
When encountering patients with angioedema, we have historically used a similar approach to anaphylaxis, mostly out of a lack of evidence supporting a particular practice. Here, we take a deep dive into ACE inhibitor-induced angioedema to provide the most up to date evidence and approach!
Angioedema
It is a non-pitting swelling of the subcutaneous or submucosal tissue of the skin, oropharynx, upper respiratory tract, and/ or gastrointestinal (GI) tract.1
How does angioedema occur?
Angioedema can be divided into two main mechanisms that are mast cell-mediated, and bradykinin-mediated.
Mast cell-mediated angioedema:2,3
After initial contact with an allergen, a person becomes sensitized to that allergen. On the next exposure to the same allergen, an IGE-mediated degranulation of mast cells occurs resulting in the release of histamine. This release of histamine triggers a cascade that causes leaky, dilated vascular endothelium leading to angioedema.
Bradykinin-mediated angioedema:3,4
-
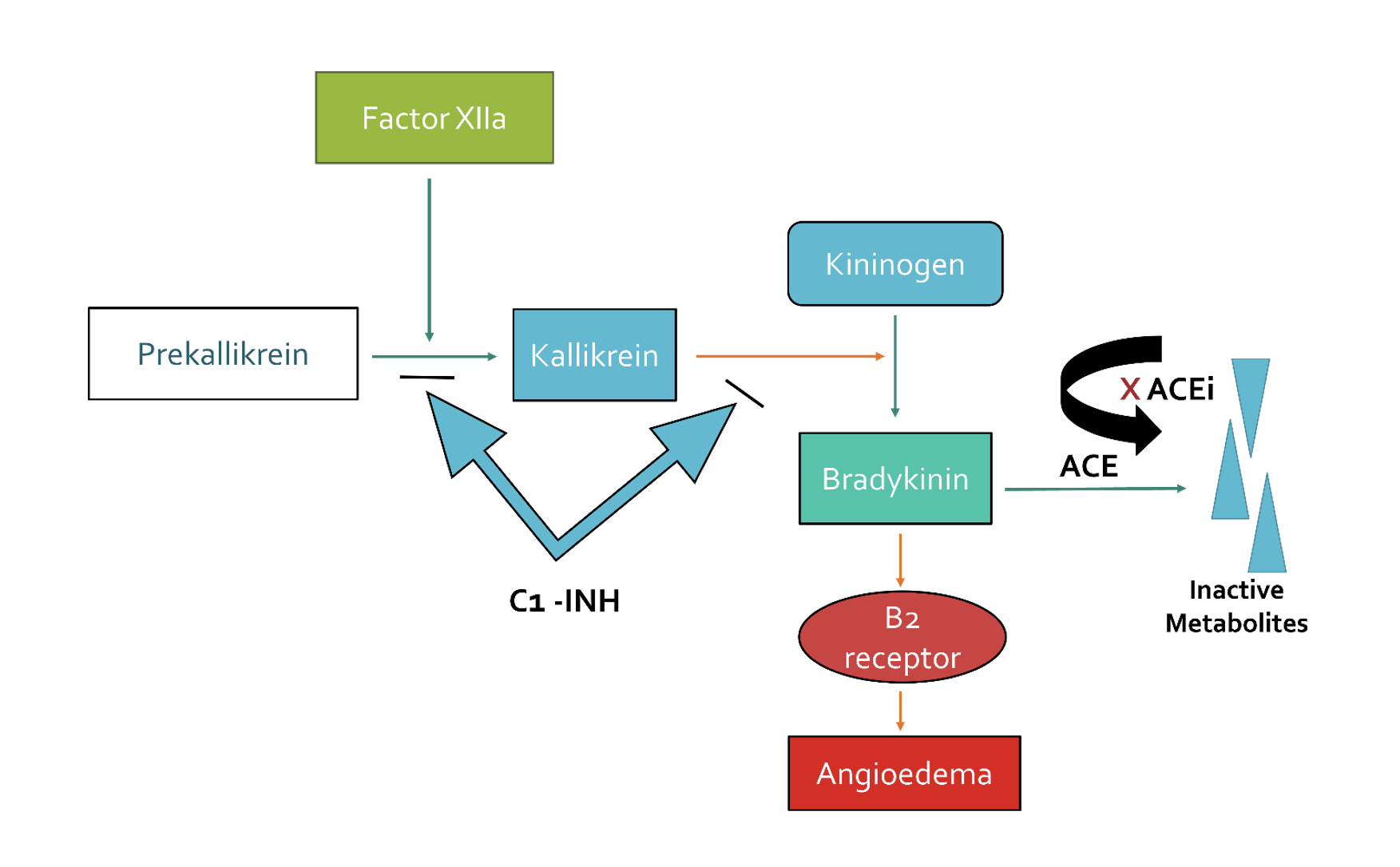
- ACEi-induced angioedema although not fully understood is a bradykinin-mediated process. In the Kinin–Kallikrein system, factor XII converts Prekallikrein to Kallikrein and subsequently Kallikrein converts Kininogen to Bradykinin (bradykinin is a vasodilator).
- Bradykinin then stimulates the B2 receptors in the endothelium leading to increase vascular permeability, manifesting as angioedema.
