In this post, we’ll discuss viscoelastic hemostatic assays (VHAs), namely ROTEM (Rotational Thromboelastometry), and how we can use this tool to enhance the care of traumatically injured patients. This is the first in a two-part series. This part reviews the Evidence for VHAs in Trauma, while Part 2, found here, reviews the practical aspects of using ROTEM in trauma.
Hemorrhage remains the leading cause of preventable death from trauma. What if I told you there was a test that reduced transfusion requirement through goal-directed care, is already used at multiple Canadian, US, and European trauma centers, and has possible mortality benefit in bleeding patients?

ROTEM (rotational thromboelastometry) provides goal-directed transfusions for individual patients during damage control and hemostatic resuscitation, adding timely laboratory data to our clinical impression. In Canada, more and more centres are adopting ROTEM for the resuscitation of critically ill bleeding patients. In the US, TEG (thromboelastography) and ROTEM have been used for many years to guide massive transfusion. In Europe, it is standard of care to use VHAs (viscoelastic hemostatic assays; i.e. ROTEM or TEG) to guide trauma resuscitations.
For the purpose of this post we will focus on ROTEM as this is predominantly used in Canada. Information on TEG can be found from multiple other FOAM sources (found below).
This post will frame the discussion around ROTEM by answering 4 questions:
- What do we do right now?
- What tests are available now, how good are they, and the idea behind ROTEM
- Why should we use ROTEM?
- We’ll cover the many advantages of ROTEM-guided resuscitation
- How do I use ROTEM to guide management?
- How to read a ROTEM output and how it guides transfusion
- Why don’t we already have ROTEM?
- Important but solvable barriers to implementation
Before we get into the meat of the post: if this test is so great and everybody’s using it already… what do the guidelines say?
TL/DR; Use it if you have it.
- The European guideline on the management of major bleeding and coagulopathy following trauma. 2019
- Recommend coagulation monitoring with traditional lab tests or VHAs (Grade 1C).
- Resuscitation measures should be continued using a goal-directed strategy, guided by standard laboratory coagulation values and/or VHAs. (Grade 1B)
- British Society for Haematology. Guideline for VHAs in the management of major bleeding. 2018
- Normal VHA results confer a high NPV for transfusion need, enabling close monitoring without immediate MTP activation of the major haemorrhage protocol. Grade 2B.
- VHAs, particularly TEG, may reduce mortality and reduce transfusion exposure and, if available, may be considered for transfusion guidance in trauma haemorrhage. Grade 2B.
- European Society of Anesthesiology guideline for Management of severe perioperative bleeding. 2017
- Recommend algorithms incorporating triggers and targets based on VHA coagulation monitoring to guide individualized hemostatic intervention in the case of perioperative bleeding. (Grade 1C)
- Practice Guidelines for perioperative blood management. ASA task force on perioperative blood management. 2015
- If coagulopathy is suspected, obtain standard coagulation tests (g., INR, aPTT, fibrinogen concentration) or viscoelastic assays (e.g., thromboelastography [TEG] and ROTEM), if available, as well as platelet count
- Both the consultants and ASA members agree that if coagulopathy is suspected, obtain viscoelastic assays (g., TEG and ROTEM), when available, as well as platelet count
- Massive transfusion policies at trauma centers participating in the ACS TQIP. 2015
- (Massive transfusion in Trauma Guidelines. ACS TQIP. 2014.)
- Goal-directed therapy based on: Standard laboratory coagulation values, and/or VHAs, if available. Once laboratory data are available, resuscitation should be goal directed based on the laboratory findings and clinical evidence of ongoing bleeding
- VHAs as the standard for transfusion or injection of blood plasma, cryoprecipitate, platelet concentrate, or anti-fibrinolytic agents in the treatment strategy for traumatic coagulopathy and hemorrhagic shock
Background: What do we do right now?
The Clotting Cascade
A brief review of the clotting cascade (…I know, I’m sorry): the goal and end result is a platelet plug reinforced by cross-linked fibrin chains.
