Trauma is the leading cause of child and youth mortality globally (1). Pediatric trauma is a high acuity but low frequency event (2). Trauma is children is 7 times less frequent in comparison to the adult population (2). There is a well-documented correlation amongst EMS and adult ED physicians who provide less pediatric care to feeling a greater degree of anxiety and discomfort surrounding these cases (3, 4).
To date, there are no randomized control trials on the topic of pediatric traumatic arrest algorithms or procedures such as emergency resuscitative thoracotomy (ERT) (5). This leads to most pediatric guidelines being extrapolated heavily from the adult literature as well as retrospective pediatric studies (5, 6). Therefore, the Western Trauma Association, the Pediatric Trauma Society and the Eastern Association for Trauma Surgery collaborated to create a pediatric traumatic cardiac arrest algorithm (5).
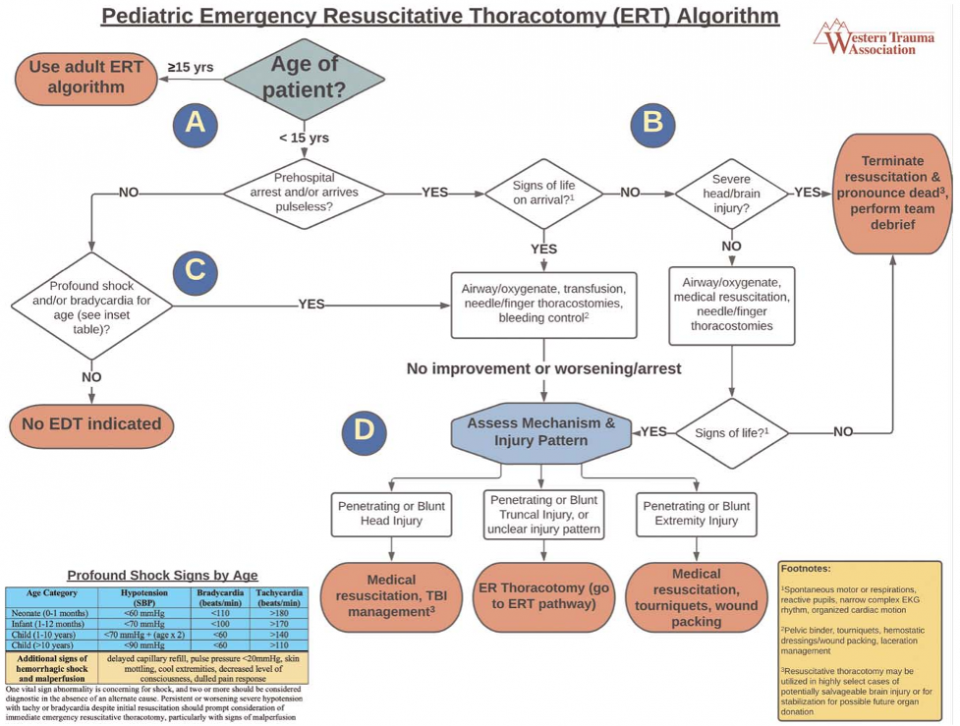
New Pediatric Traumatic Arrest Algorithm
This portion of the algorithm which focuses on the initial assessment and management of pediatric traumatic cardiac arrest (5). Age cut offs for pediatric versus adult algorithms vary widely in the literature (7-11). However, one US analysis of over 40 years of data concluded ERT survival rates of those <15 was <1% versus around 5% for adolescents 15 and older (8). A more recent analysis of the US national trauma databank revealed no survivors in pediatric patients who received ERT without signs of life for both blunt and penetrating trauma (9). This is consistent with multiple other studies showing no pediatric survivors of ERT if signs of life are absent (12-14). As such, this was used to create both the age cut off and to delineate who to consider for an ERT.
This does contradict the commonly held notion that children have a higher injury tolerance, and that pediatric survival is always better. As such, this is why the pathway for pediatric ERT is more restrictive than its adult counterpart. Children do have a unique physiology with lower total circulating blood volume than adults which puts them at higher risk for exsanguination (15). Furthermore, children are better able to compensate for blood loss with vasoconstriction to maintain their blood pressure while presenting with only tachycardia (15). Thus, hemodynamic decompensation and shock happen later and to a more severe degree than adults (15). This shorter window of tolerance for hypoxia and cerebral/ cardiac ischemia is why children with traumatic cardiac arrest/ no signs of life are more likely to be at an irreversible point of exsanguination.
