As per the 2021 updated guidelines for the management of priapism by Ericson et al, priapism is defined as a penile erection that lasts longer than 4 hours after completion of sexual activity or that is unrelated to sexual activity. The European Association of Urology (EAU) don’t include the 4-hour time limit in their definition per se, but practically speaking, reference a 4 hour cut-off necessitating intervention.
While priapism is a relatively low incidence (1.5 – 5.4 in 100, 000)1,2 presentation to the emergency department, it is a truly a life and limb medical emergency.
There are three general classifications of priapism: ischemic, non-ischemic, and recurrent intermittent (or stuttering) priapism. Our biggest focus today will be on ischemic priapism as it comprises ~ 95% or priapism presentations3, but also insofar as it is time sensitive, contrary to non-ischemic or stuttering priapism. These latter two will often be managed longitudinally by our urologist colleagues in conjunction with their family doctor. The role of the emergency physician in the latter two of these conditions will be identification and appropriate referral.
The term priapism comes from a reference to a minor Greek god of fertility, Priapus who is depicted as having a rather large and permanent erection, that ironically, due to a curse put on him by the goddess Hera, would result in impotence at the most inopportune time. This parallel bears a familiar yet cruel irony for those who suffer with priapism.
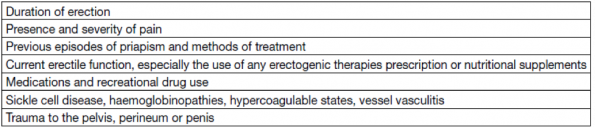
There are typically no clinical exam findings that will indicate what form of priapism a patient has; the etiology will be taken from the history. Salonia outlines questions that should be asked with every instance of unwanted erection in the emergency department:
How long has the erection been present? Is it painful? Previous history of priapism and how was it treated? Currently being managed for erectile dysfunction and what prescribed medications are they using? What other medications or recreational drugs are being used? Does the patient suffer from blood or blood vessel diseases that might predispose to priapism? Have they recently had a trauma to the pelvis? Think of the skater coming from the skatepark after losing his footing on that sweet nollie backside railslide. This latter presentation, typically associated with pelvic or straddle injuries, often will result in the non-ischemic variant of priapism due to formation of an arterio-venous shunt, resulting in persistent and partial tumescence.
The potential causes for priapism comprise a very long list, however the most common causes for priapism are related to prescribed or illicit drugs, treatments for sexual dysfunction, or as consequence of another disorder like sickle cell disease, hematologic, metabolic, or neurologic disorders, or neoplastic syndromes. But in many cases, priapism is idiopathic.3
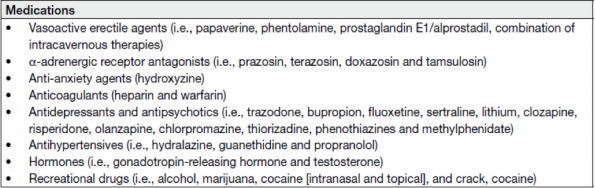
Just remember the mnemonic HAAAAARD-ED (that’s 5 A’s). This mnemonic includes Hormones (GnRH and testosterone), Antidepressants (trazodone, bupropion, fluoxetine, sertraline), Antipsychotics (clozapine, risperidone, olanzapine, chlorpromazine, phenothiazines, and methylphenidate), Anxiety meds (hydroxyzine), Anticoagulants (heparin and warfarin), Antihypertensives (hydralazine, guanethidine, and propranolol), Recreational Drugs (EtOH, Cannabis, cocaine, crack), and Erectile Dysfunction meds (papaverine, phentolamine, PGE1/alprostadil, combination intracavernous therapies.
