In Part 2 of ROTEM for Trauma: Blood is Thicker with Wine – we will review the approach to interpreting ROTEM and how it can guide massive transfusion in the injured and bleeding patient. We will also touch on the practical considerations for starting a ROTEM program at your centre. To review the evidence for ROTEM in trauma, please see Part 1, which can be found here.
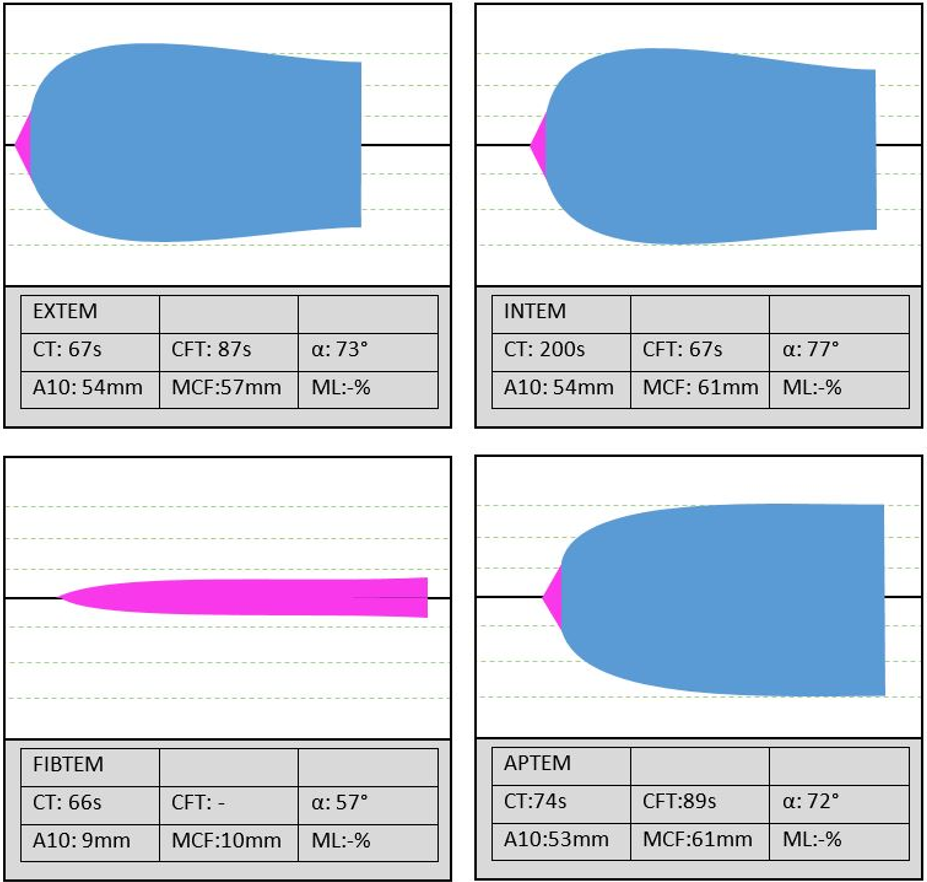
This is a normal ROTEM output. For the purposes of acute bleeding in trauma, we focus on EXTEM and use FIBTEM as required.
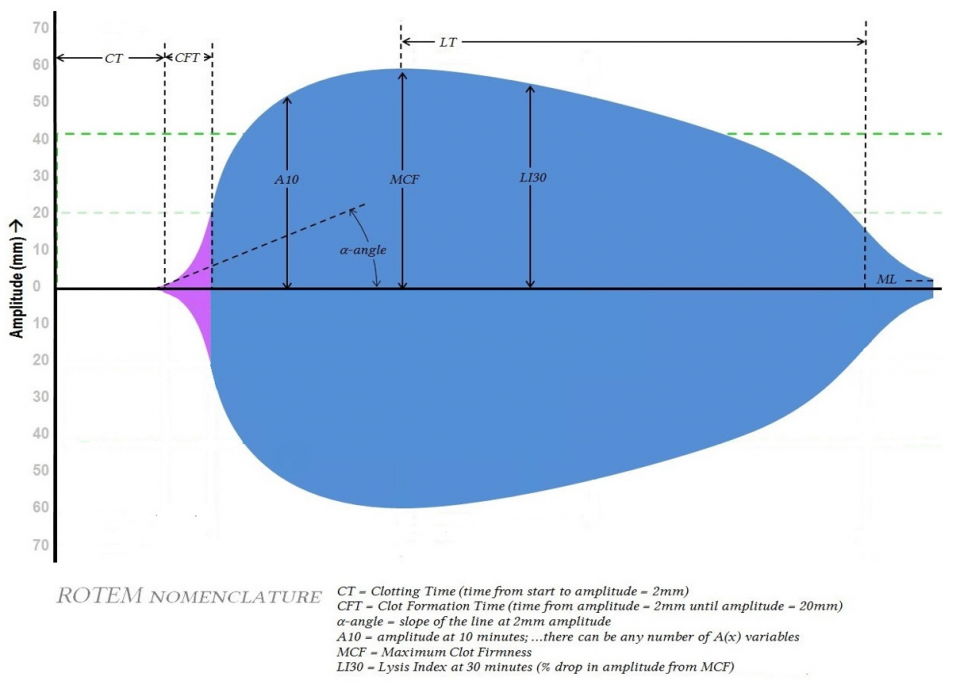
We’ll present the approach to ROTEM in multiple formats (i.e. pictorial, flowchart, chart, an analogy to drinking glasses, algorithm, etc.)