- Angiotensin-converting enzyme (ACE) is involved in the process of bradykinin breakdown into inactive metabolites preventing vascular permeability/ angioedema. Thus, Angiotensin-converting enzyme inhibitor (ACEi) inactivates ACE which in turn blocks the conversion of bradykinin into inactive metabolites, leading to the accumulation of bradykinin and hence angioedema occurs.
- C1 inhibitor protein is also an inhibitory protein in this pathway that breaks down bradykinin into inactive metabolites just like ACE. If C1 inhibitor protein is deficient, bradykinin will accumulate resulting in episodes of swelling (hereditary angioedema).
Why is ACE inhibitor-induced angioedema important?
-
- ACE inhibitors are among the most commonly prescribed drugs.
- ACE inhibitor-induced angioedema accounts for 30% of all angioedema presentations.5,6
- A retrospective cohort study with over 130,000 patients showed that 0.7% of patients taking an ACEi will develop bradykinin-mediated angioedema in the first 5 years.7
- Can occur from hours after initiation of an ACEi to years after with 10% occurring in the first month.8,9
- Although historically ARBs were thought to cause bradykinin-mediated angioedema, recent evidence including a meta analysis suggests that the rates of angioedema with ARBs use is equivalent to placebo.10,11
Differentiating ACE inhibitor-induced angioedema (bradykinin-mediated) from mast cell-mediated angioedema
Besides being on an ACEi, other clues of bradykinin-mediated angioedema in general include:
-
-
- The absence of allergen exposure, onset over hours, more involvement of the tongue and upper airways, and the absence of urticaria and pruritis.
- Both can have GI involvement.
- Mast cell-mediated tends to have more involvement of other systems including hypotension, shock and wheezes.1,2,12
-
Investigations
There are numerous blood tests that can be ordered, but unfortunately there is no diagnostic blood test that confirms ACE inhibitor-induced angioedema.1,2
Potential blood tests to consider are C4 and C1 esterase inhibitor levels, ESR/ CRP and serum tryptase. You should not worry about these tests in the emergency department, but these tests may help allergists later on their assessment later. It is reasonable to get basic blood work including CBC, basic chemistry and renal function test for the non-immunologic differential of angioedema, although this is unlikely to help diagnostically.
Treatments
- Stopping the ACEi is the number one thing that can be done to treat ACE inhibitor-induced angioedema. This will lead to the resolution of symptoms in 24 to 72 hours in the majority of patients.15
- There are numerous medications proposed in the literature to treat ACE inhibitor-induced angioedema which include:13,14
-
-
- The use of standard mast cell-mediated angioedema treatment including steroid, antihistamines and epinephrine.
- Fresh frozen plasma (FFP).
- Tranexamic Acid (TXA).
- Icatibant.
- Ecallantide.
- C1 inhibitor concentrate.
-
-
1. Corticosteroids, antihistamines and epinephrine
Historically, patients presenting to the ED with ACE inhibitor-induced angioedema have been given “trial” doses of these medications.16,17 We now have a better understanding of the Kinin–kallikrein system and all the evidence shows no benefit of these medications in the treatment of ACE inhibitor-induced angioedema.
The American Academy of Emergency Medicine (AAEM) released a 2021 Clinical Practice Committee statement (CPC) on the treatment of ACE inhibitor-induced angioedema that recommends against the routine use of these medications.1
That being said, if the diagnosis is unclear or you suspect mast cell-mediated angioedema, consider giving these medications.
2. FFP
The standard dose is 2 units.
FFP contains ACE, hence its proposed mechanism in ACE inhibitor-induced angioedema is the replacement of ACE which can subsequently break down Bradykinin to inactive metabolites. Nevertheless, what muddies the waters with FFP is that the level of ACE can vary significantly between bags of FFP potentially due to donors taking ACEi.18 In addition, FFP also contains kininogen and kallikrein which are substrates in the production of bradykinin that theoretically may worsen angioedema, although no study has demonstrated this.5
There are numerous case reports about the success of FFP in the treatment of angioedema including a 2013 case series on 7 cases showing “temporal association between the administration of FFP and improvement in ACE inhibitor-induced angioedema”.17,19–21 The highest level of evidence that we have on FFP is a retrospective cohort study done by Saeb and colleagues in 2016. This study included all adults admitted to the ICU with angioedema in a community teaching hospital in Detroit MI between 2007-2012.
-
-
- The study excluded age less than 18 years, food allergy or hereditary angioedema as the culprit, and intubation unrelated to airway angioedema.
- 128 charts met the inclusion criteria and were reviewed.
- ACEi were used in 90% of the FFP-treated group and 85% of the control group.
- They found that patients treated with FFP had lower rates of intubation and shorter ICU stays.
- Intubation done in 35% of the FFP-treated group and 60% of control group (p=0.05).
- FFP-treated patients had shorter ICU stays (1.5 days versus 3.5 days; p < 0.001).
-
That said, this was a small cohort study with only 20 patients in the FFP group. Potentially patients receiving FFP were not as sick, and FFP was given as an attempt to avoid an intubation that they would have never needed.