- Enzymatic phase: requires coagulation factors
- Platelet phase: thrombin generation on activated platelets generate fibrin from fibrinogen which crosslinks building the clot
- Fibrinolytic phase: clot dissolution is dependent on fibrin and tPA from endothelial cells

The clotting cascade can be “measured” by standard lab tests (SLTs):
- Local tissue effects – no clinically usable way to assess
- Intrinsic (F8, 9, 11, 12) – PTT
- Extrinsic (TF, F7) – PT/INR
- Common pathway (FXa, thrombin, fibrinogen) – TT, fibrinogen
- Platelet portion: CBC (platelet count)
- Fibrinolytic phase: d-dimer
The issues with SLTs are:
- Originally designed for monitoring therapeutic anticoagulation, not expeditious identification of trauma-related coagulation defects
- Not ideal to predict risk of bleed or clotting ability as these tests are run with plasma alone, not accounting for cellular clotting components (i.e. lack of in vivo applicability)
- And they have: longer turnover time, lack of specificity, and lack of ability to guide transfusion of blood products acutely
Acute Trauma Coagulopathy
- Hemorrhagic shock secondary to trauma is associated with intrinsic coagulopathy beyond the lethal triad, termed acute trauma coagulopathy (ATC) or trauma-induced coagulopathy (TIC)
- In severely injured patients, 25-35% already have a trauma-induced coagulopathy at presentation to ED
- This coagulopathy poses 8x higher risk of death from trauma
- ATC occurs early after injury, independent of coagulopathy from acidosis, hypothermia, or hemodilution and factor dilution
- Hypoperfusion is a large determinant of coagulopathy
- Hyperfibrinolysis: Injured endothelium releases tPA leading to early breakdown of necessary clot, therefore, increased need for blood products
- Fibrinolytic shutdown: not all trauma patients are coagulopathic, some can be hypercoagulable, reducing transfusion requirements
- Trauma patients are at high risk of hemorrhage-related morbidity and mortality, and ATC further increases the risk of complications, need for transfusion, and length of stay
Balanced 1:1:1 transfusion strategies have been found to improve mortality in this population but given the unique nature of each patient’s coagulopathy, is this one-size-fits-all approach appropriate for everyone? Is Balanced transfusion enough?
What is ROTEM?
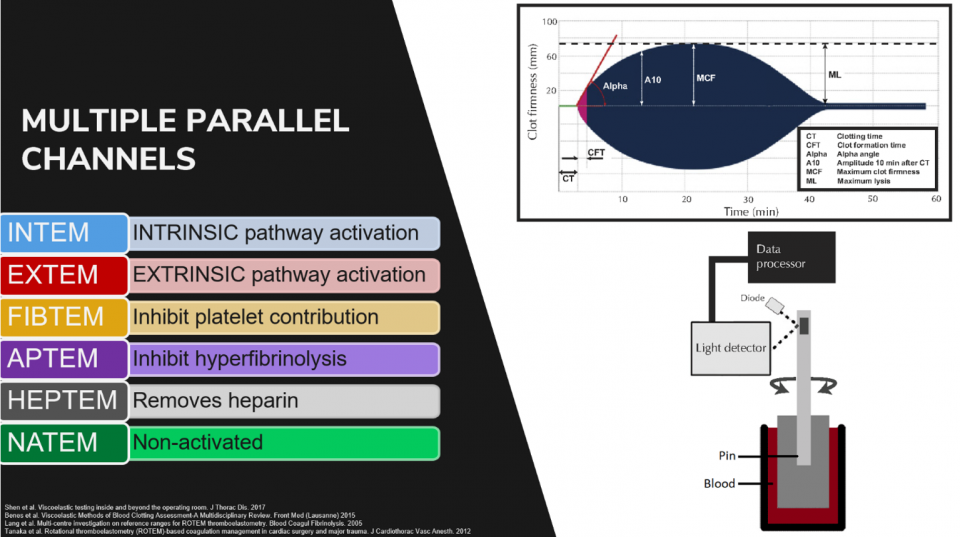
ROTEM measures clot formation over time. It is a dynamic assessment of clot formation, strength, stability, and dissolution. It measures the global viscoelastic properties of whole blood clot formation.

- A small sample of patient blood is placed into a cup
- A sensor rod is inserted into the blood sample
- The rod is then gently rotated, with a subsequent clot forming between the cup and rod
- As blood clots form and strengthen, they exhibit increasing viscosity and elasticity
- This leads to increased resistance on the pin’s rotation
- This is converted to electrical signals to create 2D graphical/numerical output
- In ROTEM, the pin rotates. In TEG, the cup rotates.
- There are multiple parallel channels that test different aspects of clotting using different reagents, but for our purposes, we will focus on EXTEM and FIBTEM
What are the Advantages of ROTEM?
Minor Advantages
- Faster
- Point-of-care test – first results 5-10min, full results 30min
- Results are displayed in both graphical and numerical format with reference ranges
- Assessment of global hemostatic potential (whole blood, not just plasma and fibrin formation)
- Detects specific portion of clotting that’s disrupted
- Detect specific defects in coagulation
- e. hypofibrinogenemia, hyperfibrinolysis, factor deficiency, heparin effect
- Can assess hyperfibrinolysis
- This is unique to VHAs over SLTs; No SLT to assess hyperfibrinolysis
- This is an important aspect of ATC as it has been associated with increased transfusion requirements and mortality
- Can perform at multiple temperatures
- Therefore: rapid diagnosis of specific coagulopathies, suggesting specific treatments which may reduce transfusion requirement, decrease hemorrhage, and decrease mortality…?
Goal-directed Hemostatic Resuscitation
- ROTEM correlates with SLTs, assesses TIC/ATC, and predicts massive transfusion, need for transfusion, fibrinolysis, and coagulopathy-related mortality
- Numerous studies verifying the above, looking at different aspects of ROTEM/TEG with different measurements and cut-offs which are the basis for management algorithms

- In terms of Trauma-specific evidence for goal-directed hemostatic resuscitation, there’s the Da Luz 2014 systematic review
- The purpose was to review the evidence for VHAs in diagnosing early coagulopathies, guiding blood transfusion, and reducing mortality in injured patients
- Looked at observational studies and RCTs
- 55 studies (n = 12,489). 38 prospective cohort. 15 retrospective cohort. 2 before-after. No RCTs.
