This comprehensive, 2-part blog post by Dr. Hashim Kareemi reviews the current literature and guidelines on elevated ICP in the emergeny department. Part 1 reviews the pathophysiology and detrimental effects of sustained elevated ICP, and provides an excellent comparison of the options for diagnosing elevated ICP in the ED.
Background
- Elevated intracranial pressure or ICP is associated with mortality (1-3) and poor neurologic outcomes (4,5).
- Our ability to quickly recognize and treat elevated ICP can influence patient outcomes.
-
- Traumatic brain injury (TBI) patients with fewer hours of elevated ICP in the first day after injury had better neurologic outcomes (6).
- Elevated ICP was identified as one of only two treatable independent risk factors for mortality in TBI patients, along with systemic hypotension (7).
Pathophysiology of Elevated ICP
Central nervous system neuron perfusion depends on maintenance of cerebral blood flow, which is defined by the equation:
Autoregulation: the response in cerebrovascular resistance to physiologic changes (e.g. systolic blood pressure, respiratory status, physical posture) in order to maintain cerebral blood flow (8).
Two important autoregulatory responses include:
- ↓ MAP = vasodilation
- ↑ PaCO2 = vasodilation
Cerebral blood flow is closely related to the cerebral perfusion pressure, which is defined as:
Intracranial pressure (ICP) is an important component of cerebral perfusion pressure. It is defined as the pressure exerted by fluids inside the cranium. Normal adult ICP is 7-15 mmHg.
- Blood:
- Arterial
- Venous
- Cerebrospinal fluid
- Brain parenchyma (mostly water)
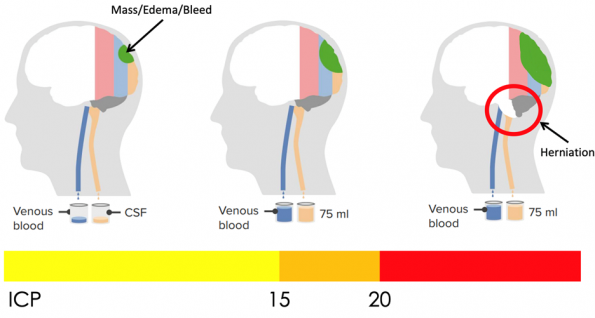
Figure 3: Relationship of relative intracranial volume and intracranial pressure. Adapted from: https://www.lecturio.com/magazine/increased-intracranial-pressure/
In the neurologically injured brain, we add an extra component such as a hematoma or cerebral edema. The Monro-Kellie Doctrine states that because the cranium is a fixed inelastic container, the increase in volume of one component must be compensated by the decrease in volume of another component, which proceeds as follows (8):
- Venous blood, then CSF are shunted out first to compensate and maintain normal ICP
- With a large enough mass, or with reduced capacity to drain CSF or venous blood, this equilibrium is lost and instead ICP begins to rise
- With sustained elevated ICP, brain parenchyma itself may be forced through anatomic openings in the cranium, compressing critical structures such as the brainstem, cerebral arteries, and nerves. This is called herniation.
In addition to herniation, sustained elevated ICP is decrease in cerebral perfusion pressure secondary to decreased cerebral perfusion pressure.
In a healthy brain, cerebral blood flow is directly correlated to the cerebral perfusion pressure except for a large intermediate zone between about 50-150 mm Hg in which cerebral blood flow remains normal (8). This is the normalizing effect of autoregulation.
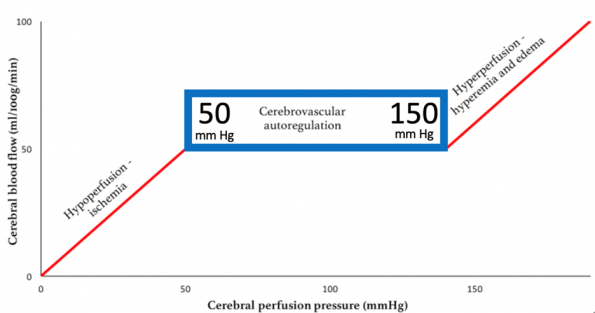
Figure 4: Cerebral blood flow versus cerebral perfusion pressure in the normal brain, highlighting zone of autoregulation. (Adapted from: Harary 2018)
In the neurologically injured brain, the physiologic response is to increase MAP (usually by bolstering cardiac output) in an attempt to bring cerebral perfusion pressure back into the range in which cerebral vessels can autoregulate, thereby maintaining cerebral blood flow.
However, neurologic injury can independently disrupt autoregulation (9). Therefore, cerebral blood flow becomes more directly correlated to cerebral perfusion pressure, and acute rises in ICP can quickly result in cerebral hypoperfusion and ischemia.