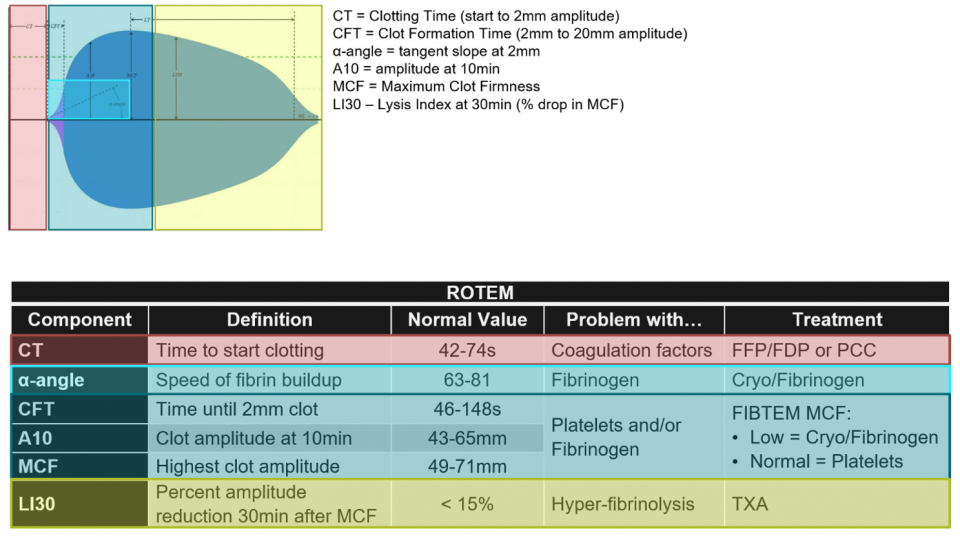
- The output shows the amplitude of the forming clot over time
- The important variables for acute management of trauma are:
- Clotting Time (CT) –time from test start to amplitude 2mm; initiation of clotting by clotting factors
- α-angle – slope of tangent at 2mm; speed of fibrin accumulation (thrombin burst, propagation)
- Maximum clot firmness (MCF) – maximum clot amplitude; dependent on fibrinogen and platelets
- Now we can get fancy and talk about the clot formation time (CFT ) and A10
- CFT – time from amplitude 2 to 20mm; amplification, mostly fibrinogen dependent
- A10 – amplitude at 10min, surrogate for MCF (allows same guided approach without waiting the full test time for MCF)
- Lysis index at 30mins (LI30) – % drop in amplitude from MCF at 30min; hyperfibrinolysis
- Now we can get fancy and talk about the clot formation time (CFT ) and A10
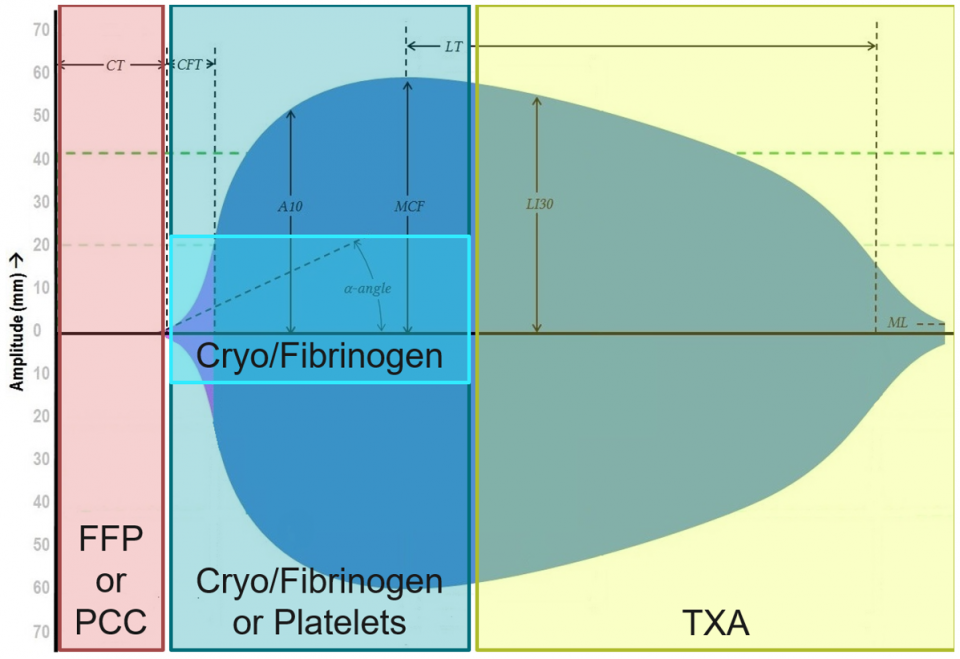
- Basically:
- Increased clotting time (EXTEM) – clotting factor issue – give FFP/FDP
- Decreased alpha angle or CFT – fibrinogen issue – cryoprecipitate/fibrinogen
- Decreased MCF – platelets or fibrinogen – examine FIBTEM to determine which to give
- Increased LI30 – Hyperfibrinolysis – give tranexamic acid
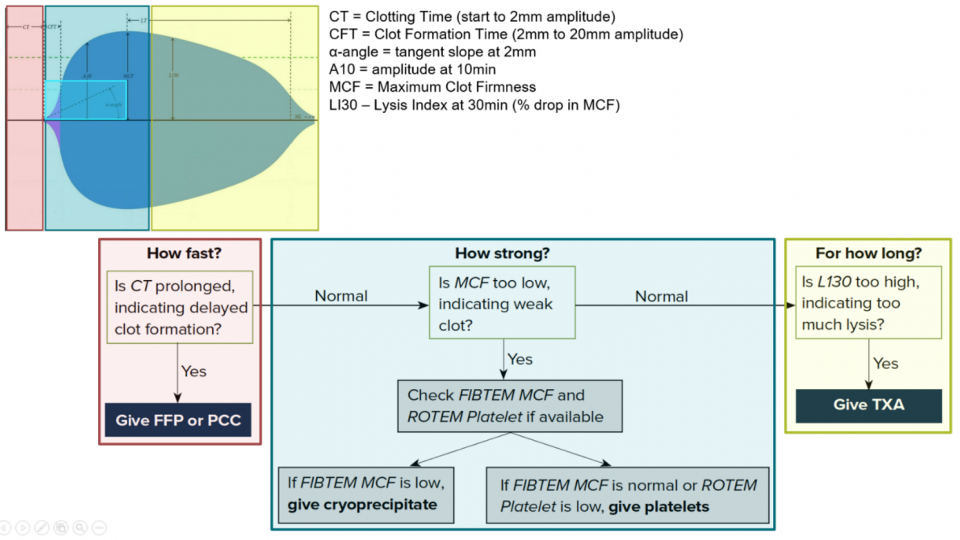
The approach below was originally posted by EMRA and is focused on 3 simple questions: (1) how fast, (2) how strong, and (3) for how long?