- Studies investigated TEG/ROTEM for diagnosis of early coagulopathies (n = 40) or for associations with blood-product transfusion (n = 25) or mortality (n = 24).
- Methodologic quality was moderate. Only 3 low risk of bias.
- Most (n = 52) were single-center studies.
- Standard measures of diagnostic accuracy were inconsistently reported.
- Many abnormalities predicted the need for massive transfusion and death, but predictive performance was not consistently superior to routine tests.
- CONCLUSIONS: Limited evidence from observational data suggest that TEG/ROTEM diagnose early trauma coagulopathy and may predict blood-product transfusion and mortality in trauma. Effects on blood-product transfusion, mortality, and other patient-important outcomes remain unproven in randomized trials.
Conservation of Blood Products
In terms of conservation of blood products – which is important from a resource stewardship, cost, and safety standpoint – there are two meta analyses and one systematic review.
- Wikkelsø et al.
- Included 17 trials (n = 1493), most involving cardiac surgery.
- VHA-guided vs standard MTP significantly reduced the proportion transfused with RBCs, FFP, and platelets.
- Conclusion: Transfusion strategies guided by VHAs may reduce the need for blood products in bleeding, but results are mainly based on elective cardiac surgery involving cardiopulmonary bypass, with low-quality evidence.
- Fahrendorff et al. 2017
- Also looked at RCTs. VHA algorithm vs standard. Outcomes were bleeding, transfusion, mortality. 15 RCTs (n = 1238). 9 cardiothoracic, 1 liver transplant, 1 surgical burn wounds, 1 trauma, 1 cirrhotic, 1 PPH.
- Conclusion: The amount of transfused RBCs, FFP, and bleeding volume was found to be significantly reduced in the VHA-guided groups, whereas no significant difference was found for platelet transfusion requirements or mortality.
- Whiting et al. 2014
- RCTs and observational studies. 31 studies. 11 RCTs (n = 1089, 6 TEG, 5 ROTEM).
- Goal: Clinical effectiveness and cost-effectiveness of VHAs in cardiac surgery, trauma-induced coagulopathy, and PPH.
- Conclusion: Reduction in RBC transfusion, platelet transfusion, and FFP transfusion compared with control. Some evidence suggests these algorithms might reduced transfusions, further study is needed to assess patient outcomes
- Clinical outcomes did not differ significantly. No differences b/w TEG/ROTEM.
There is also a 2016 Cochrane review which we will talk about below, but this also found decreased transfusion requirements with VHAs, concluding there is growing evidence to support conservation of blood products.
All in all, there is low quality, mostly cardiac surgery evidence that VHA-guided MTPs reduce transfusion and decreased bleeding.
Mortality Benefit
There are two Cochrane reviews, a meta-analysis that we just went over by Wikkelsø et al., and a Haematology guideline that suggest mortality benefit.
- Cochrane 2011
- The first Cochrane review was in 2011 and concluded there was an absence of evidence that VHAs improve M/M in patients with severe bleeding
- Cochrane 2016 (update)
- The same group revisited the question 5 years later.
- Including only RCTs they found 8 new studies (n = 617) for a total of 17 studies (n = 1493). 6 ongoing trials (unable to get data).
- 15 trials were suitable for meta-analysis.
- Majority = cardiac surgery patients (1435/1493 patients); though there were also liver transplantation, trauma, and orthopedic surgery
- Results:
- Reduce overall mortality – tendency towards improved mortality outcomes (3.9% vs 7.4%, RR 0.52, 95% CI, 0.28–0.95), though only 8 trials had data (n = 717), 2 being zero event trials. They stated that firm conclusions could not be made due to limitations of study design and power.
- They also found:
- Decreased dialysis dependent renal failure.
- No difference in surgical re-interventions, excessive bleeding events, massive transfusion.
- Low quality evidence based on high risk of bias (only 2 studies were low risk), large heterogeneity, low number of events, imprecision, indirectness.
- TSA indicates 54% information size for mortality.
- Conclusion: There is growing evidence that VHA-guided transfusion may reduce the need for blood products and improve morbidity in patients with bleeding. However, these results are primarily based on trials of elective cardiac surgery involving cardiopulmonary bypass, and the level of evidence remains low. Further evaluation of TEG- or ROTEM-guided transfusion in acute settings and other patient categories in low risk of bias studies is needed.
- Anesthesia Meta-analysis. 2017.
- Same review we just went over for reduction in blood products
- VHA seemed to reduce overall mortality, however, quality of evidence graded as low due to high risk of bias, heterogeneity, imprecision, and low event rate.
- British Society for Haematology Guideline 2018
- Conclusion: based on current evidence = VHA, particularly TEG, may reduce mortality and reduce transfusion exposure and, if available, may be considered for transfusion guidance in trauma haemorrhage. Grade 2B.