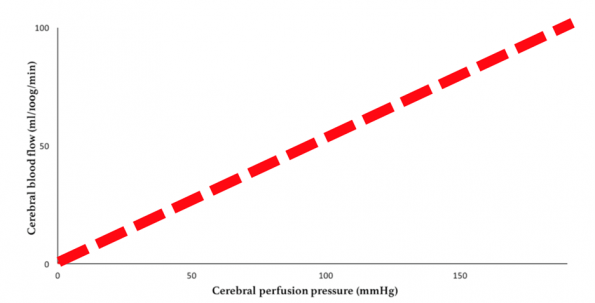
Figure 5: Cerebral blood flow versus cerebral perfusion pressure in the neurologically injured brain.
“Brain codes” represent these deleterious effects of sustained elevated ICP. Similar to cardiopulmonary conditions such as tension pneumothorax or cardiac tamponade, brain codes must be treated by relieving the obstructing entity (i.e. decrease ICP) while ensuring adequate perfusion (i.e. maintain MAP). Brain codes can be thought of as obstructive shock of the CNS.
Etiologies of Elevated ICP
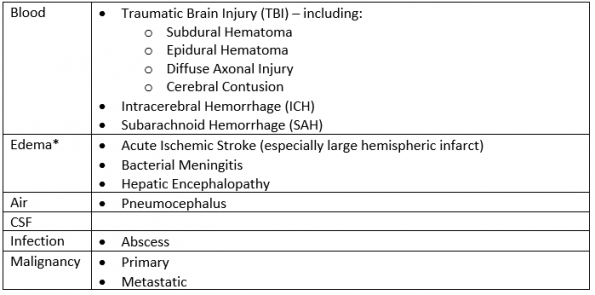
*Cerebral edema may play a role in several of the other listed etiologies (10). Table 1: Etiologies of elevated intracranial pressure.
Diagnosis of Elevated ICP
Gold Standard: invasive ICP monitor (e.g. extra-ventricular drain, bolt monitor) – often not available in initial phase of care.
History: limited evidence, often patients unable to provide history as they have altered mental status. Classic symptoms include headache, vomiting and visual changes.
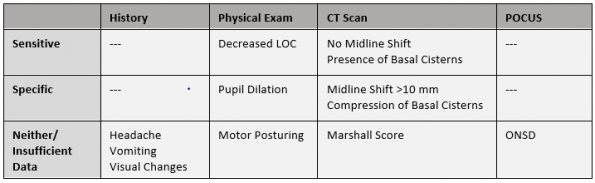
Physical Exam and Imaging: a recent meta-analysis by Fernando et al. examined the accuracy of different non-invasive (physical exam, CT or MRI) methods compared to invasive ICP monitor for adult patients with suspected elevated ICP (11). They found that no single non-invasive finding was diagnostic in isolation, whether to rule in or rule out elevated ICP. However, the following signs have diagnostic value:
- Any pupillary dilation is highly specific
- Absence or compression of basal cisterns on CT is highly sensitive
- Significant midline shift of >10 mm on CT is highly specific
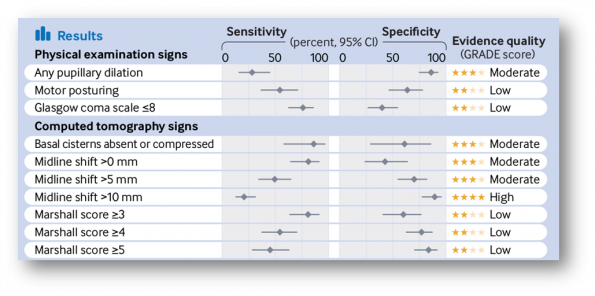
Table 2: Sensitivity and specificity of non-invasive methods for diagnosis of elevated intracranial pressure.
(Copied from: Visual Abstract, Fernando 2019)
Ultrasound: several bedside ultrasound techniques have gained popularity for diagnosing elevated ICP.
- Transcranial doppler, which involves measuring blood flow velocity in cerebral vessels via bone windows (e.g. temporal) in the cranium, was shown by Fernando et al. to have limited area under the curve (AUC 0.55-0.72 for pulsatility index, AUC 0.85 for arterial blood pressure), and requires further investigation before clinical application (11).
- Optic nerve sheath diameter (ONSD), measured 3 mm behind the globe via ocular ultrasound, had a better AUC of 0.94 among 10 studies included in Fernando et al’s meta-analysis. However, the constituent studies used a variety of cut-offs for the diameter.
- Another meta-analysis similarly found high sensitivity and specificity (12). However, they did not limit comparison to the gold standard of invasive ICP monitor. These results should therefore be interpreted with caution.
- Taking these two meta-analyses together, and without consensus on an ideal cut-off nor a large, prospective trial with a gold standard comparator, ONSD cannot be recommended for the diagnosis of elevated ICP at this time.
