Your next patient is an 80-year-old male, who had been functioning well until about a month ago. He has a history of type II diabetes, hypertension, and dyslipidemia, and has been working full-time.
Upon reading the triage note and assessing the patient, you discover a troubling recent medical history. Over the past few months, he has experienced worsening weakness in his legs and hands, along with issues related to balance and multiple falls. Additionally, he complains of bilateral hand numbness. Further investigation of the electronic medical record reveals that he has visited a peripheral emergency department four times in the last two months all for the same set of symptoms.
During these visits, the initial diagnoses ranged from joint pain to knee osteoarthritis. As his symptoms progressed, he was diagnosed with weakness, possibly related to polymyositis, due to an elevation in inflammatory markers. He was started on steroids. On a subsequent visit, the diagnosis remained the same. There were attempts to transfer him to a larger hospital for a CT scan of the head, but this was declined due to capacity issues. The patient was offered admission peripherally but declined.
Upon your evaluation, you find that the patient is now unable to walk safely. While unsure about the exact nature of the problem, you strongly suspect a neurological issue. Recognizing the need for admission due to his mobility challenges, you decide to consult the neurology team to investigate further.
What is a Myelopathy
A myelopathy is a broad term used to describe conditions that result in damage or dysfunction in the spinal cord, meningeal, or perimenigeal spaces.1 These conditions can arise from various factors, such as traumatic injuries, vascular diseases, infections, inflammation, or autoimmune disorders, and they can lead to serious outcomes like quadriplegia, paraplegia, or sensory deficits. Early recognition and intervention are crucial to potentially reverse these effects.
It’s important to differentiate myelopathy from myelitis; which specifically involves inflammation of the spinal cord and is considered a type of myelopathy. Myelopathies can be further classified into compressive causes (more common in older patients), and non-compressive causes; which encompass a wide range of potential underlying factors, including infections, toxins, metabolic issues, and paraneoplastic processes.1 Recognizing when to suspect myelopathy, understanding examination findings, and knowing when to order an MRI evaluation are essential skills for emergency physicians.
How can we improve our Physical Exam?
One simple way to think about the physical examination is from an anatomical perspective. The spinal cord is made of nervous tissue that extends from the medulla oblongata in the brain stem, all the way to L2 in the vertebral column. After the spinal cord tapers out, the spinal nerves continue to branch out and form the cauda equina.
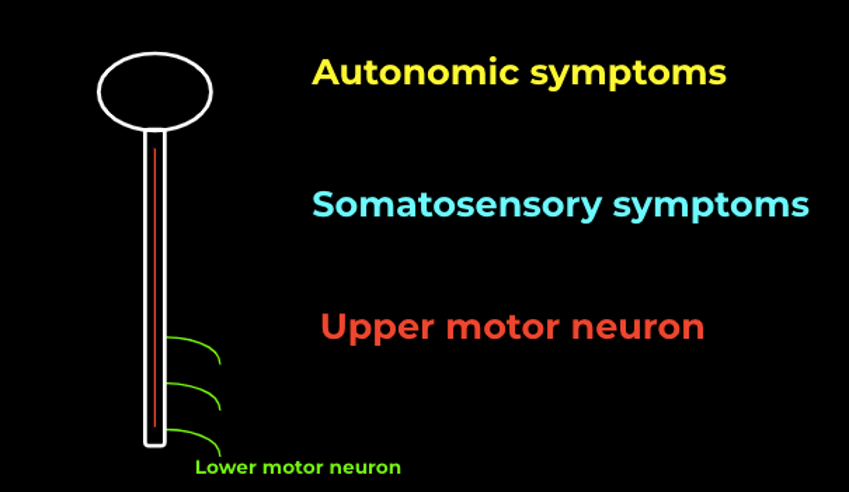
When thinking about compression or inflammation of the spinal cord, the simplest approach is to think about what symptoms this can generate. In its simplest format, these symptoms can be categorized into three main categories (figure 1.):
- Autonomic symptoms
- The spinal cord contains axons responsible for autonomic functions, including those related to bowel, bladder, and genital function. Understanding the importance of autonomic symptoms, especially urinary retention, is vital. It serves as a crucial indicator, alerting clinicians to promptly screen for possible spinal cord compression or dysfunction spanning its entire length.
- Somatosensory symptoms
- Axons convey somatosensory information through specific tracts: pain and temperature via the spinothalamic tract, and proprioception and vibration through the dorsal column. This highlights the concept of a sensory level, aiding in localizing spinal cord involvement, and even pinpointing a specific sensory transmission column.
- Upper motor neuron findings
- Understanding the differentiation between upper and lower motor neurons (UMNs and LMNs) is crucial in spinal cord assessment. LMNs extend from the spinal cord to innervate skeletal muscles, and their dysfunction can result in specific findings like muscle wasting, decreased tone, and absent reflexes.
- Conversely, UMNs, which extend from the brain through the spinal cord, have unique clinical characteristics when damaged, including increased tone, clonus, decreased power, hyperreflexia, and the presence of signs like the Hoffmann and Babinski signs. The Hoffmann sign, often less familiar, involves flicking the middle finger’s nail to observe quick adduction of the thumb and index finger. A positive Hoffmann sign suggests UMN issues, primarily in the cervical spine, and is akin to the upper limb version of the Babinski sign.
Understanding this is crucial as a compression/inflammation at T12 will present with predominantly upper motor neuron findings, where as a compression at L3 will present with predominantly lower motor neuron findings. In the case of lower thoracic pathology, charting the classic “no feature of cauda” would be insufficient.
What is Degenerative Cervical Myelopathy (DCM)?