- How fast?
- Initiation phase: Clotting factor dependent
- CT on EXTEM – FFP/FDP, PCC
- How strong?
- Fibrin and platelet dependent
- MCF on EXTEM and FIBTEM
- MCF represents overall stability of clot (platelets 80%, fibrin 20% interacting via GPIIb/IIIa)
- If abnormal, must assess fibrin and platelet contribution separately
- If FIBTEM abnormal – replace fibrinogen (fibrinogen or cryo)
- If MCF abnormal but FIBTEM normal – replace platelet
- If BOTH abnormal – treat and repeat test; whenever you replace one, run test again to detect if deficiency in other
- For how long?
- Fibrinolysis
- Overactive Plasmin causes hyperfibrinolysis
- Lysis at 30min and 60min – (LY30, LY60) – TXA
Let’s look at the approach in tabular format:
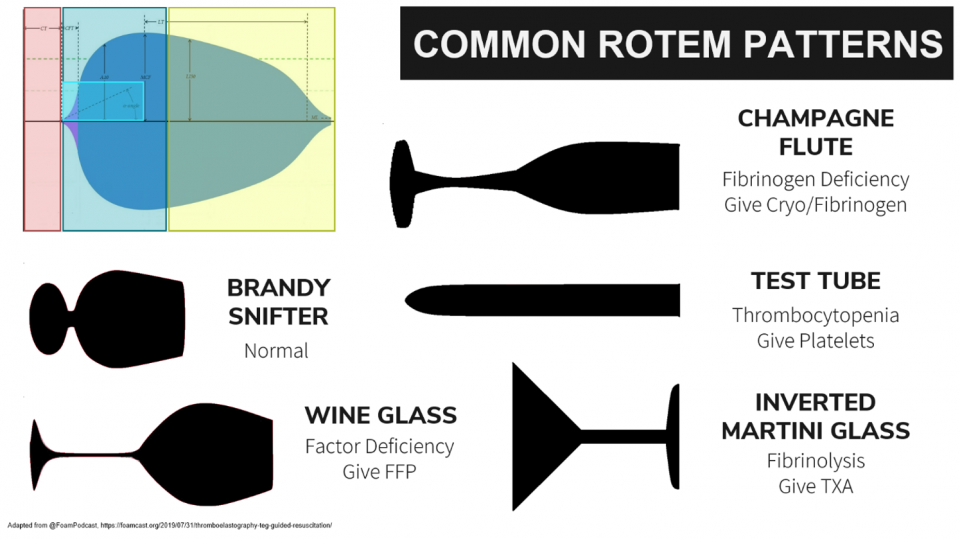
That brings us to likely the most famous of interpretation techniques – the comparison of ROTEM/TEG outputs to drinking glasses:
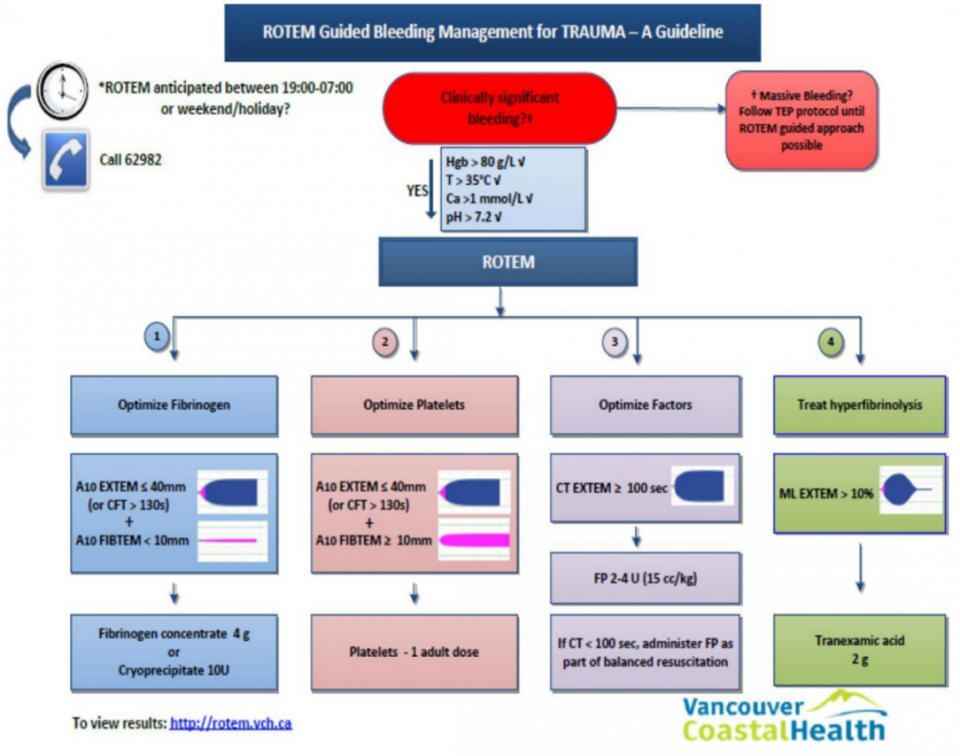
Below is a Canadian transfusion algorithm currently in use based on ROTEM:
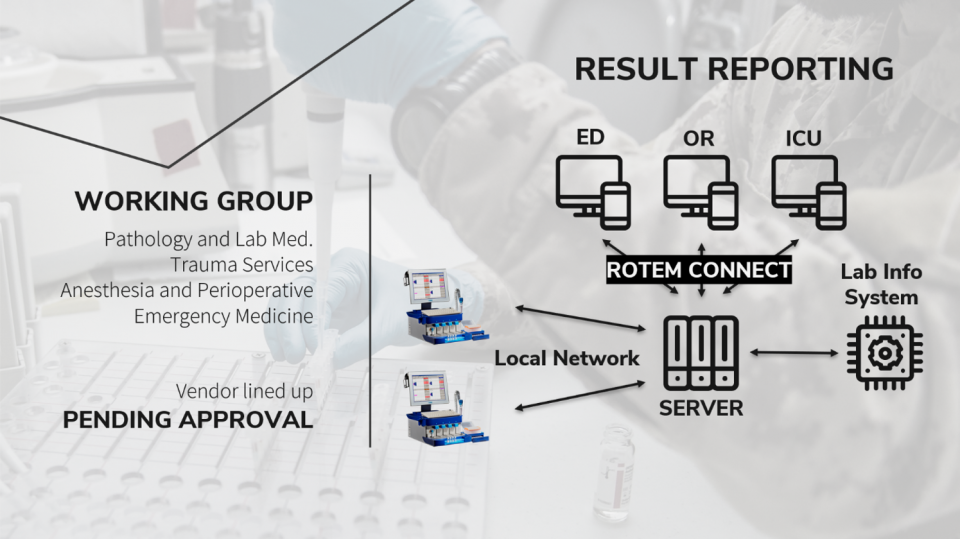
The Implementation of ROTEM
- Establishing ROTEM/TEG at any centre will require champions and a Working Group with support and leadership from Laboratory medicine, Trauma, Anesthesia, ICU, Emergency Medicine
- The centre will then have to decide if the test will be performed at the bedside or centrally:
- Point-of-care would have:
- Faster turnaround time (no transport)
- BUT:
- Multiple analyzers needed in multiple areas
- Requires dedicated staff in multiple areas (taken away from active patient care)
- No automated entry of results
- No QA/QC protocols unless oversight from lab
- Lab-based
- QA and accreditation standards maintained
- Standardized users, no need to train staff in multiple clinical areas (prevents mistakes from inappropriate use)
- Increased involvement of hematopathologists and transfusion medicine specialists
- BUT:
- Requires good communication b/w care areas and lab for fast turnaround time
- Specimen transport can take 5-10min
- Well-established/reliable transport mechanisms required
- In the several Canadian centers that utilize ROTEM, the ROTEM device is centrally located and lab-run
- Samples are taken in the various clinical care areas – which are transported by porter or pneumatic tube to the lab for rapid turnaround. These samples are processed centrally, and a live output of the test is available at any computer terminal via ROTEM Connect.
The Main Barrier to Implementation: Cost
- A consensus conference of the US JTACS stated:
- The costs of starting a program have been found to range from 100,000 to 125,000 USD. Though in the US, the ongoing costs are comparable to those of other conventional coagulation tests
- Cost-effectiveness has been demonstrated in the non-trauma setting and is in large part caused by the decrease in the consumption of blood products, which may result from improved hemostatic management