On the other hand, there has been some contradictory evidence in recent years with some negative case reports published on the use of FFP in treating ACE inhibitor-induced angioedema. A case published in 2017 reports a patient with ACE inhibitor-induced angioedema whose symptoms got worse 2 hours after the administration of 2 units of FFP.22 This potentially signal that previous unanimously positive case reports on the use of FFP in treating ACE inhibitor-induced angioedema may be the result of reporting bias.
In summary, there are a lot of case reports and a small retrospective cohort study, but we are not sure if FFP works as well as FFP can have variable levels of ACE enzyme between bags of FFP. FFP also carries a risk associated with blood administration and 2 units of FFP may not be tolerated by patients with chronic kidney disease and/ or congestive heart failure who disproportionately use ACE inhibitor.
3. Tranexamic Acid (TXA)
The standard dose is 1 g.
It works by blocking the conversion of plasminogen to plasmin leading to decreased activation of factor XII which is needed for bradykinin production. This makes theoretical sense, but a counter argument is that the proposed issue with ACE inhibitor-induced angioedema is not overproduction but instead too much existing bradykinin.
Similar to FFP, there are numerous case reports on the effective use of TXA in treating ACE inhibitor-induced angioedema.23,24 There are 2 main studies that offer the highest level of evidence. The first is a 2018 study done by Beauchence et al in 2018 that was a retrospective analysis of medical files of patient who had consults between 2010 – 2016 in 2 French tertiary care hospitals for ACE inhibitor-induced angioedema.25
-
-
- All had received TXA as a first line treatment.
- 33 patients in total who had severe episode of angioedema.
- 27 significantly improved when treated with TXA alone.
- 6 remaining patients were treated with alternate therapy with icatibant (5/33) or C1 inhibitor concentrate (1/33) due to partial improvement after tranexamic acid therapy.
- None of the patients were intubated, no fatalities and no side effects were reported.
-
The second study was done in 2021 by Hasara et al which was a single site retrospective cohort study in Lakeland USA.26
-
-
- Included 16 patients treated with TXA in the ED for suspected ACE inhibitor-induced angioedema from Jan 2019 – March 2021.
- Primary outcome was proportion of patients who required intubation.
- All but one received one histamine directed therapy.
- 15 received 1000 mg, but 1 received 100 mg.
- 4 received FFP and TXA.
- Mean time from TXA administration to resolution was 100 min SD 26.
- 1 required repeat TXA dose.
- 2 were intubated prior to administration of TXA.
- The remaining 14 patients did not require intubation.
- No adverse effects reported.
-
So, what is the verdict on TXA use now? While there are no randomized control trials, the best evidence does show some signal of potential for benefit in terms of symptom improvement and decreasing the need for intubation with no evidence of harm.
4. Icatibant
A synthetic peptidomimetic drug that works as a competitive antagonist to the bradykinin B2 receptor located on the endothelial cells.27 Initial randomized control trials in 2015 had positive results for the use of icatibant for improving symptoms and even preventing intubation.28,29 However, 2 larger trials done in 2017 showed negative results. The first trial by Straka et al was a single center double-blind, placebo-controlled study protocol involved 33 patients where 13 were in the icatibant group and 18 in the placebo. They found no statistical difference in time to symptom resolution.27 The second study by Sinert et al was an international multicenter double blinded RCT that included Canadian sites where 121 patients underwent randomization. 60/121 received Icatibant (1 withdrew consent) and 58/121 received placebo. There were no statistically significant differences between the 2 treatment groups in the primary efficacy end point of time to meeting discharge criteria (p= 0.63), and no statistical difference in time of symptoms resolution (P= 0.57) or any other secondary end point.30 In Canada, Cai et all 2020 performed a retrospective review of the Winnipeg Regional Health Authority’s (WRHA) angioedema patients who were dispensed icatibant in hospital. This study involved a total of 21 patients where half received it for the treatment of ACE inhibitor-induced angioedema. They found no benefit for Icatibant use in the treatment of ACE inhibitor-induced angioedema.31
In summary, there is strong evidence against the use of Icatibant, suggesting there is more to ACE inhibitor-induced angioedema than simply an excess of bradykinin.
5. Ecallantide
It inhibits the conversion of high molecular weight kininogen to bradykinin by inhibiting plasma kallikrein.32 There are case reports that did show promising results and specifically 2 main studies that investigated the effectiveness of Ecallantide in ACE inhibitor-induced angioedema. The first study was done by Bernstein et al 2015 and was a phase 2 triple blinded RCT. The primary efficacy end point of the study was achieving discharge criteria from the ED within 4 hours after initiating study-related treatment. 50 patients enrolled (86% had moderate or severe severity) and divided to 24 patients in the placebo group and 26 in the treatment (Ecallantide) group. Discharge criteria from the ED was met in 4 hours or less in 8 patients (31%) of 26 patients receiving Ecallantide Vs. 5 patients of (21%) 24 patients receiving placebo (difference in proportions 10%; 95% confidence interval -14% to 34%) with no adverse reactions reported.33 Another multicenter double blinded RCT done by Lewis et all 2015 with 79 patients were randomized. 58 patients were randomized to the Ecallantide group (10 mg n=20, 30 mg n=19, 60 mg n=19) and 18 were in the placebo. The primary endpoint of the study was defined as meeting predetermined discharge eligibility criteria within 6 hours of study drug administration. The interim analysis revealed a substantially higher-than-anticipated response rate in the placebo group and because of this, the study was determined to be inadequately powered to detect a statistically significant difference between the 2 groups, and the trial was stopped early.32
Bottom line, the evidence is clear that Ecallantide is not useful in ACE inhibitor-induced angioedema.