The reported frequency of these etiologies varies among studies, but Salonia reports some of the following numbers, which were taken from various sources:
- Idiopathic – 35%
- Sickle Cell Disease – 63% pediatric cases; 23% adult cases (among hemoglobinopathies)
- Intracavernosal injections – 0.4 – 35%
- Second generation Antipsychotics – 33.8%
- Alpha-adrenergic antagonists – 8.8%
- Other medications – 11.3%
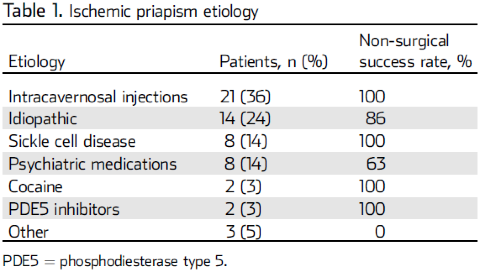
You’ll note that SCD comprises a large number of cases and there can certainly be overlap of multiple etiologies, i.e. a patient with SCD taking an antipsychotic for schizophrenia or using intracavernosal injections for baseline erectile dysfunction. The relative risk can stack in this way, but it is hard to put a number to it. Of note, men with SCD will have a life-time probability of 29-42% of developing ischemic priapism.3 A small study of 58 patients by Ridyard et al reported a slight variation on these numbers but would give you a general idea based on a single patient population:
Physiology of Erection
For us to understand the pathophysiology and treatment of priapism, we must understand the anatomy of the penis and physiology of a normal erection. The majority of the penile tissue is comprised of twin corpora cavernosa comprised of arteriolar and arterial smooth muscle, encased in the tunica albuginea, and the corpus spongiosum which houses the urethra on the ventral side. While flaccid, the smooth muscle of the cavernosa is fully contracted, allowing for just enough blood flow to provide nutrition, but when sexually aroused, neurotransmitters from the cavernous nerve terminals result in smooth muscle relaxation and subsequent dilatation and engorgement with blood. The blood is trapped in the cavernosa by expanding sinusoids, and further maintained by compression of the subtunical venular plexuses and emissary veins, maintaining its turgid state. Parasympathetic innervation, arising from the pelvic plexus, originating from spinal nerves S2-S4, induces tumescence, whereas sympathetic stimulation from the same plexus originating at T10 – L2 (often as a result of ejaculation in normal anatomy and physiology) results in detumescence.4 At a molecular level, nitric oxide (NO) is released from endothelial cells and the cavernous nerve, promoting cGMP production (via guanylate cyclase), activating protein kinase G and resultant smooth muscle relaxation by decreasing intracellular calcium.5 NO induced cGMP then also upregulates PDE-5, eventually resulting in detumescence by generating a negative feedback cycle.
Physiology of Priapism
As for the pathophysiology of priapism, you’ll note in the diagram here that phosphodiesterase contributes to the hydrolysis of cGMP into 5’GMP, so a phosphodiesterase inhibitor would result in higher quantities of cGMP intracellularly resulting in sustained tumescence. Typically, oral PDEi agents like sildenafil have not been directly correlated with high incidence of priapism, rather, intracavernous injection therapies like prostaglandin E1, papaverine, papaverine/phentolamine, or Trimix (all 3 combined) have a far higher likelihood of causing the condition (0.4-35%). The prosed mechanisms for the large part of the other medications on this list are due to alpha-adrenergic blockade, resulting in absent sympathetic activity to facilitate detumescence, i.e., anti-hypertensives, antipsychotics, and antidepressants all seem to be linked to alpha-blockade. However, there are some exceptions on the list: heparin use – anti-platelet antibodies –> platelet aggregation –> poor outflow; cocaine results in norepinephrine depletion from sympathetic nerve terminals.
According to Idris et al, in sickle cell disease, the main cause of failure in detumescence is intracavernosal stasis resulting in veno-occlusive crisis and obstruction of venous drainage. 7 However, there are other pathways that are implicated. Endothelial damage and chronic hemolysis results in endothelial NO depletion, resulting in overall decrease of PDE-5, impairing detumescence. There are other proposed mechanisms but the minutiae would not be contributory to ED management. SCD typically results in the stuttering form of priapism, resulting in multiple instances of priapism over a short period of time. Often each instance will be managed the same as ischemic priapism, however, early urology/hematology involvement will be necessary to either start medications for secondary prevention or to consider shunt procedure or penile implant.