- Two systematic reviews have looked at the issue of cost-effectiveness:
- Holcomb and colleagues examined 1974 consecutive patients with trauma comparing rTEG with CCTs in 2012.
- Found that the cost of rTEG ($317) was not excessive compared with the CCTs ($286) on the basis that rTEG was available long before CCT results.
- They concluded that rTEG could replace CCTs entirely.
- Whiting et al, in 2015, looked at the clinical effectiveness and cost-effectiveness of VE devices in cardiac surgery, trauma-induced coagulopathy, and PPH.
- For the trauma population, the cost-savings owing to VE testing were more substantial, amounting to per-patient savings of £688 for ROTEM compared with SLTs, £721 for TEG, and £818 for Sonoclot. This finding was entirely dependent on material costs, which are slightly higher for ROTEM. The increased savings were due to the higher volume of blood products transfused in trauma patients. VE testing remained cost-saving following various scenario analyses.
- This conclusion is predicated on the assumption that viscoelastic testing leads to improved clinical outcomes in the trauma population, which again could not be established from existing studies, and in the absence of convincing data, the cost-savings remain more theoretical.
- The per-patient cost-savings were £43 for ROTEM, £79 for TEG, and £132 for Sonoclot, as a result of the cost of purchasing each base viscoelastic system, with the basic ROTEM system being most expensive, and Sonoclot the least expensive
- The difference in the Canadian system is that hospital’s themselves do not pay for blood products, they are provided by the Canadian Blood Services – so these cost-savings would be seen on a SYSTEMS-WIDE level and not a per hospital level.
- That being what it is, the potential benefit to the system from decreased blood product usage, the decreased transfusion risk to the patient, the ability to tailor hemostatic resuscitation, and the growing evidence of possible mortality benefit make ROTEM an attractive option.
- Holcomb and colleagues examined 1974 consecutive patients with trauma comparing rTEG with CCTs in 2012.
FOAM Resources
- https://www.emra.org/emresident/article/teg-and-rotem/
- https://litfl.com/thromboelastogram-teg/
- https://foamcast.org/2019/07/31/thromboelastography-teg-guided-resuscitation/
- http://www.emdocs.net/thromboelastography-teg-traum/
- https://rebelem.com/rebel-review/rebel-review-54-thromboelastogram-teg/thromboelastogram-teg/
- https://derangedphysiology.com/main/required-reading/haematology-and-oncology/Chapter%20120/viscoelastic-tests-clotting-function-teg-and-rotem
- https://www.acep.org/how-we-serve/sections/critical-care-medicine/news/april-2019/the-role-of-thromboelastometry-in-monitoring-critical-patients/
References
- Abdelfattah K, Cripps MW. Thromboelastography and rotational thromboelastometry use in trauma. Int J Surg. 2016;33:196–201.