6. C1 esterase Inhibitor Concentrate
It acts by inhibiting the kallikrein kininogen system. There have been numerous case reports of its use in improving ACE inhibitor-induced angioedema.34–37 Greve et al 2015 did a proof-of-concept case series with historical control of ACE inhibitor-induced angioedema patients. 10 patients in the C1 esterase inhibitor group and 47 in the corticosteroid/ antihistamine group and time to complete resolution of symptoms was considerably longer in the historical control group (33.1 +/- 19.4 hours) than in the C1 esterase inhibitor group (10.1 +/-3.0 hours). No intubation or tracheotomy was needed in the C1 esterase inhibitor group (0/10 patients), whereas three out of the 47 historical controls required tracheotomy and two were intubated (5/47).38 A more recent 2021 retrospective propensity-matched cohort study by Mohoron et al compared patients who received C1 esterase inhibitor for ACE inhibitor-induced angioedema to those who did not. 82 patients were included with 57 patients in non-C1 esterase inhibitor group and 25 patients in the C1 esterase inhibitor group. After propensity score matching; 22 patients in the C1 esterase inhibitor group and 22 in the non-C1 esterase inhibitor group. The study showed no difference in proportion of intubation (13.6% in the C1 esterase inhibitor group vs. 9.1% in the non-C1 esterase inhibitor group, p >0.999). Mean cost of angioedema related medication was also higher in the C1 esterase inhibitor group ($8758.95 (± $2959.30) vs. $15.91 (± $7.32), p <0.001).39 Although not a RCT, this study hints at what has become a common theme.
Simply put, there is not sufficient evidence to justify the use of C1-esterase inhibitor concentrate in ACE inhibitor-induced angioedema.
The single best treatment for ACE inhibitor-induced angioedema is the discontinuation of the ACE inhibitor
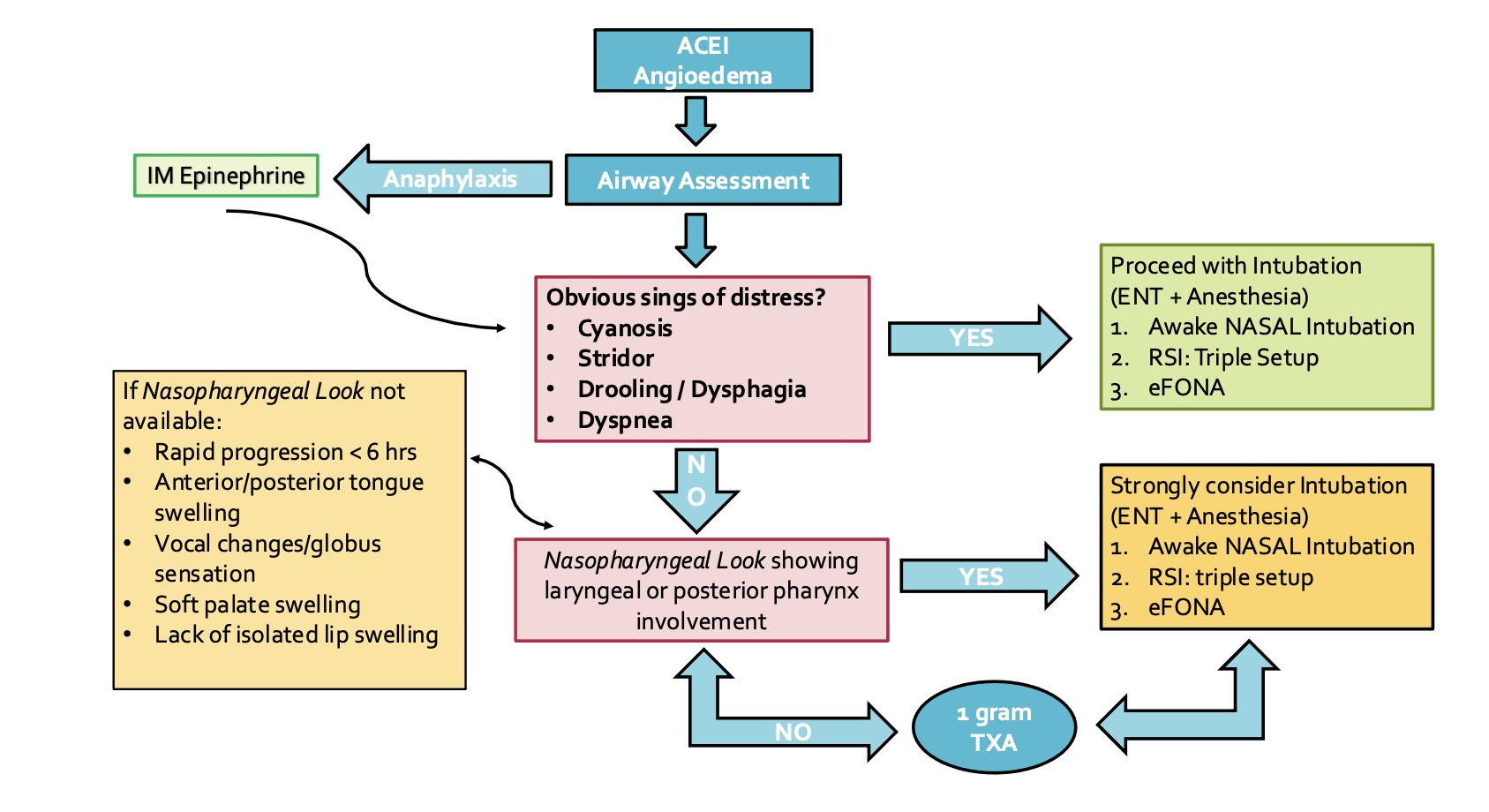
Airway management of ACE inhibitor-induced angioedema
We can think about the airway management in 2 steps. The first step is the assessment followed by the second step that is intubation.