Diagnosis
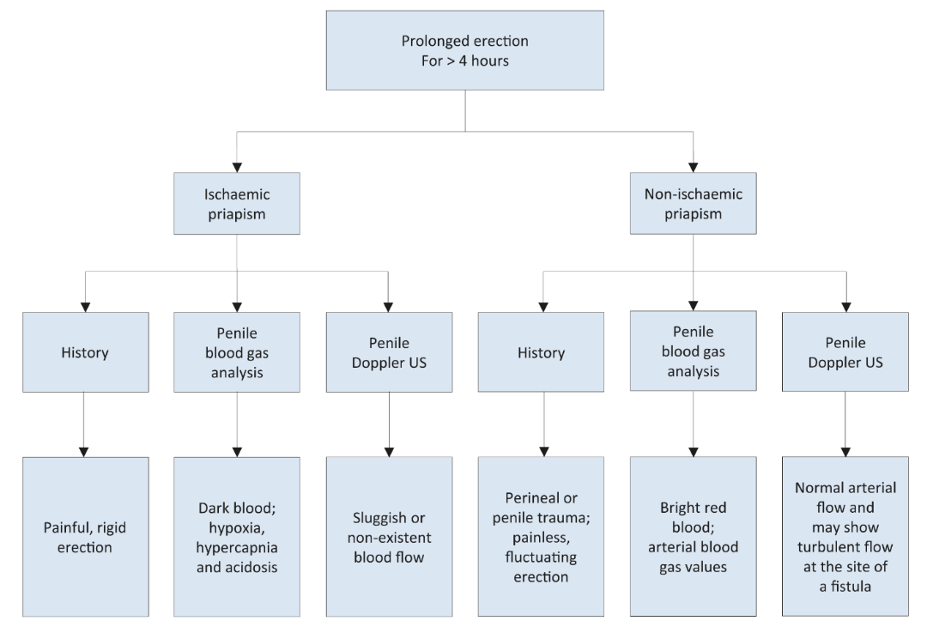
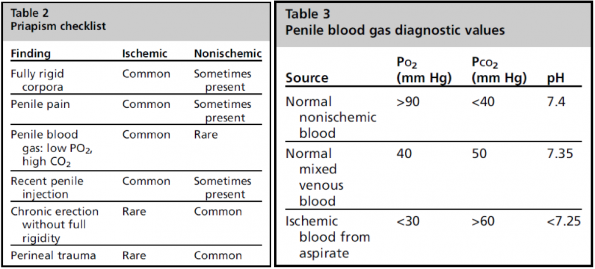
The Urologic Clinics of North American, updated in 2021, have published the following tables to assist in the diagnosis and investigation of priapism. The first is a diagnostic checklist that takes into account history and blood gas readings to help the clinician differentiate between ischemic and non-ischemic priapism. A fully rigid corpora, severe penile, pain, and history of recent penile injection is much more common in the ischemic variant. History of chronic, partially rigid erection or recent perineal trauma would be more suggestive of nonischemic etiology.
However, what will help really cinch the diagnosis is penile aspirate blood gas. A normal arterial blood gas typically has a PO2 of > 90 mmHg, PC02 of <40 mmHg and pH of 7.4. Ischemic blood from an aspirate is typically deoxygenated with pO2 of < 30, pCO2 > 60, and an acidotic pH of < 7.25.8
The EAU make several strong recommendations for the diagnosis of ischemic priapism, including thorough history; examination of the penis, testicles, perineum and abdomen; laboratory testing including CBC, coag studies, and blood gas analysis of blood aspirated from the corpus cavernosa; and colour doppler ultrasound prior to aspiration for detumescence or phenylephrine to accurately differentiate ischemic from nonischemic priapism.
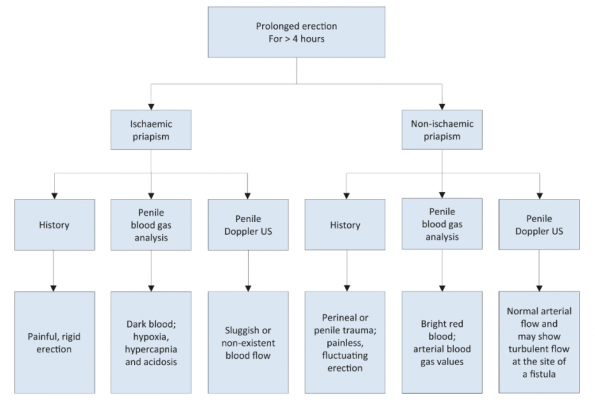
These guidelines also provide this nice flow chart, which combines history, penile blood gas and penile doppler ultrasound to help distinguish ischemic from nonischemic forms of priapism.