- Afshari A, Wikkelsø A, Brok J, Møller AM, Wetterslev J. Thrombelastography (TEG) or thromboelastometry (ROTEM) to monitor haemotherapy versus usual care in patients with massive transfusion. Cochrane Database Syst Rev. 2011 Mar 16;(3):CD007871
- Benes J, Zatloukal J, Kletecka J. Viscoelastic Methods of Blood Clotting Assessment-A Multidisciplinary Review. Front Med (Lausanne) 2015;2:62.
- Bolliger D, Seeberger MD, Tanaka KA. Principles and practice of thromboelastography in clinical coagulation management and transfusion practice. Transfus Med Rev. 2012 Jan;26(1):1-13
- Borgman MA, Spinella PC, Perkins JG, Grathwohl KW, Repine T, Beekley AC, Sebesta J, Jenkins D, Wade CE, Holcomb JB. The ratio of blood products transfused affects mortality in patients receiving massive transfusions at a combat support hospital. J Trauma 2007;63:805–13.doi:10.1097/TA.0b013e3181271ba3
- Briggs C, Kimber S, Green L. Where are we at with point- of- care testing in haematology? Br J Haematol 2012;158:679-90. 10.1111/j.1365-2141.2012.09207.x
- Brohi K, Cohen MJ, Ganter MT, Schultz MJ, Levi M, Mackersie RC, Pittet JF. Acute coagulopathy of trauma: hypoperfusion induces systemic anticoagulation and hyperfibrinolysis. J Trauma 2008;64:1211–7. discussion 7.doi:10.1097/TA.0b013e318169cd3
- Brohi K, Singh J, Heron M, Coats T. Acute traumatic coagulopathy. J Trauma 2003;54:1127–30.doi:10.1097/01.TA.0000069184.82147.06
- Camazine MN, Hemmila MR, Leonard JC, Jacobs RA, Horst JA, Kozar RA, et al. Massive transfusion policies at trauma centers participating in the American College of Surgeons Trauma Quality Improvement Program. J Trauma Acute Care Surg. 2015;78:S48–53
- Carroll RC, Craft RM, Langdon RJ, Clanton CR, Snider CC, Wellons DD, Dakin PA, Lawson CM, Enderson BL, Kurek SJ. Early evaluation of acute traumatic coagulopathy by thrombelastography. Transl Res. 2009;154:34–9.
- Chapman MP, Moore EE, Moore HB, et al. The “Death Diamond.” J Trauma Acute Care Surg 2015;79:925-9. 10.1097/TA.0000000000000871
- Chapman MP, Moore EE, Ramos CR, et al. Fibrinolysis greater than 3% is the critical value for initiation of antifibrinolytic therapy. J Trauma Acute Care Surg. 2013;75(6):961-967; discussion 967.
- Chee YL, Greaves M. Role of coagulation testing in predicting bleeding risk. Hematol J Off J Eur Haematol Assoc 2003;4:373-8.
- Chen J, Gorman M, O’Reilly B, et al. Analytical evaluation of the epoc® point-of-care blood analysis system in cardiopulmonary bypass patients. Clin Biochem 2016;49:708-12. 10.1016/j.clinbiochem.2015.12.015
- Coakley, Margaret, et al. “Transfusion triggers in orthotopic liver transplantation: a comparison of the thromboelastometry analyzer, the thromboelastogram, and conventional coagulation tests.” Journal of cardiothoracic and vascular anesthesia 20.4 (2006): 548-553.
- Cohen MJ, Call M, Nelson M, Calfee CS, Esmon CT, Brohi K, Pittet JF. Critical role of activated protein C in early coagulopathy and later organ failure, infection and death in trauma patients. Ann Surg 2012;255:379–85.doi:10.1097/SLA.0b013e318235d9e6
- Collins PW, Solomon C, Sutor K, Crispin D, Hochleitner G, Rizoli S, et al. Theoretical modelling of fibrinogen supplementation with therapeutic plasma, cryoprecipitate, or fibrinogen concentrate. Br J Anaesth. 2014;113:585–95.
- Corredor C, Wasowicz M, Karkouti K, et al. The role of point-of-care platelet function testing in predicting postoperative bleeding following cardiac surgery: a systematic review and meta-analysis. Anaesthesia 2015;70:715-31. 10.1111/anae.13083
- Cotton BA, Faz G, Hatch QM, et al. Rapid thrombelastography delivers real-time results that predict transfusion within 1 hour of admission. J Trauma 2011;71:407-14; discussion 414-7. 10.1097/TA.0b013e31821e1bf0
- CRASH-2 Trial Collaborators. Effects of Tranexamic Acid on Death, Vascular Occlusive Events, and Blood Transfusion in Trauma Patients with Significant Haemorrhage (CRASH-2): A Randomised, Placebo-Controlled Trial. Lancet. 2010;376 (9734):23–32.
- Curry NS, Davenport R, Pavord S, Mallett SV, Kitchen D, Klein AA, Maybury H, Collins PW, and Laffan M. The use of viscoelastic haemostatic assays in the management of major bleeding: A British Society for Haematology Guideline. First published:02 August 2018 https://doi.org/10.1111/bjh.15524
- Da Luz LT, Nascimento B, Rizoli S. Thrombelastography (TEG(R)): practical considerations on its clinical use in trauma resuscitation. Scand J Trauma Resusc Emerg Med. 2013 Apr 16;21(1):29.