- The Bottom line is that: there is low quality evidence, mainly in cardiac surgery, but growing evidence of possible mortality benefit

Several studies are emerging and underway looking into the question of mortality benefit. One that wasn’t included in the Cochrane review was that of Gonzalez et al. from 2016.
- Pragmatic RCT of TEG-guided vs conventional test guided MTP.
- Level 1 trauma center in Denver USA. Patients enrolled during MTP activation.
- The primary outcome was 28-d survival.
- N = 111. IIT.
- Mortality 19.6% (n = 11) vs 36.4% (n = 20). Most deaths w/n 6h.
- Numbers of haemorrhagic deaths lower (8.9% vs. 20%).
- TEG-guided resuscitation = fewer blood products overall, increased ICU- and ventilator-free days.
- Conclusion: Utilization of a goal-directed, TEG-guided MTP to resuscitate severely injured patients improves survival compared with an MTP guided by SLTs and utilizes less plasma and platelet transfusions during the early phase of resuscitation
- This study provides evidence that VHA‐guided transfusion may be beneficial for the management of acute bleeding in trauma, over and above the effects of the empiric 1:1:1 transfusion.
iTACTIC (underway)
- The Implementing Treatment Algorithms for the Correction of Trauma-Induced Coagulopathy Trial (iTACTIC) aims to recruit 400 patients across Europe, randomizing them to a TEG-guided or CCT-guided resuscitation
FOAM Resources
- https://www.emra.org/emresident/article/teg-and-rotem/
- https://litfl.com/thromboelastogram-teg/
- https://foamcast.org/2019/07/31/thromboelastography-teg-guided-resuscitation/
- http://www.emdocs.net/thromboelastography-teg-traum/
- https://rebelem.com/rebel-review/rebel-review-54-thromboelastogram-teg/thromboelastogram-teg/
- https://derangedphysiology.com/main/required-reading/haematology-and-oncology/Chapter%20120/viscoelastic-tests-clotting-function-teg-and-rotem
- https://www.acep.org/how-we-serve/sections/critical-care-medicine/news/april-2019/the-role-of-thromboelastometry-in-monitoring-critical-patients/
References
- Abdelfattah K, Cripps MW. Thromboelastography and rotational thromboelastometry use in trauma. Int J Surg. 2016;33:196–201.
- Afshari A, Wikkelsø A, Brok J, Møller AM, Wetterslev J. Thrombelastography (TEG) or thromboelastometry (ROTEM) to monitor haemotherapy versus usual care in patients with massive transfusion. Cochrane Database Syst Rev. 2011 Mar 16;(3):CD007871
- Benes J, Zatloukal J, Kletecka J. Viscoelastic Methods of Blood Clotting Assessment-A Multidisciplinary Review. Front Med (Lausanne) 2015;2:62.
- Bolliger D, Seeberger MD, Tanaka KA. Principles and practice of thromboelastography in clinical coagulation management and transfusion practice. Transfus Med Rev. 2012 Jan;26(1):1-13
- Borgman MA, Spinella PC, Perkins JG, Grathwohl KW, Repine T, Beekley AC, Sebesta J, Jenkins D, Wade CE, Holcomb JB. The ratio of blood products transfused affects mortality in patients receiving massive transfusions at a combat support hospital. J Trauma 2007;63:805–13.doi:10.1097/TA.0b013e3181271ba3
- Briggs C, Kimber S, Green L. Where are we at with point- of- care testing in haematology? Br J Haematol 2012;158:679-90. 10.1111/j.1365-2141.2012.09207.x
- Brohi K, Cohen MJ, Ganter MT, Schultz MJ, Levi M, Mackersie RC, Pittet JF. Acute coagulopathy of trauma: hypoperfusion induces systemic anticoagulation and hyperfibrinolysis. J Trauma 2008;64:1211–7. discussion 7.doi:10.1097/TA.0b013e318169cd3
- Brohi K, Singh J, Heron M, Coats T. Acute traumatic coagulopathy. J Trauma 2003;54:1127–30.doi:10.1097/01.TA.0000069184.82147.06
- Camazine MN, Hemmila MR, Leonard JC, Jacobs RA, Horst JA, Kozar RA, et al. Massive transfusion policies at trauma centers participating in the American College of Surgeons Trauma Quality Improvement Program. J Trauma Acute Care Surg. 2015;78:S48–53
- Carroll RC, Craft RM, Langdon RJ, Clanton CR, Snider CC, Wellons DD, Dakin PA, Lawson CM, Enderson BL, Kurek SJ. Early evaluation of acute traumatic coagulopathy by thrombelastography. Transl Res. 2009;154:34–9.
- Chapman MP, Moore EE, Moore HB, et al. The “Death Diamond.” J Trauma Acute Care Surg 2015;79:925-9. 10.1097/TA.0000000000000871
- Chapman MP, Moore EE, Ramos CR, et al. Fibrinolysis greater than 3% is the critical value for initiation of antifibrinolytic therapy. J Trauma Acute Care Surg. 2013;75(6):961-967; discussion 967.