How does doppler ultrasound perform in confirming the diagnosis of priapism? Wakrim et al conducted a systematic review in 2022, comprising 12 articles with 363 patients, seeking to compare penile doppler ultrasound to cavernosal blood gas analysis (BGA) or pudendal artery angiography (PAA) in differentiating ischemic from non-ischemic priapism. When compared to PAA, PDU showed sensitivity of 100%, specificity of 73%, PPV of 100% and NPV of 86%. There was no comparable difference between BGA or PDU across all studies.9 I know we did not talk about pudendal artery angiography in its role for diagnosis of priapism, however, I don’t believe we need to as it is not recommended in any of the guidelines and there are two alternative tests that are easily accessible, neither of which subject the patient to radiation.
Management
Management of priapism in the ED if focused on two main approaches, aspiration +/- saline irrigation and injection of phenylephrine. However, there are also some more conservative techniques that can be tried prior to these more invasive methods. Exercise, ejaculation, application of an ice pack, cold baths, or cold water enemas have been proposed, however, there is no good quality evidence to suggest these are effective; I found one case study in which a 34M achieved detumescence after climbing hospital stairs for 7 minutes.10 But let’s be honest, leading your patient through an orange theory work-out in the urgent care hallway or stairwell in the state they presented to the ED is probably poor for the hospital’s optics.
Treatment
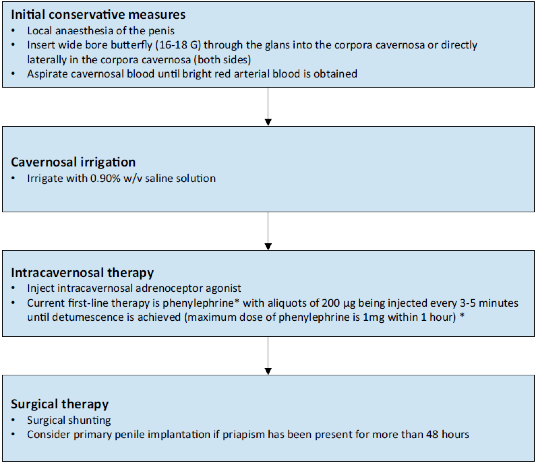
A stepwise approach is proposed by the EAU starting with local anesthesia of the penis using a dorsal penile nerve or circumferential penile block followed by aspiration of cavernosal blood, trial of irrigation with normal saline, and lastly intracavernosal injection of phenylephrine. If these techniques fail, you should be getting on the phone with urology. Let’s look at each of these procedures in turn.
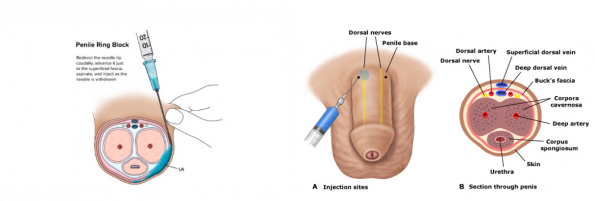
Penile Nerve Block/Ring block
To perform the dorsal penile nerve block, the clinician should prepare 1-2% lidocaine or bupivacaine 0.5% without epinephrine in a 10 mL syringe with a 25-27 g needle and landmark the 10 and 2 positions at the base of the shaft of the penis. As with any nerve block procedure, you should draw back on the syringe first, confirming you’re not in a vessel, before injecting 1-2 mL. This will anesthetize only the dorsum of the penis, so if you are aiming for circumferential anesthesia, you can perform a ring block (as seen here).
Aspiration
Once anesthetized, the next step will be aspiration of cavernosal blood with a butterfly needle and a large capacity syringe. The clinician can, once again, landmark at 10 and 2 but anywhere along the cavernosa would be appropriate, provided the corpus spongiosum on the ventral side of the penis is avoided as this could result in urethral injury. The gauge of the needle used is a question of practicality, i.e., the larger the gauge, the faster you should expect to be able to aspirate the cavernosal blood, however, you might also expect a larger hematoma at the site upon successful completion. EMRAP coverage of Priapism (1:52-2:25).
Phenylephrine Irrigation
Phenylephrine is a selective alpha-1-adrenergic receptor agonist without concomitant beta-adrenergic agonism resulting in inotropic or chronotropic cardiac effects. The solution can be mixed in concentrations anywhere from 100-500 mcg/mL and is administered in aliquots of 200 mcg every 3 -5 minutes directly into the corpora up to a maximum of 1 mg per hour.11 Cardiac monitoring is required for this part of the algorithm as phenylephrine can result in reflex bradycardia, venoconstriction resulting in increased preload and decreased arterial constriction, increasing afterload. Ensure that you are also monitoring that the needle is truly in the cavernosum as extravasation into subcutaneous tissues could theoretically result in skin necrosis. EMRAP coverage (2:44-3:30).