- Da Luz, LT, Nascimento, B, Shankarakutty, AK, Rizoli, S, Adhikari, NKJ. (2014). Effect of thromboelastography (TEG®) and rotational thromboelastometry (ROTEM®) on diagnosis of coagulopathy, transfusion guidance and mortality in trauma: descriptive systematic review. Critical Care 2014, 18:518
- Davenport R, Manson J, De’Ath H, et al. Functional definition and characterization of acute traumatic coagulopathy. Crit Care Med 2011;39:2652-8. 10.1097/CCM.0b013e3182281af5
- Delaney M, Wendel S, Bercovitz RS, et al. Transfusion reactions: prevention, diagnosis, and treatment. Lancet 2016;388:2825-36. 10.1016/S0140-6736(15)01313-6
- Deppe A-C, Weber C, Zimmermann J, et al. Point-of-care thromboelastography/ thromboelastometry-based coagulation management in cardiac surgery: a meta-analysis of 8332 patients. J Surg Res. 2016 Jun 15;203(2):424-33.
- Dias JD, Norem K, Doorneweerd DD, et al. Use of thromboelastography (TEG) for detection of new oral anticoagulants. Arch Pathol Lab Med. 2015 May;139(5):665-73.
- Dzik WH1, Blajchman MA, Fergusson D, Hameed M, Henry B, Kirkpatrick AW, Korogyi T, Logsetty S, Skeate RC, Stanworth S, MacAdams C, Muirhead B. Clinical review: Canadian National Advisory Committee on Blood and Blood Products–Massive transfusion consensus conference 2011: report of the panel. Crit Care. 2011;15(6):242. doi: 10.1186/cc10498. Epub 2011 Dec 8.
- Einersen PM, et al. Rapid thrombelastography thresholds for goal-directed resuscitation of patients at risk for massive transfusion. J Trauma Acute Care Surg. 2017;82(1):114.
- Fahrendorff M, Oliveri RS, Johansson PI. The Use of Viscoelastic Haemostatic Assays in Goal-Directed Treatment with Allogeneic Blood Products – A Systematic Review and Meta-Analysis. Scand J Trauma Resusc Emerg Med. 2017.
- Ganter MT, Hofer CK. Coagulation monitoring: current techniques and clinical use of viscoelastic point-of-care coagulation devices. Anesth Analg. 2008 May;106(5):1366-75
- Gonzalez, Eduardo, et al. “Goal-directed hemostatic resuscitation of trauma-induced coagulopathy: a pragmatic randomized clinical trial comparing a viscoelastic assay to conventional coagulation assays.” Annals of surgery 263.6 (2016): 1051.
- Goodman MD, Makley AT, Hanseman DJ, Pritts TA, Robinson BR. All the bang without the bucks: Defining essential point-of-care testing for traumatic coagulopathy. J Trauma Acute Care Surg. 2015;79(1):117-24.
- Hartert H. Blutgerinnungsstudien mit der Thrombelastographie, einem neuen Untersuchungsverfahren. Klin Wochenschr 1948;26:577-83. 10.1007/BF01697545
- Holcomb JB, Minei KM, Scerbo ML, Radwan ZA, Wade CE, Kozar RA, Gill BS, Albarado R, McNutt MK, Khan S, et al. Admission rapid thrombelastography can replace conventional coagulation tests in the emergency department: experience with 1974 consecutive trauma patients. Ann Surg 2012;256:476–86.doi:10.1097/SLA.0b013e3182658180
- Holcomb JB, Tilley BC, Baraniuk S, Fox EE, Wade CE, Podbielski JM, del Junco DJ, Brasel KJ, Bulger EM, Callcut RA, et al. Transfusion of plasma, platelets, and red blood cells in a 1:1:1 vs a 1:1:2 ratio and mortality in patients with severe trauma: the PROPPR randomized clinical trial. JAMA 2015;313:471–82.doi:10.1001/jama.2015.12
- Howley IW, Haut ER, Jacobs L, et al. Thromboelastography (TEG)-based resuscitation better than empirical 1:1 transfusion? Trauma Surgery & Acute Care Open 2018;3:e000140. doi: 10.1136/tsaco-2017-000140
- Hunt H, Stanworth S, Curry N, et al. Thromboelastography (TEG) and rotational thromboelastometry (ROTEM) for trauma-induced coagulopathy in adult trauma patients with bleeding. In: Hunt H. editor. Cochrane Database of Systematic Reviews. Chichester, UK: John Wiley & Sons, Ltd, 2015:CD010438.
- Hunt H, Stanworth S, Curry N, Woolley T, Cooper C, Ukoumunne O, Zhelev Z, Hyde C. TEG and ROTEM for diagnosing trauma induced coagulopathy (disorder of the clotting system) in adult trauma patients with bleeding. Cochcane Review. 2015
- Inaba, Kenji MD; Rizoli, Sandro MD; Veigas, Precilla V. MSc; Callum, Jeannie MD; Davenport, Ross MD; Hess, John MD; Maegele, Marc MD and the Viscoelastic Testing in Trauma Consensus Panel. 2014 Consensus conference on viscoelastic test–based transfusion guidelines for early trauma resuscitation: Report of the panel. Journal of Trauma and Acute Care Surgery: June 2015 – Volume 78 – Issue 6 – p 1220-1229. doi: 10.1097/TA.0000000000000657
- Jansen JO, Luke D, Davies E, Spencer P, Kirkman E, Midwinter MJ. Temporal changes in ROTEM®-measured coagulability of citrated blood samples from coagulopathic trauma patients. Injury. 2013;44:36–9.
- Kander T, Larsson A, Taune V, Schött U, Tynngård N (2016) Assessment of Haemostasis in Disseminated Intravascular Coagulation by Use of Point-of-Care Assays and Routine Coagulation Tests, in Critically Ill Patients; A Prospective Observational Study. PLoS ONE 11(3): e0151202. doi:10.1371/ journal.pone.0151202
- Kang YG, Martin DJ, Marquez J, et al. Intraoperative changes in blood coagulation and thrombelastographic monitoring in liver transplantation. Anesth Analg 1985;64:888-96. 10.1213/00000539-198509000-00008
- Karkouti K, Callum J, Wijeysundera DN, Rao V, Crowther M, Grocott HP, Pinto R, Scales DC; TACS Investigators. Point-of-Care Hemostatic Testing in Cardiac Surgery: A Stepped-Wedge Clustered Randomized Controlled Trial. Circulation. 2016 Oct 18;134(16):1152-1162.