Step 1: Assessment
The question is who needs a tube and what can we use to predict that?
-
Based on symptoms and signs of distress:
Mudd et all in 2020 did a retrospective study with 183 patients presenting to their Emergency Department with ACE inhibitor-induced angioedema of whom 9.5% needed intubation. Those needing intubation were more likely to have
1. Rapid progression of symptoms within the first 6 hours
2. Anterior tongue swelling
3. Vocal changes
4. Drooling and dyspnea. Isolated lip swelling was significantly more common in patients that do not require intubation.44 Other signs that may predict intubation are can’t swallow, inspiratory stridor or cyanosis.45
-
Nasopharyngeal look
Another tool to use in the initial assessment of these patients is the nasopharyngeal look. AAEM recommends this as a first line in the airway assessment in patients who do not need immediate airway management.1 A recent retrospective study by Gayen et al 2022 supports this too. They found that the nasopharyngeal look was normal in angioedema patients who did not require intubation.46
Locally, you can use the nasopharyngeal scope, or the new portable bronchoscope inserted in the nose. Look for laryngeal edema +/- posterior pharynx involvement. In their retrospective study, Chant et al 2014 found 65% of patients with laryngeal edema needed airway intervention.47 If laryngeal and pharyngeal edema are present, then immediate intervention is needed.48 The presence of epiglottic, aryepiglottic, or laryngeal edema suggests the need for definitive airway management.5
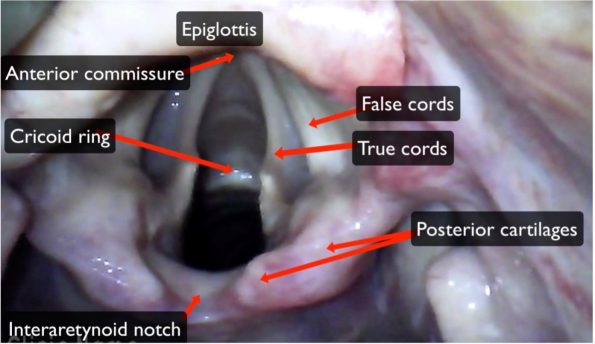
Normal Anatomy49
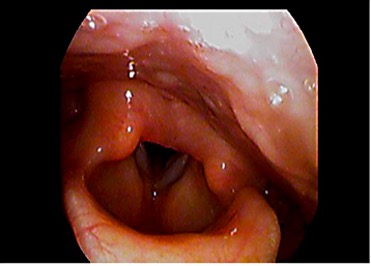
Laryngeal Edema50
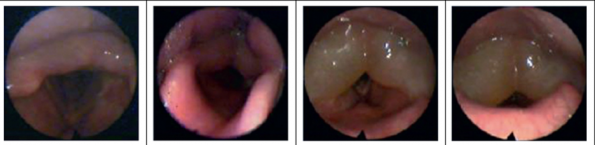
Spectrum of Laryngeal Edema51
Step 2: Intubation
Expert opinion
This past month I had the opportunity to do the AIME difficult airway month elective as well as complete the AIME course. I was able to discuss the airway management of ACE inhibitor-induced angioedema with Dr. Parks who is an AIME instructor and Ottawa emergency alumni. Although dynamic, there may be time “don’t be a cowboy/cowgirl/cowperson. Acknowledge that likely the safest place for this patient to be intubated is in the OR with ENT present to perform an awake trach PRN.”
That said, based on center, consultant availability and/ or rapidly progressive pathology, there may be a need for ED intubation. In these cases in the ED, the primary first line should be awake nasotracheal intubation and it should be done by a provider comfortable with the procedure as manipulation of airway can lead to worsening angioedema due to release of factor XII.1
Want to know more about the nasal awake fiberoptic intubation? The AIME textbook chapter on this topic will provide you with photos and videos on how to perform it.
In terms of preparation, the extended airway exam with a nasopharyngeal scope is a must. Mark the neck of all these patients. If there is difficult anatomy, as per the DAS guidelines “if you can’t confidently identify anterior neck anatomy, your initial vertical incision through skin should be a large one (8-10 cm), to help you then identify anatomy using blunt dissection”.52
After discussing this with Dr. Parks, here is the approach we should take:
The first step is deciding if the patient is maintaining airway/ventilation or if this will be a crash intubation.