- Chee YL, Greaves M. Role of coagulation testing in predicting bleeding risk. Hematol J Off J Eur Haematol Assoc 2003;4:373-8.
- Chen J, Gorman M, O’Reilly B, et al. Analytical evaluation of the epoc® point-of-care blood analysis system in cardiopulmonary bypass patients. Clin Biochem 2016;49:708-12. 10.1016/j.clinbiochem.2015.12.015
- Coakley, Margaret, et al. “Transfusion triggers in orthotopic liver transplantation: a comparison of the thromboelastometry analyzer, the thromboelastogram, and conventional coagulation tests.” Journal of cardiothoracic and vascular anesthesia 20.4 (2006): 548-553.
- Cohen MJ, Call M, Nelson M, Calfee CS, Esmon CT, Brohi K, Pittet JF. Critical role of activated protein C in early coagulopathy and later organ failure, infection and death in trauma patients. Ann Surg 2012;255:379–85.doi:10.1097/SLA.0b013e318235d9e6
- Collins PW, Solomon C, Sutor K, Crispin D, Hochleitner G, Rizoli S, et al. Theoretical modelling of fibrinogen supplementation with therapeutic plasma, cryoprecipitate, or fibrinogen concentrate. Br J Anaesth. 2014;113:585–95.
- Corredor C, Wasowicz M, Karkouti K, et al. The role of point-of-care platelet function testing in predicting postoperative bleeding following cardiac surgery: a systematic review and meta-analysis. Anaesthesia 2015;70:715-31. 10.1111/anae.13083
- Cotton BA, Faz G, Hatch QM, et al. Rapid thrombelastography delivers real-time results that predict transfusion within 1 hour of admission. J Trauma 2011;71:407-14; discussion 414-7. 10.1097/TA.0b013e31821e1bf0
- CRASH-2 Trial Collaborators. Effects of Tranexamic Acid on Death, Vascular Occlusive Events, and Blood Transfusion in Trauma Patients with Significant Haemorrhage (CRASH-2): A Randomised, Placebo-Controlled Trial. Lancet. 2010;376 (9734):23–32.
- Curry NS, Davenport R, Pavord S, Mallett SV, Kitchen D, Klein AA, Maybury H, Collins PW, and Laffan M. The use of viscoelastic haemostatic assays in the management of major bleeding: A British Society for Haematology Guideline. First published:02 August 2018 https://doi.org/10.1111/bjh.15524
- Da Luz LT, Nascimento B, Rizoli S. Thrombelastography (TEG(R)): practical considerations on its clinical use in trauma resuscitation. Scand J Trauma Resusc Emerg Med. 2013 Apr 16;21(1):29.
- Da Luz, LT, Nascimento, B, Shankarakutty, AK, Rizoli, S, Adhikari, NKJ. (2014). Effect of thromboelastography (TEG®) and rotational thromboelastometry (ROTEM®) on diagnosis of coagulopathy, transfusion guidance and mortality in trauma: descriptive systematic review. Critical Care 2014, 18:518
- Davenport R, Manson J, De’Ath H, et al. Functional definition and characterization of acute traumatic coagulopathy. Crit Care Med 2011;39:2652-8. 10.1097/CCM.0b013e3182281af5
- Delaney M, Wendel S, Bercovitz RS, et al. Transfusion reactions: prevention, diagnosis, and treatment. Lancet 2016;388:2825-36. 10.1016/S0140-6736(15)01313-6
- Deppe A-C, Weber C, Zimmermann J, et al. Point-of-care thromboelastography/ thromboelastometry-based coagulation management in cardiac surgery: a meta-analysis of 8332 patients. J Surg Res. 2016 Jun 15;203(2):424-33.
- Dias JD, Norem K, Doorneweerd DD, et al. Use of thromboelastography (TEG) for detection of new oral anticoagulants. Arch Pathol Lab Med. 2015 May;139(5):665-73.
- Dzik WH1, Blajchman MA, Fergusson D, Hameed M, Henry B, Kirkpatrick AW, Korogyi T, Logsetty S, Skeate RC, Stanworth S, MacAdams C, Muirhead B. Clinical review: Canadian National Advisory Committee on Blood and Blood Products–Massive transfusion consensus conference 2011: report of the panel. Crit Care. 2011;15(6):242. doi: 10.1186/cc10498. Epub 2011 Dec 8.
- Einersen PM, et al. Rapid thrombelastography thresholds for goal-directed resuscitation of patients at risk for massive transfusion. J Trauma Acute Care Surg. 2017;82(1):114.
- Fahrendorff M, Oliveri RS, Johansson PI. The Use of Viscoelastic Haemostatic Assays in Goal-Directed Treatment with Allogeneic Blood Products – A Systematic Review and Meta-Analysis. Scand J Trauma Resusc Emerg Med. 2017.
- Ganter MT, Hofer CK. Coagulation monitoring: current techniques and clinical use of viscoelastic point-of-care coagulation devices. Anesth Analg. 2008 May;106(5):1366-75
- Gonzalez, Eduardo, et al. “Goal-directed hemostatic resuscitation of trauma-induced coagulopathy: a pragmatic randomized clinical trial comparing a viscoelastic assay to conventional coagulation assays.” Annals of surgery 263.6 (2016): 1051.