- Kaufner L, Henkelmann A, von Heymann C, et al. Can prepartum thromboelastometry-derived parameters and fibrinogen levels really predict postpartum hemorrhage? J Perinat Med. 2017 May 24;45(4):427-35.
- Kornblith LZ, Kutcher ME, Redick BJ, Calfee CS, Vilardi RF, Cohen MJ. Fibrinogen and platelet contributions to clot formation: implications for trauma resuscitation and thromboprophylaxis. J Trauma Acute Care Surg. 2014;76:255–63.
- Kozek-Langenecker SA, Ahmed AB, Afshari A, Albaladejo P, Aldecoa C, Barauskas G, De Robertis E, Faraoni D, Filipescu DC, Fries D, Haas T, Jacob M, Lancé MD, Pitarch JVL, Mallett S, Meier J, Molnar ZL, Rahe-Meyer N, Samama CM, Stensballe J, Van der Linden PJF, Wikkelsø AJ, Wouters P, Wyffels P, Zacharowski K. Management of severe perioperative bleeding: guidelines from the European Society of Anesthesiology First update 2016. Eur J Anaesthesiol. 2017;34:332–395.
- Kumar M, Ahmad J, Maiwall R, et al. Thromboelastography-Guided Blood Component Use in Patients With Cirrhosis With Nonvariceal Bleeding: A Randomized Controlled Trial. Hepatology. 2019; In Press
- Lang T, Bauters A, Braun SL, et al. Multi-centre investigation on reference ranges for ROTEM thromboelastometry. Blood Coagul Fibrinolysis 2005;16:301-10. 10.1097/01.mbc.0000169225.31173.19
- Lee FM, Chan AK, Lau KK, et al. Reversal of New, Factor-specific Oral Anticoagulants by rFVIIa, Prothrombin Complex Concentrate and Activated Prothrombin Complex Concentrate: A Review of Animal and Human Studies. Thromb Res 2014;133:705-13. 10.1016/j.thromres.2014.01.031
- Leemann H, Lustenberger T, Talving P, et al. The role of rotation thromboelastometry in early prediction of massive transfusion. J Trauma 2010;69:1403-8-9.
- Levy JH, Welsby I, Goodnough LT. Fibrinogen as a therapeutic target for bleeding: a review of critical levels and replacement therapy. Transfusion. 2014;54:1389–405.
- Luddington RJ. Thrombelastography/thromboelastometry. Clin Lab Haematol 2005;27:81-90. 10.1111/j.1365-2257.2005.00681.x
- Mahamad S, Chaudhry H, Nisenbaum R, et al. Exploring the effect of factor Xa inhibitors on rotational thromboelastometry: a case series of bleeding patients. J Thromb Thrombolysis. 2019 Feb;47(2):272-9.
- Midwinter MJ , Woolley T. Resuscitation and coagulation in the severely injured trauma patient. Philos Trans R Soc Lond B Biol Sci 2011;366:192–203.doi:10.1098/rstb.2010.0220
- Morrison JJ. Noncompressible torso hemorrhage. Crit Care Clin 2017;33:37–54.doi:10.1016/j.ccc.2016.09.001
- Muszynski JA, Spinella PC, Cholette JM, et al. Transfusion-related immunomodulation: review of the literature and implications for pediatric critical illness. Transfusion 2017;57:195-206. 10.1111/trf.13855
- Nielsen, Vance G. “A comparison of the Thrombelastograph and the ROTEM.” Blood Coagulation & Fibrinolysis 18.3 (2007): 247-252
- Nystrup KB, Windeløv NA, Thomsen AB, Johansson PI. Reduced clot strength upon admission, evaluated by thrombelastography (TEG®), in trauma patients is independently associated with increased 30-day mortality. Scand J Trauma Resusc Emerg Med. 2011;19:52.
- Park MS, Salinas J, Wade CE, Wang J, Martini W, Pusateri AE, Merrill GA, Chung K, Wolf SE, Holcomb JB. Combining early coagulation and inflammatory status improves prediction of mortality in burned and nonburned trauma patients. J Trauma. 2008;64:S188–94.
- Pezold M, Moore EE, Wohlauer M, et al. Viscoelastic clot strength predicts coagulation-related mortality within 15 minutes. Surgery 2012;151:48-54. 10.1016/j.surg.2011.06.023
- Practice guidelines for perioperative blood management: an updated report by the American Society of Anesthesiologists Task Force on Perioperative Blood Management. American Society of Anesthesiologists Task Force on Perioperative Blood Management. Anesthesiology. 2015 Feb;122(2):241-75. doi: 10.1097/ALN.0000000000000463. PMID: 25545654 DOI: 10.1097/ALN.0000000000000463
- Rizoli S, Min A, Sanchez AP, Shek P, Grodecki R, Veigas P, Peng HT. In Trauma, Conventional ROTEM and TEG Results Are Not Interchangeable But Are Similar in Clinical Applicability. Mil Med. 2016 May;181(5 Suppl):117-26. doi: 10.7205/MILMED-D-15-00166.
- Rossaint R, Bouillon B, Cerny V, et al. The European guideline on management of major bleeding and coagulopathy following trauma: fourth edition. Crit Care 2016;20:100.
- Rout G, Shalimar, Gunjan D, et al. Thromboelastography-guided Blood Product Transfusion in Cirrhosis Patients With Variceal Bleeding: A Randomized Controlled Trial. J Clin Gastroenterol. 2019; In Press.
- Rugeri L, et al. Diagnosis of early coagulation abnormalities in trauma patients by rotation thrombelastography. J Thromb Haemost. 2007;5(2):289.