- Maintaining airway/ ventilation intubation:
- Double set up
- Neck marked and prepped +/- local with epi.
- Exit strategies available with RSI drugs, BMV, VL and SGA.
- Use smaller tube 7.0 (tight anatomy)
- Smaller than size 7.0 will be shorter, so will need nasal ETT to reach glottic inlet (especially in taller people).
- Double set up
- Crash intubation: (my approach)
-
- Optimize oxygenation as best you can (use two sources high FiO2).
- PEEP may help (BVM, BiPAP).
- RSI with “triple setup” (i.e. flexible scope, VL, eFONA)
- One attempt with oral VL (to lift tongue) and nasal flexible scope (with nasal 7.0 ETT) used to find glottis.
- If unsuccessful, come out and reoxygenate with iGel.
- If you’re successful, intubate through the iGel with the flexible scope and 6.0 ETT
- If you’re unsuccessful with this, or if at any point you reach significant desaturation, go with eFONA.
Summary of the Management in the ED
References
- Rosenbaum S, Gentry Wilkerson R, Winters ME, Vilke GM, Yung M, Wu C. Clinical Practice Statement: What is the Emergency Department Management of Patients with Angioedema Secondary to an ACE-Inhibitor? J Emerg Med [Internet]. 2021 [cited 2022 Oct 18];61(1):105–12. Available from: https://doi.org/10.1016/j.jemermed.2021.02.038
- Bernstein JA, Cremonesi P, Hoffmann TK, Hollingsworth J. Angioedema in the emergency department: a practical guide to differential diagnosis and management. Vol. 10, International Journal of Emergency Medicine. Springer London; 2017.
- Hahn J, Hoffmann TK, Bock B, Nordmann-Kleiner M, Trainotti S, Greve J. M E D I C I N E Angioedema An Interdisciplinary Emergency. Dtsch Arztebl Int. 2017;114:489–96.
- Bernstein JA, Moellman J. Emerging concepts in the diagnosis and treatment of patients with undifferentiated angioedema. Int J Emerg Med [Internet]. 2012 [cited 2022 Oct 16];5(39). Available from: http://www.intjem.com/content/5/1/39
- Long BJ, Koyfman A, Gottlieb M. Evaluation and Management of Angioedema in the Emergency Department. West J Emerg Med Integr Emerg Care with Popul Heal. 2019;20(4).
- Culley CM, Dibridge JN, Wilson GL. Off-Label Use of Agents for Management of Serious or Life-threatening Angiotensin Converting Enzyme Inhibitor–Induced Angioedema. Ann Pharmacother [Internet]. 2015 Sep 28 [cited 2022 Oct 19];50(1):47–59. Available from: https://journals-sagepub-com.proxy.bib.uottawa.ca/doi/full/10.1177/1060028015607037
- Banerji A, Blumenthal KG, Lai KH, Zhou L. Epidemiology of ACE Inhibitor Angioedema Utilizing a Large Electronic Health Record. J Allergy Clin Immunol Pract. 2017 May 1;5(3):744–9.
- Quickfall MDCM D, Jakubovic B, Zipursky JS, Zipursky J. Angiotensin-converting-enzyme (ACE) inhibitors are the leading cause of drug-induced angioedema Angiotensin-converting-enzyme inhibitor-induced angioedema 5 Angiotensin receptor blockers can be used if there is a clinical indication for renin-angiotensin-aldosterone system blockade. C | MAY [Internet]. 2021 [cited 2022 Oct 25];17:735. Available from: www.cmaj.ca/lookup/doi/10.1503/
- Allen Kaplan AP, Greaves MW, Carolina S. Angioedema. J Am Acad Dermatol. 2005;53(3).
- Sharma P, Nagarajan V. Q: Can an ARB be given to patients who have had angioedema on an ACE inhibitor? [cited 2022 Oct 21]; Available from: www.ccjm.org
- Makani H, Messerli FH, Romero J, Wever-Pinzon O, Korniyenko A, Berrios RS, et al. Meta-Analysis of Randomized Trials of Angioedema as an Adverse Event of Renin–Angiotensin System Inhibitors. Am J Cardiol. 2012 Aug 1;110(3):383–91.
- Pines JM, Poarch K, Hughes S. Recognition and Differential Diagnosis of Hereditary Angioedema in the Emergency Department. J Emerg Med. 2021 Jan 1;60(1):35–43.
- Hébert J, Boursiquot JN, Chapdelaine H, Laramée B, Desjardins M, Gagnon R, et al. Bradykinin-induced angioedema in the emergency department. Int J Emerg Med. 2022 Dec 1;15(1).
- Jaiganesh T, Wiese M, Hollingsworth J, Hughan C, Kamara M, Wood P, et al. Acute angioedema: Recognition and management in the emergency department. Eur J Emerg Med. 2013 Feb;20(1):10–7.
- Zingale LC, Beltrami L, Zanichelli A, Maggioni L, Pappalardo E, Cicardi B, et al. Angioedema without urticaria: a large clinical survey. CMAJ. 2006;175(9):1065.