- Goodman MD, Makley AT, Hanseman DJ, Pritts TA, Robinson BR. All the bang without the bucks: Defining essential point-of-care testing for traumatic coagulopathy. J Trauma Acute Care Surg. 2015;79(1):117-24.
- Hartert H. Blutgerinnungsstudien mit der Thrombelastographie, einem neuen Untersuchungsverfahren. Klin Wochenschr 1948;26:577-83. 10.1007/BF01697545
- Holcomb JB, Minei KM, Scerbo ML, Radwan ZA, Wade CE, Kozar RA, Gill BS, Albarado R, McNutt MK, Khan S, et al. Admission rapid thrombelastography can replace conventional coagulation tests in the emergency department: experience with 1974 consecutive trauma patients. Ann Surg 2012;256:476–86.doi:10.1097/SLA.0b013e3182658180
- Holcomb JB, Tilley BC, Baraniuk S, Fox EE, Wade CE, Podbielski JM, del Junco DJ, Brasel KJ, Bulger EM, Callcut RA, et al. Transfusion of plasma, platelets, and red blood cells in a 1:1:1 vs a 1:1:2 ratio and mortality in patients with severe trauma: the PROPPR randomized clinical trial. JAMA 2015;313:471–82.doi:10.1001/jama.2015.12
- Howley IW, Haut ER, Jacobs L, et al. Thromboelastography (TEG)-based resuscitation better than empirical 1:1 transfusion? Trauma Surgery & Acute Care Open 2018;3:e000140. doi: 10.1136/tsaco-2017-000140
- Hunt H, Stanworth S, Curry N, et al. Thromboelastography (TEG) and rotational thromboelastometry (ROTEM) for trauma-induced coagulopathy in adult trauma patients with bleeding. In: Hunt H. editor. Cochrane Database of Systematic Reviews. Chichester, UK: John Wiley & Sons, Ltd, 2015:CD010438.
- Hunt H, Stanworth S, Curry N, Woolley T, Cooper C, Ukoumunne O, Zhelev Z, Hyde C. TEG and ROTEM for diagnosing trauma induced coagulopathy (disorder of the clotting system) in adult trauma patients with bleeding. Cochcane Review. 2015
- Inaba, Kenji MD; Rizoli, Sandro MD; Veigas, Precilla V. MSc; Callum, Jeannie MD; Davenport, Ross MD; Hess, John MD; Maegele, Marc MD and the Viscoelastic Testing in Trauma Consensus Panel. 2014 Consensus conference on viscoelastic test–based transfusion guidelines for early trauma resuscitation: Report of the panel. Journal of Trauma and Acute Care Surgery: June 2015 – Volume 78 – Issue 6 – p 1220-1229. doi: 10.1097/TA.0000000000000657
- Jansen JO, Luke D, Davies E, Spencer P, Kirkman E, Midwinter MJ. Temporal changes in ROTEM®-measured coagulability of citrated blood samples from coagulopathic trauma patients. Injury. 2013;44:36–9.
- Kander T, Larsson A, Taune V, Schött U, Tynngård N (2016) Assessment of Haemostasis in Disseminated Intravascular Coagulation by Use of Point-of-Care Assays and Routine Coagulation Tests, in Critically Ill Patients; A Prospective Observational Study. PLoS ONE 11(3): e0151202. doi:10.1371/ journal.pone.0151202
- Kang YG, Martin DJ, Marquez J, et al. Intraoperative changes in blood coagulation and thrombelastographic monitoring in liver transplantation. Anesth Analg 1985;64:888-96. 10.1213/00000539-198509000-00008
- Karkouti K, Callum J, Wijeysundera DN, Rao V, Crowther M, Grocott HP, Pinto R, Scales DC; TACS Investigators. Point-of-Care Hemostatic Testing in Cardiac Surgery: A Stepped-Wedge Clustered Randomized Controlled Trial. Circulation. 2016 Oct 18;134(16):1152-1162.
- Kaufner L, Henkelmann A, von Heymann C, et al. Can prepartum thromboelastometry-derived parameters and fibrinogen levels really predict postpartum hemorrhage? J Perinat Med. 2017 May 24;45(4):427-35.
- Kornblith LZ, Kutcher ME, Redick BJ, Calfee CS, Vilardi RF, Cohen MJ. Fibrinogen and platelet contributions to clot formation: implications for trauma resuscitation and thromboprophylaxis. J Trauma Acute Care Surg. 2014;76:255–63.
- Kozek-Langenecker SA, Ahmed AB, Afshari A, Albaladejo P, Aldecoa C, Barauskas G, De Robertis E, Faraoni D, Filipescu DC, Fries D, Haas T, Jacob M, Lancé MD, Pitarch JVL, Mallett S, Meier J, Molnar ZL, Rahe-Meyer N, Samama CM, Stensballe J, Van der Linden PJF, Wikkelsø AJ, Wouters P, Wyffels P, Zacharowski K. Management of severe perioperative bleeding: guidelines from the European Society of Anesthesiology First update 2016. Eur J Anaesthesiol. 2017;34:332–395.