Sickle Cell Disease
In patients with SCD, the approach should be much the same but should be coordinated with our hematology colleagues. Proceeding with aspiration followed by phenylephrine irrigation is still the mainstay however, Idris et al recommends that these patients be referred to urology earlier and treatment initiated earlier as the risk of long-term erectile dysfunction is much higher in patients with SCD.
The one thing that should be acknowledged, however, is avoidance of exchange transfusion in these patients due to ASPEN syndrome, or Association of Sickle Cell Disease, Priapism and Exchange Transfusion and Neurologic Events. This uncommon outcome of exchange transfusion was first described in 1993 by JF Siegel described as severe headache followed by neurologic events like seizure and decreased level of consciousness requiring ventilatory support. This was conjectured to be due to cerebral ischemia from a sudden increase in percent total hemoglobin and release of vasoactive substances following penile detumescence.15 However, beyond this initial article, the resources are virtually non-existent.
Ultimately, the EAU recommends the following algorithm for management in the ED:
References
- Roghmann F, Becker A, Sammon JD, et al. Incidence of Priapism in Emergency Departments in the United States. The Journal of Urology. 2013;190(4):1275-1280.
- Eland IA, van der Lei J, Stricker BHC, Sturckenboom MJCM. Incidence of priapism in the general population. Urology. 2001;57(5):970-972.
- Salonia A, Bettochi C, Capogrosso P, et al. EAU Guidelines on Sexual and Reproductive Health. Edn. presented at the EAU Annual Congress of Milan 2023. European Association of Urology; 2023.
- Dean RC. Physiology of Penile Erection and Pathophysiology of Erectile Dysfunction. Published online 2006.
- Idris IM, Burnett AL, DeBaun MR. Epidemiology and treatment of priapism in sickle cell disease. Hematology. 2022;2022(1):450-458. doi:10.1182/hematology.2022000380
- Sangiorgi G, Cereda A, Benedetto D, et al. Anatomy, Pathophysiology, Molecular Mechanisms, and Clinical Management of Erectile Dysfunction in Patients Affected by Coronary Artery Disease: A Review. Biomedicines. 2021;9(4):432. doi:10.3390/biomedicines9040432
- Ahuja G, Ibecheozor C, Okorie NC, et al. Priapism and Sickle Cell Disease: Special Considerations in Etiology, Management, and Prevention. Urology. 2021;156:e40-e47. doi:10.1016/j.urology.2021.06.010
- Ericson C, Baird B, Broderick GA. Management of Priapism 2021 Update. Urol Clin N Am. 2021;48(4):565-576.
- Wakrim S, Ziouziou I, Ralph D, Khabbal Y. Penile Doppler ultrasound study in priapism: A systematic review. Progrès en Urologie. 2022;32(1):61-69. doi:10.1016/j.purol.2021.03.009
- Gravel J, LeBlanc C, Varner C. Management of priapism with a trial of exercise in the emergency department. CJEM. 2019;21(1):150-153. doi:10.1017/cem.2018.3
- Moussa M, Abou Chakra M, Papatsoris A, et al. An update on the management algorithms of priapism during the last decade. Arch Ital Urol Androl. 2022;94(2):237-247. doi:10.4081/aiua.2022.2.237
- Bobo GA, Almajed W, Conlon J, Morenas RA, Hellstrom WJG. An overview of emergency pharmacotherapy for priapism. Expert Opinion on Pharmacotherapy. 2022;23(12):1371-1380. doi:10.1080/14656566.2022.2099271
- Martin C, Cocchio C. Effect of phenylephrine and terbutaline on ischemic priapism: a retrospective review. The American Journal of Emergency Medicine. 2016;34(2):222-224. doi:10.1016/j.ajem.2015.10.029
- Ridyard DG, Phillips EA, Vincent W, Munarriz R. Use of High-Dose Phenylephrine in the Treatment of Ischemic Priapism: Five-Year Experience at a Single Institution. The Journal of Sexual Medicine. 2016;13(11):1704-1707. doi:10.1016/j.jsxm.2016.09.010
- Siegel J, Rich M, Brock W. Association of sickel cell disease, priapism exchange transfusion and neurological events: ASPEN syndrome. Journal of Urology. November1993;150(5):1480-1482.