of Diazepam 20mg q1h x3 is supported by the METAPHI guidelines to prevent seizures.4 Early case series and RCTs specifically targeting higher-risk patients found no seizures after diazepam loading.13,14
Key points:
- In the recently intoxicated patient, the PAWSS score can predict severe alcohol withdrawal. Clinical impression and previous seizure/delirium should also be used in predicting seizure and delirium risk.
- Diagnose alcohol withdrawal using the DSM5 criteria. Physical signs are the most common, and the alcohol intake history should fit.
- Use the CIWA-Ar score, but recognize that its limitations (subjectivity and the need to be able to communicate). Using the SHOT score and vital signs may help prevent discharging a patient too early when they still have significant tremor or signs of withdrawal that put them at risk for decompensation.
- In the patient at high risk for severe alcohol withdrawal, use a loading dose in the emergency department as seizure prophylaxis before continuing with usual CIWA symptom-guided therapy
References