Degenerative Cervical Myelopathy (DCM) is a neurological disorder characterized by spinal cord compression in the neck due to age-related degenerative changes. Patients with DCM can experience a range of non-specific symptoms, including limb pain, numbness, coordination problems, imbalance, and bladder issues. These symptoms arise from the pressure placed on the spinal cord by factors such as disc herniation, ligament thickening, ossification, and bone spurs collectively referred to as spondylosis.
Degenerative Cervical Myelopathy is likely more common than reported, as incidental cord compression is frequently detected in MRI scans, and a significant proportion of individuals with asymptomatic cord compression may eventually develop DCM.2 The exact prevalence remains uncertain, and many cases go undiagnosed, making it an important differential for emergency physicians to consider in the elderly presenting with falls or weakness.
How is Degenerative Cervical Myelopathy Diagnosed? Why is it missed?
The diagnosis of DCM relies on a combination of clinical suspicion, a targeted physical examination, and imaging studies. Historically, a key feature of myelopathy is the evolution of symptoms over months, often involving neck pain/stiffness, limb/body pain, upper limb – weakness/numbness/loss of dexterity, lower limb – stiffness/weakness/sensory loss, paraesthesia, autonomic symptoms, imbalance, and falls. However, these symptoms can be non-specific and may overlap with age-related decline or other neurological conditions. Physical examination findings, such as the Hoffman sign, can be indicative but lack the sensitivity and specificity to confirm the diagnosis on its own.3 When clinical suspicion is high, the next step is typically an MRI of the cervical spine, which reveals central stenosis with cord compression.
DCM is often missed due to its non-specific and subtle early features, which can be mistaken for other neurological conditions. Delayed diagnosis is common, with some patients experiencing an average delay of 2.2 years before receiving a correct diagnosis.4 One proposed contributing factor is the incomplete neurological assessment by healthcare professionals who may lack awareness of the disease. A study comparing assessments by primary care providers and specialists revealed significant differences in documenting key examination findings, such as hyperreflexia, the Hoffman sign, and lower limb spasticity.5 These variations in assessment can contribute to delayed diagnosis and referral to spinal surgery specialists, emphasizing the importance of raising awareness and improving clinical assessments for myelopathy.
How is Degenerative Cervical Myelopathy managed?
The treatment approach for DCM typically involves referral to a spine surgeon for assessment. Patients with the appropriate constellation of myelopathy symptoms and consistent MRI findings should undergo evaluation by a spine surgeon. In cases of severe or rapidly progressing symptoms, prompt consultation with a spine surgeon, either through an on-call service or in the emergency department, is recommended. Patients with milder or slowly progressive symptoms can be assessed as outpatients.
Surgery is the primary treatment option for individuals with moderate or severe DCM or those experiencing disease progression. Surgical decompression can arrest the advancement of the condition and offer limited yet meaningful improvements in pain, function, and quality of life.4 It’s essential to note that surgery does not guarantee the restoration of previously lost neurological function. For patients with mild, stable DCM or non-specific symptoms without upper motor neuron findings on examination, the focus may be on symptom management and regular follow-up to monitor for progression.
Case Resolution:
In the initial case, the patient presented with progressive bilateral arm and leg weakness over a month, without neck pain, and a loss of hand dexterity. There were no reported bowel or bladder symptoms. On physical examination, cranial nerves appeared normal, while strength was mostly normal, but upper motor neuron findings were present, including bilateral Babinski and Hoffman signs. The patient also had an inability to ambulate safely. Based on their clinical assessment, neurology recommended an MRI of the cervical spine.
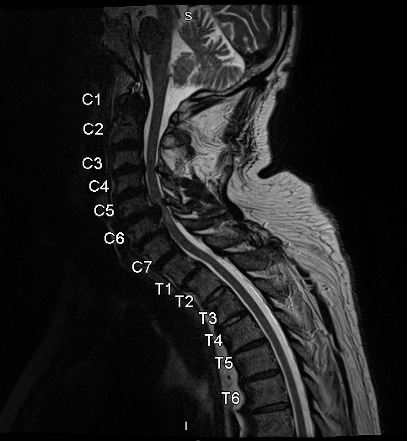
The MRI findings revealed central disc extrusion with inferior migration at C5-C6 causing severe cord compression and T2 signal abnormality (figure 2.). Moderate narrowing was also observed at C3-C4 and C4-C5. As a result, the patient was transferred to neurosurgery for decompressive surgery. Several months after the surgery, the patient showed significant improvement in neurological deficits compared to their pre-surgical state.
References
- Granados Sánchez AM, María L, Posada G, Andrés C, Toscano O, López López A. review articles DIAGNOSTIC APPROACH TO MYELOPATHIES ENfOquE DIAGNóSTICO DE LAS MIELOPATíAS.
- Milligan J, Ryan K, Fehlings M, Frcsc F, Bauman C. Degenerative cervical myelopathy Diagnosis and management in primary care.
- Grijalva RA, Hsu FPK, Wycliffe ND, et al. Hoffmann sign: clinical correlation of neurological imaging findings in the cervical spine and brain. Spine (Phila Pa 1976). 2015;40(7):475-479. doi:10.1097/BRS.0000000000000794
- Davies BM, Mowforth OD, Smith EK, Kotter MRN. Degenerative cervical myelopathy. BMJ. 2018;360. doi:10.1136/BMJ.K186
- Hilton B, Tempest-Mitchell J, Davies B, Kotter M. Assessment of degenerative cervical myelopathy differs between specialists and may influence time to diagnosis and clinical outcomes. PLoS One. 2018;13(12). doi:10.1371/JOURNAL.PONE.0207709