- Zuraw BL, Bernstein JA, Lang DM, Contributors W, Craig T, Dreyfus D, et al. Practice parameter A focused parameter update: Hereditary angioedema, acquired C1 inhibitor deficiency, and angiotensin-converting enzyme inhibitor-associated angioedema. J Allergy Clin Immunol [Internet]. 2013 [cited 2022 Oct 19];131:1491-1493.e25. Available from: http://dx.doi.org/10.1016/j.jaci.2013.03.034
- Hassen GW, Kalantari H, Parraga M, Chirurgi R, Meletiche C, Chan C, et al. Fresh frozen plasma for progressive and refractory angiotensin-converting enzyme inhibitor-induced angioedema. J Emerg Med. 2013 Apr;44(4):764–72.
- Chen SX, Hermelin D, Weintraub SJ. Possible donor-dependent differences in efficacy of fresh frozen plasma for treatment of ACE inhibitor–induced angioedema. J Allergy Clin Immunol Pract. 2019 Jul 1;7(6):2087–8.
- Warrier MR, Copilevitz CA, Dykewicz MS, Slavin RG. Fresh frozen plasma in the treatment of resistant angiotensin-converting enzyme inhibitor angioedema. Ann Allergy, Asthma Immunol. 2004 May 1;92(5):573–5.
- Stewart M, McGlone R. Fresh frozen plasma in the treatment of ACE inhibitor-induced angioedema. BMJ Case Rep. 2012;
- Shiber J. Lingual Angioedema due to ACE-Inhibitor. Trop Med Surg. 2014;2:1.
- Adebayo O, Wilkerson RG. Angiotensin-converting enzyme inhibitor–induced angioedema worsened with fresh frozen plasma. Am J Emerg Med. 2017 Jan 1;35(1):192.e1-192.e2.
- Stoldt J, Cox C, Matusz E. Tranexamic acid use in the setting of ACE inhibitor induced angioedema. Am J Emerg Med. 2022 May 1;55:230.e3-230.e4.
- Wang K, Geiger H, McMahon A. Tranexamic acid for ACE inhibitor induced angioedema – A case report. Am J Emerg Med [Internet]. 2020 Oct 21 [cited 2022 Oct 22];43:292.e5-292.e7. Available from: http://www.ncbi.nlm.nih.gov/pubmed/34756369
- Beauchêne C, Martins-Héricher J, Denis D, Martin L, Maillard H. Intérêt de l’acide tranexamique en traitement d’urgence de première intention des crises d’angiœdème bradykinique sous IEC. La Rev Médecine Interne. 2018 Oct 1;39(10):772–6.
- Hasara S, Wilson K, Amatea J, Anderson J. Tranexamic Acid for the Emergency Treatment of Angiotensin-Converting Enzyme Inhibitor-Induced Angioedema. Cureus. 2021 Sep 20;13(9).
- Straka BT, Ramirez CE, Byrd JB, Stone E, Woodard-Grice A, Nian H, et al. Effect of bradykinin receptor antagonism on ACE inhibitor-associated angioedema. J Allergy Clin Immunol. 2017 Jul 1;140(1):242-248.e2.
- Baş M, Greve J, Stelter K, Havel M, Strassen U, Rotter N, et al. A Randomized Trial of Icatibant in ACE-Inhibitor–Induced Angioedema. N Engl J Med [Internet]. 2015 Jan 29 [cited 2022 Oct 19];372(5):418–25. Available from: https://www-nejm-org.proxy.bib.uottawa.ca/doi/full/10.1056/NEJMoa1312524
- Fok JS, Katelaris CH, Brown AF, Smith WB. Icatibant in angiotensin-converting enzyme (ACE) inhibitor-associated angioedema. Intern Med J [Internet]. 2015 Aug 1 [cited 2022 Oct 23];45(8):821–7. Available from: https://onlinelibrary-wiley-com.proxy.bib.uottawa.ca/doi/full/10.1111/imj.12799
- Sinert R, Levy P, Bernstein JA, Body R, Sivilotti MLA, Moellman J, et al. Randomized Trial of Icatibant for Angiotensin-Converting Enzyme Inhibitor–Induced Upper Airway Angioedema. J Allergy Clin Immunol Pract. 2017 Sep 1;5(5):1402-1409.e3.
- Cai G, Barber C, Kalicinsky C. Review of icatibant use in the Winnipeg Regional Health Authority. Allergy, Asthma Clin Immunol. 2020 Dec 1;16(1).
- Lewis LM, Graffeo C, Crosley P, Klausner HA, Clark CL, Frank A, et al. Ecallantide for the Acute Treatment of Angiotensin-Converting Enzyme Inhibitor–Induced Angioedema: A Multicenter, Randomized, Controlled Trial. Ann Emerg Med. 2015 Feb 1;65(2):204–13.
- Bernstein JA, Moellman JJ, Collins SP, Hart KW, Lindsell CJ. Effectiveness of ecallantide in treating angiotensin-converting enzyme inhibitor–induced angioedema in the emergency department. Ann Allergy, Asthma Immunol. 2015 Mar 1;114(3):245–9.
- Rasmussen ER, Bygum A. ACE-inhibitor induced angio-oedema treated with complement C1-inhibitor concentrate. BMJ [Internet]. 2013 [cited 2022 Oct 19]; Available from: http://group.bmj.com/group/rights-licensing/permissions.