Summary
In this post, Dr. Kareemi described the pathophysiology of elevated ICP according to the Monro-Kellie Doctrine, and explained the deletirious effects or ‘brain codes’ of sustained elevated ICP that include cerebral hypoperfusion and ischemia. This post also reviewed and compared available diagnostic options: invasive ICP monitoring (gold standard), CT head findings, physical exam and pupillary dilatation, transcranial doppler and optic nerve sheath diameter. The takeaway: diagnosing elevated ICP requires multiple non-invasive signs, and we should not necessarily wait for the CT to initiate treatment. Part 2 will cover the guidelines and approach to management of elevated ICP, stay tuned!
References
- Lannoo E, Van Rietvelde F, Colardyn F, Lemmerling M, Vandekerckhove T, Jannes C, De Soete G. Early predictors of mortality and morbidity after severe closed head injury. J Neurotrauma. 2000 May;17(5):403-14. doi: 10.1089/neu.2000.17.403. PMID: 10833059.
- Narayan RK, Greenberg RP, Miller JD, Enas GG, Choi SC, Kishore PR, Selhorst JB, Lutz HA 3rd, Becker DP. Improved confidence of outcome prediction in severe head injury. A comparative analysis of the clinical examination, multimodality evoked potentials, CT scanning, and intracranial pressure. J Neurosurg. 1981 Jun;54(6):751-62. doi: 10.3171/jns.1981.54.6.0751. PMID: 7241184.
- Lindvall P, Ahlm C, Ericsson M, Gothefors L, Naredi S, Koskinen LO. Reducing intracranial pressure may increase survival among patients with bacterial meningitis. Clin Infect Dis. 2004 Feb 1;38(3):384-90. doi: 10.1086/380970. Epub 2004 Jan 14. PMID: 14727209.
- Czosnyka M, Guazzo E, Whitehouse M, Smielewski P, Czosnyka Z, Kirkpatrick P, Piechnik S, Pickard JD. Significance of intracranial pressure waveform analysis after head injury. Acta Neurochir (Wien). 1996;138(5):531-41; discussion 541-2. doi: 10.1007/BF01411173. PMID: 8800328.
- Sorrentino E, Diedler J, Kasprowicz M, Budohoski KP, Haubrich C, Smielewski P, Outtrim JG, Manktelow A, Hutchinson PJ, Pickard JD, Menon DK, Czosnyka M. Critical thresholds for cerebrovascular reactivity after traumatic brain injury. Neurocrit Care. 2012 Apr;16(2):258-66. doi: 10.1007/s12028-011-9630-8. PMID: 21964774.
- Farahvar A, Gerber LM, Chiu YL, Carney N, Härtl R, Ghajar J. Increased mortality in patients with severe traumatic brain injury treated without intracranial pressure monitoring. J Neurosurg. 2012 Oct;117(4):729-34. doi: 10.3171/2012.7.JNS111816. Epub 2012 Aug 17. PMID: 22900846.
- Schreiber MA, Aoki N, Scott BG, Beck JR. Determinants of Mortality in Patients With Severe Blunt Head Injury. Arch Surg. 2002;137(3):285–290. doi:10.1001/archsurg.137.3.285
- Harary M, Dolmans RGF, Gormley WB. Intracranial Pressure Monitoring-Review and Avenues for Development. Sensors (Basel). 2018 Feb 5;18(2):465. doi: 10.3390/s18020465. PMID: 29401746; PMCID: PMC5855101.
- Rangel-Castilla L, Gasco J, Nauta HJ, Okonkwo DO, Robertson CS. Cerebral pressure autoregulation in traumatic brain injury. Neurosurg Focus. 2008 Oct;25(4):E7. doi: 10.3171/FOC.2008.25.10.E7. PMID: 18828705.
- Hinson HE, Stein D, Sheth KN. Hypertonic saline and mannitol therapy in critical care neurology. J Intensive Care Med. 2013 Jan-Feb;28(1):3-11. doi: 10.1177/0885066611400688. Epub 2011 Mar 24. PMID: 21436162.
- Fernando SM, Tran A, Cheng W, Rochwerg B, Taljaard M, Kyeremanteng K, English SW, Sekhon MS, Griesdale DEG, Dowlatshahi D, McCredie VA, Wijdicks EFM, Almenawer SA, Inaba K, Rajajee V, Perry JJ. Diagnosis of elevated intracranial pressure in critically ill adults: systematic review and meta-analysis. BMJ. 2019 Jul 24;366:l4225. doi: 10.1136/bmj.l4225. PMID: 31340932; PMCID: PMC6651068.
- Koziarz A, Sne N, Kegel F, Nath S, Badhiwala JH, Nassiri F, Mansouri A, Yang K, Zhou Q, Rice T, Faidi S, Passos E, Healey A, Banfield L, Mensour M, Kirkpatrick AW, Nassar A, Fehlings MG, Hawryluk GWJ, Almenawer SA. Bedside Optic Nerve Ultrasonography for Diagnosing Increased Intracranial Pressure: A Systematic Review and Meta-analysis. Ann Intern Med. 2019 Dec 17;171(12):896-905. doi: 10.7326/M19-0812. Epub 2019 Nov 19. PMID: 31739316.