- Kumar M, Ahmad J, Maiwall R, et al. Thromboelastography-Guided Blood Component Use in Patients With Cirrhosis With Nonvariceal Bleeding: A Randomized Controlled Trial. Hepatology. 2019; In Press
- Lang T, Bauters A, Braun SL, et al. Multi-centre investigation on reference ranges for ROTEM thromboelastometry. Blood Coagul Fibrinolysis 2005;16:301-10. 10.1097/01.mbc.0000169225.31173.19
- Lee FM, Chan AK, Lau KK, et al. Reversal of New, Factor-specific Oral Anticoagulants by rFVIIa, Prothrombin Complex Concentrate and Activated Prothrombin Complex Concentrate: A Review of Animal and Human Studies. Thromb Res 2014;133:705-13. 10.1016/j.thromres.2014.01.031
- Leemann H, Lustenberger T, Talving P, et al. The role of rotation thromboelastometry in early prediction of massive transfusion. J Trauma 2010;69:1403-8-9.
- Levy JH, Welsby I, Goodnough LT. Fibrinogen as a therapeutic target for bleeding: a review of critical levels and replacement therapy. Transfusion. 2014;54:1389–405.
- Luddington RJ. Thrombelastography/thromboelastometry. Clin Lab Haematol 2005;27:81-90. 10.1111/j.1365-2257.2005.00681.x
- Mahamad S, Chaudhry H, Nisenbaum R, et al. Exploring the effect of factor Xa inhibitors on rotational thromboelastometry: a case series of bleeding patients. J Thromb Thrombolysis. 2019 Feb;47(2):272-9.
- Midwinter MJ , Woolley T. Resuscitation and coagulation in the severely injured trauma patient. Philos Trans R Soc Lond B Biol Sci 2011;366:192–203.doi:10.1098/rstb.2010.0220
- Morrison JJ. Noncompressible torso hemorrhage. Crit Care Clin 2017;33:37–54.doi:10.1016/j.ccc.2016.09.001
- Muszynski JA, Spinella PC, Cholette JM, et al. Transfusion-related immunomodulation: review of the literature and implications for pediatric critical illness. Transfusion 2017;57:195-206. 10.1111/trf.13855
- Nielsen, Vance G. “A comparison of the Thrombelastograph and the ROTEM.” Blood Coagulation & Fibrinolysis 18.3 (2007): 247-252
- Nystrup KB, Windeløv NA, Thomsen AB, Johansson PI. Reduced clot strength upon admission, evaluated by thrombelastography (TEG®), in trauma patients is independently associated with increased 30-day mortality. Scand J Trauma Resusc Emerg Med. 2011;19:52.
- Park MS, Salinas J, Wade CE, Wang J, Martini W, Pusateri AE, Merrill GA, Chung K, Wolf SE, Holcomb JB. Combining early coagulation and inflammatory status improves prediction of mortality in burned and nonburned trauma patients. J Trauma. 2008;64:S188–94.
- Pezold M, Moore EE, Wohlauer M, et al. Viscoelastic clot strength predicts coagulation-related mortality within 15 minutes. Surgery 2012;151:48-54. 10.1016/j.surg.2011.06.023
- Practice guidelines for perioperative blood management: an updated report by the American Society of Anesthesiologists Task Force on Perioperative Blood Management. American Society of Anesthesiologists Task Force on Perioperative Blood Management. Anesthesiology. 2015 Feb;122(2):241-75. doi: 10.1097/ALN.0000000000000463. PMID: 25545654 DOI: 10.1097/ALN.0000000000000463
- Rizoli S, Min A, Sanchez AP, Shek P, Grodecki R, Veigas P, Peng HT. In Trauma, Conventional ROTEM and TEG Results Are Not Interchangeable But Are Similar in Clinical Applicability. Mil Med. 2016 May;181(5 Suppl):117-26. doi: 10.7205/MILMED-D-15-00166.
- Rossaint R, Bouillon B, Cerny V, et al. The European guideline on management of major bleeding and coagulopathy following trauma: fourth edition. Crit Care 2016;20:100.
- Rout G, Shalimar, Gunjan D, et al. Thromboelastography-guided Blood Product Transfusion in Cirrhosis Patients With Variceal Bleeding: A Randomized Controlled Trial. J Clin Gastroenterol. 2019; In Press.
- Rugeri L, et al. Diagnosis of early coagulation abnormalities in trauma patients by rotation thrombelastography. J Thromb Haemost. 2007;5(2):289.
- Sakamoto, Y., Koami, H. & Miike, T. Monitoring the coagulation status of trauma patients with viscoelastic devices. j intensive care 5, 7 (2017).
- Sankarankutty, Ajith, et al. “TEG® and ROTEM® in trauma: similar test but different results.” World J Emerg Surg 7.Suppl 1 (2012): S3.