- Sakamoto, Y., Koami, H. & Miike, T. Monitoring the coagulation status of trauma patients with viscoelastic devices. j intensive care 5, 7 (2017).
- Sankarankutty, Ajith, et al. “TEG® and ROTEM® in trauma: similar test but different results.” World J Emerg Surg 7.Suppl 1 (2012): S3.
- Schöchl H, Cotton B, Inaba K, et al. FIBTEM provides early prediction of massive transfusion in trauma. Crit Care 2011;15:R265. 10.1186/cc10539
- Schochl H, Cotton B, Inaba K, Nienaber U, Fischer H, Voelckel W, et al. FIBTEM provides early prediction of massive transfusion in trauma. Crit Care. 2011;15:R265.
- Schöchl H, Frietsch T, Pavelka M, Jambor C. Hyperfibrinolysis after major trauma: differential diagnosis of lysis patterns and prognostic value of thromboelastometry. J Trauma. 2009;67:125–31.
- Scholchl H, Nienaber U, Hofer G, Voelckel W, Jambor C, Scharbert G, et al. Goal-directed coagulation management of major trauma patients using thromboelastometry (ROTEM®)-guided administration of fibrinogen concentrate and prothrombin complex concentrate. Crit Care. 2010;14:R55.
- Shen L, Tabaie S, Ivascu N. Viscoelastic testing inside and beyond the operating room. J Thorac Dis. 2017;9(Suppl 4):S299–S308. doi:10.21037/jtd.2017.03.85
- Smart L, Mumtaz K, Scharpf D, et al. Rotational thromboelastometry or conventional coagulation tests in liver transplantation: comparing blood loss, transfusions, and cost. Ann Hepatol. 2017 Nov-Dec;16(6):916-23.
- Snegovskikh D, Souza D, Walton Z, et al. Point-of-care viscoelastic testing improves the outcome of pregnancies complicated by severe postpartum hemorrhage. J Clin Anesth. 2018 Feb;44:50-6.
- Spahn DR, Bouillon B, Cerny V, Coats TJ, Duranteau J, Fernandez-Mondejar E, et al. Management of bleeding and coagulopathy following major trauma: an updated European guideline. Crit Care. 2013;17:R76.
- Tanaka KA, Bolliger D, Vadlamudi R, Nimmo A. Rotational thromboelastometry (ROTEM)-based coagulation management in cardiac surgery and major trauma. J Cardiothorac Vasc Anesth. 2012;26:1083–93.
- Tapia NM et al. TEG-guided resuscitation is superior to standardized MTP resuscitation in massively transfused penetrating trauma patients. J Trauma Acute Care Surg. 2013;74:378-386
- Tapia NM, Chang A, Norman M, et al. TEG-guided resuscitation is superior to standardized MTP resuscitation in massively transfused penetrating trauma patients. J Trauma Acute Care Surg. 2013 Feb;74(2):378-85; discussion 385-6.
- Tauber H, Innerhofer P, Breitkopf R, Westermann I, Beer R, El Attal R, Strasak A, Mittermayr M. Prevalence and impact of abnormal ROTEM® assays in severe blunt trauma: results of the ‘Diagnosis and Treatment of Trauma-Induced Coagulopathy (DIA-TRE-TIC) study’. Br J Anaesth. 2011;107:378–87.
- Thakur M, Ahmed AB. A review of thromboelastography. Int J Periop Ultrasound Appl Technol 2012;1(1):25-29.
- Trentalange MJ, Walts LF. A comparison of thromboelastogram and template bleeding time in the evaluation of platelet function after aspirin ingestion. J Clin Anesth. 1991 Sep-Oct;3(5):377-81.
- Venema, Lieneke F., et al. “An assessment of clinical interchangeability of TEG® and ROTEM® thromboelastographic variables in cardiac surgical patients.” Anesthesia & Analgesia 111.2 (2010): 339-344.
- Whiting D, DiNardo JA. 2014. TEG and ROTEM: Technology and Clinical Applications. Am J Hematol. 2014;89(2):228–232.
- Whiting P, Al M, Westwood M, et al. Viscoelastic Point-of-Care Testing to Assist with the Diagnosis, Management and Monitoring of Haemostasis: A Systematic Review and Cost-Effectiveness Analysis. Health Technol Assess. 2015;19(58)1-228.
- Wikkelsø A, Wetterslev J, Møller AM, Afshari A. Thromboelastography (TEG) or Rotational Thromboelastometry (ROTEM) to Monitor Haemostatic Treatment in Bleeding Patients: A Systematic Review with Meta-Analysis and Trial Sequential Analysis. Anaesthesia. 2017;72(4):519–531
- Wikkelsø A, Wetterslev J, Møller AM, Afshari A. Thromboelastography (TEG) or thromboelastometry (ROTEM) to monitor haemostatic treatment versus usual care in adults or children with bleeding. Cochrane Database Syst Rev. 2016;(8):CD007871
- Wikkelsø A, Wetterslev J, Møller AM, et al. Thromboelastography (TEG) or thromboelastometry (ROTEM) to monitor haemostatic treatment versus usual care in adults or children with bleeding. In: Afshari A. editor. Cochrane Database of Systematic Reviews. Chichester, UK: John Wiley & Sons, Ltd, 2016:CD007871.
- Yin J, Zhao Z, Li Y, Wang J, Yao D, Zhang S, et al. Goal-directed transfusion protocol via thrombelastography in patients with abdominal trauma: a retrospective study. World J Emerg Surg. 2014;9:28.



Hi there, this is an excellent resource. Thank you very much for such a comprehensive review.
I was interested in the Vancouver Coastal Health algorithm and was wondering if you could help put me find the original article? Thank you in advance!
http://www.phsa.ca/health-professionals-site/Documents/CPG_Massive%20Hemorrhage_Algorithm%20removed_2023-09-12.pdf