- Perza M, Koczirka S, Nomura JT. C1 Esterase Inhibitor for Ace-Inhibitor Angioedema: A Case Series and Literature Review. J Emerg Med. 2020 Mar 1;58(3):e121–7.
- Ibarra F, Cruz M. C1 esterase inhibitor use in the management of lisinopril-induced angioedema: A case series. J Am Pharm Assoc (2003) [Internet]. 2020 Nov 1 [cited 2022 Oct 19];60(6):e361–4. Available from: https://pubmed.ncbi.nlm.nih.gov/32727672/
- Donaldson IA, Alonzi R, Barratt D, Barret E, Berge V, Bott S, et al. Focal Therapy: Patients, Interventions, and Outcomes-A Report from a Consensus Meeting. Eur Urol [Internet]. 2015 [cited 2021 Nov 10];67(4):771–7. Available from: http://dx.doi.org/10.1016/j.eururo.2014.09.018
- Greve J, Bas M, Hoffmann TK, Schuler PJ, Weller P, Kojda G, et al. Effect of C1-Esterase-inhibitor in angiotensin-converting enzyme inhibitor-induced angioedema. Laryngoscope [Internet]. 2015 Jun 1 [cited 2022 Oct 23];125(6):E198–202. Available from: https://onlinelibrary-wiley-com.proxy.bib.uottawa.ca/doi/full/10.1002/lary.25113
- Mohorn PL, Roebuck LD, Raybon-Rojas E, Duncan C. C1 esterase inhibitor for angiotensin-converting enzyme inhibitor-induced angioedema at a community teaching health system: A brief retrospective propensity-matched cohort study. Am J Emerg Med [Internet]. 2021 [cited 2022 Oct 18];49:6–9. Available from: https://doi.org/10.1016/j.ajem.2021.05.036
- Hospitals shun cheap drug used to stop bleeding – The Globe and Mail [Internet]. [cited 2022 Oct 19]. Available from: https://www.theglobeandmail.com/life/health-and-fitness/hospitals-shun-cheap-drug-used-to-stop-bleeding/article4178385/
- Canadian Agency for Drugs and Technologies in Health. Common Drug Review Pharmacoeconomic Review Report. 2018.
- Oksana Prokopchuk-Gauk, MD FRCPC Transfusing Wisely: Appropriate Blood Use in Patient Care [Internet]. 2021 [cited 2022 Oct 19]. Available from: https://saskblood.ca/transfusion-best-practice-recommendations/
- William Cardarelli P. Managed Care Implications of Hereditary Angioedema. Am J Manag Care [Internet]. 2013 [cited 2022 Oct 30];19(7 Suppl). Available from: https://www.ajmc.com/view/ace010_13jun_cardarelli_s119to24
- Mudd PA, Hooker EA, Stolz U, Hart KW, Bernstein JA, Moellman JJ. Emergency department evaluation of patients with angiotensin converting enzyme inhibitor associated angioedema. Am J Emerg Med. 2020 Dec 1;38(12):2596–601.
- Khan DA, Kocatürk E, Bauer A, Aygören-Pürsün E. Clinical Commentary Review What’s New in the Treatment of Urticaria and Angioedema. J Allergy Clin Immunol [Internet]. 2021 [cited 2022 Oct 18];9(6). Available from: https://doi.org/10.1016/j.jaip.2021.03.012
- Gayen S, Sinha T, Dronamraju V, Lashari B, Zhao H, Dhungana S. Nasopharyngolaryngoscopy as a Triage Tool for Airway Compromise in Angioedema: A Retrospective Cohort Study. Cureus. 2022 Apr 2;14(4).
- Chan NJ, Soliman AMS. Angiotensin Converting Enzyme Inhibitor-Related Angioedema. https://doi.org/101177/0003489414543069 [Internet]. 2014 Jul 24 [cited 2022 Oct 18];124(2):89–96. Available from: https://journals.sagepub.com/doi/10.1177/0003489414543069
- Bentsianov BL, Parhiscar A, Azer M, Har-El G. The role of fiberoptic nasopharyngoscopy in the management of the acute airway in angioneurotic edema. Laryngoscope. 2000;110(12):2016–9.
- Aime Airway [Internet]. [cited 2022 Oct 27]. Available from: https://aimeairway.ca/announcement/62/excerpts-from-airway-physiology-and-anatomy-a-deep-dive-chapter-in-airway-management-in-emergencies-the-infinity-edition
- Laryngoscopic photograph showing diffuse edema of the glottic and… | Download High-Quality Scientific Diagram [Internet]. [cited 2022 Oct 27]. Available from: https://www.researchgate.net/figure/Laryngoscopic-photograph-showing-diffuse-edema-of-the-glottic-and-supraglottic-mucosa_fig1_273469483
- Bas M. Evidence and evidence gaps of medical treatment of non-tumorous diseases of the head and neck. GMS Curr Top Otorhinolaryngol Head Neck Surg [Internet]. 2016 [cited 2022 Oct 27];15:Doc02. Available from: /pmc/articles/PMC5169075/
- Difficult Airway society. DAS Difficult Intubation guidelines – overview . 2015.


Trackbacks/Pingbacks