- Schöchl H, Cotton B, Inaba K, et al. FIBTEM provides early prediction of massive transfusion in trauma. Crit Care 2011;15:R265. 10.1186/cc10539
- Schochl H, Cotton B, Inaba K, Nienaber U, Fischer H, Voelckel W, et al. FIBTEM provides early prediction of massive transfusion in trauma. Crit Care. 2011;15:R265.
- Schöchl H, Frietsch T, Pavelka M, Jambor C. Hyperfibrinolysis after major trauma: differential diagnosis of lysis patterns and prognostic value of thromboelastometry. J Trauma. 2009;67:125–31.
- Scholchl H, Nienaber U, Hofer G, Voelckel W, Jambor C, Scharbert G, et al. Goal-directed coagulation management of major trauma patients using thromboelastometry (ROTEM®)-guided administration of fibrinogen concentrate and prothrombin complex concentrate. Crit Care. 2010;14:R55.
- Shen L, Tabaie S, Ivascu N. Viscoelastic testing inside and beyond the operating room. J Thorac Dis. 2017;9(Suppl 4):S299–S308. doi:10.21037/jtd.2017.03.85
- Smart L, Mumtaz K, Scharpf D, et al. Rotational thromboelastometry or conventional coagulation tests in liver transplantation: comparing blood loss, transfusions, and cost. Ann Hepatol. 2017 Nov-Dec;16(6):916-23.
- Snegovskikh D, Souza D, Walton Z, et al. Point-of-care viscoelastic testing improves the outcome of pregnancies complicated by severe postpartum hemorrhage. J Clin Anesth. 2018 Feb;44:50-6.
- Spahn DR, Bouillon B, Cerny V, Coats TJ, Duranteau J, Fernandez-Mondejar E, et al. Management of bleeding and coagulopathy following major trauma: an updated European guideline. Crit Care. 2013;17:R76.
- Tanaka KA, Bolliger D, Vadlamudi R, Nimmo A. Rotational thromboelastometry (ROTEM)-based coagulation management in cardiac surgery and major trauma. J Cardiothorac Vasc Anesth. 2012;26:1083–93.
- Tapia NM et al. TEG-guided resuscitation is superior to standardized MTP resuscitation in massively transfused penetrating trauma patients. J Trauma Acute Care Surg. 2013;74:378-386
- Tapia NM, Chang A, Norman M, et al. TEG-guided resuscitation is superior to standardized MTP resuscitation in massively transfused penetrating trauma patients. J Trauma Acute Care Surg. 2013 Feb;74(2):378-85; discussion 385-6.
- Tauber H, Innerhofer P, Breitkopf R, Westermann I, Beer R, El Attal R, Strasak A, Mittermayr M. Prevalence and impact of abnormal ROTEM® assays in severe blunt trauma: results of the ‘Diagnosis and Treatment of Trauma-Induced Coagulopathy (DIA-TRE-TIC) study’. Br J Anaesth. 2011;107:378–87.
- Thakur M, Ahmed AB. A review of thromboelastography. Int J Periop Ultrasound Appl Technol 2012;1(1):25-29.
- Trentalange MJ, Walts LF. A comparison of thromboelastogram and template bleeding time in the evaluation of platelet function after aspirin ingestion. J Clin Anesth. 1991 Sep-Oct;3(5):377-81.
- Venema, Lieneke F., et al. “An assessment of clinical interchangeability of TEG® and ROTEM® thromboelastographic variables in cardiac surgical patients.” Anesthesia & Analgesia 111.2 (2010): 339-344.
- Whiting D, DiNardo JA. 2014. TEG and ROTEM: Technology and Clinical Applications. Am J Hematol. 2014;89(2):228–232.
- Whiting P, Al M, Westwood M, et al. Viscoelastic Point-of-Care Testing to Assist with the Diagnosis, Management and Monitoring of Haemostasis: A Systematic Review and Cost-Effectiveness Analysis. Health Technol Assess. 2015;19(58)1-228.
- Wikkelsø A, Wetterslev J, Møller AM, Afshari A. Thromboelastography (TEG) or Rotational Thromboelastometry (ROTEM) to Monitor Haemostatic Treatment in Bleeding Patients: A Systematic Review with Meta-Analysis and Trial Sequential Analysis. Anaesthesia. 2017;72(4):519–531
- Wikkelsø A, Wetterslev J, Møller AM, Afshari A. Thromboelastography (TEG) or thromboelastometry (ROTEM) to monitor haemostatic treatment versus usual care in adults or children with bleeding. Cochrane Database Syst Rev. 2016;(8):CD007871
- Wikkelsø A, Wetterslev J, Møller AM, et al. Thromboelastography (TEG) or thromboelastometry (ROTEM) to monitor haemostatic treatment versus usual care in adults or children with bleeding. In: Afshari A. editor. Cochrane Database of Systematic Reviews. Chichester, UK: John Wiley & Sons, Ltd, 2016:CD007871.
- Yin J, Zhao Z, Li Y, Wang J, Yao D, Zhang S, et al. Goal-directed transfusion protocol via thrombelastography in patients with abdominal trauma: a retrospective study. World J Emerg Surg. 2014;9:28.