The first determination in any trauma activation is whether there has truly been a prehospital arrest, or the patient is pulseless on arrival to hospital. If the patient has a pulse, a rapid primary survey, vital signs, and cardiac monitoring is initiated. Profound shock was defined as the presence of at least two of bradycardia, tachycardia, or hypotension for age. A blue age-based vital abnormality table is provided in the bottom left of the algorithm (5). If the patient has these, treatment of reversible causes of traumatic arrest, such as hemorrhage, pneumothorax and hemothorax are recommended. This includes securing an airway (oxygenation/ ventilation), transfusion, needle/ finger thoracostomies and bleeding control (pelvic binder, tourniquets, hemostatic dressings/ wound packing, laceration repair). Given children can maintain relatively normal BP even with ongoing large hemorrhage, worsening tachycardia is usually the primary sign of bleeding (16). The change to bradycardia and hypotension is an ominous sign of full arrest (16). This population may also benefit from resuscitative endovascular balloon occlusion of the aorta (REBOA) (17-19). Unfortunately, there is a lack of pediatric data to incorporate REBOA use in pediatric traumatic arrest currently, though the development of pediatric REBOA catheter sizes is ongoing (20).
If the patient did present pulseless and/or had a prehospital arrest, the next consideration is whether there are “signs of life”. These are denoted in the yellow footnote as spontaneous motor activity, respirations, reactive pupils, narrow complex ECG rhythm or organized cardiac activity on ultrasound (21-23). If the answer to ANY ONE of these questions is “yes”, then you move on to treating reversible causes of arrest as previously described. If ROSC is not immediately obtained, consideration of mechanism/ injury pattern is the next guiding principal to decide on an ERT. Simultaneously, PALS cardiac arrest algorithm suggestions like CPR, epinephrine and defibrillation should also be ongoing as per protocol (5). A wide variety of findings have been used to denote a “sign of life” in the literature, with no consensus in current pediatric guidelines (21-23). The above were chosen based on expert opinion via the three associations making this guideline as well as definitions from the US National Trauma Databank, several European pediatric guidelines, and the current EAST adult guidelines (5, 22, 24).
If a patient has no signs of life and clear signs of severe brain injury, early termination of resuscitation is recommended given the high associated mortality and likelihood of poor neurologic outcome. Multiple analyses have shown that the leading cause of death in children with cardiac arrest after blunt trauma was cranial or spinal cord injuries (8, 25, 26). One multicenter review showed that up to 88% of death secondary to cardiac arrest in pediatric trauma was secondary to cranial trauma (25). In fact, one more recent study reviewing head trauma as a cause of pediatric arrest showed that half of patients in this category were pronounced dead on arrival to ED (26). Of those who were admitted, all passed within a median of 24 hours (26). The sole survivor had persistent neurologic deficits.
If a patient remains asystolic/ no signs of life on arrival and resuscitation/ intervention for reversible causes of arrest does not yield any result, termination of resuscitation is recommended as survival after ERT is essentially nil (13, 14).
The decision to go forward with an ERT involves consideration of injury pattern/ mechanism and the presence of AT LEAST 1 sign of life (8, 13, 24, 27-29). If the patient has an isolated head injury ongoing medical resuscitation/ TBI management is reasonable, with an ERT not being indicated. If there is penetrating/ blunt truncal injury, that includes chest, abdomen and pelvic, an ERT can be considered. If there is isolated penetrating or blunt injury to an extremity, medical resuscitation, tourniquet, and wound packing are recommended and an ERT is not indicated (5).
The reasoning to avoid ERT in isolated head trauma was previously discussed and not recommended given the abysmal survival rates and likelihood of good neurologic outcome (8, 25, 26). The recommendation is to consider ERT in patients with blunt or penetrating truncal trauma/ unclear mechanism of injury given its potential benefit. This choice is interesting, given multiple decade long analyses of pediatric ERT patients indicate no survivors of those sustaining blunt trauma (8, 13, 14, 23). Many European guidelines also recommended using ERT only in penetrating truncal trauma (22). With respect to isolated extremity injuries, several of studies indicate there have been no survivors of ERT in the pediatric population who suffered isolated extremity injuries as the cause of arrest (8, 9, 12, 14).
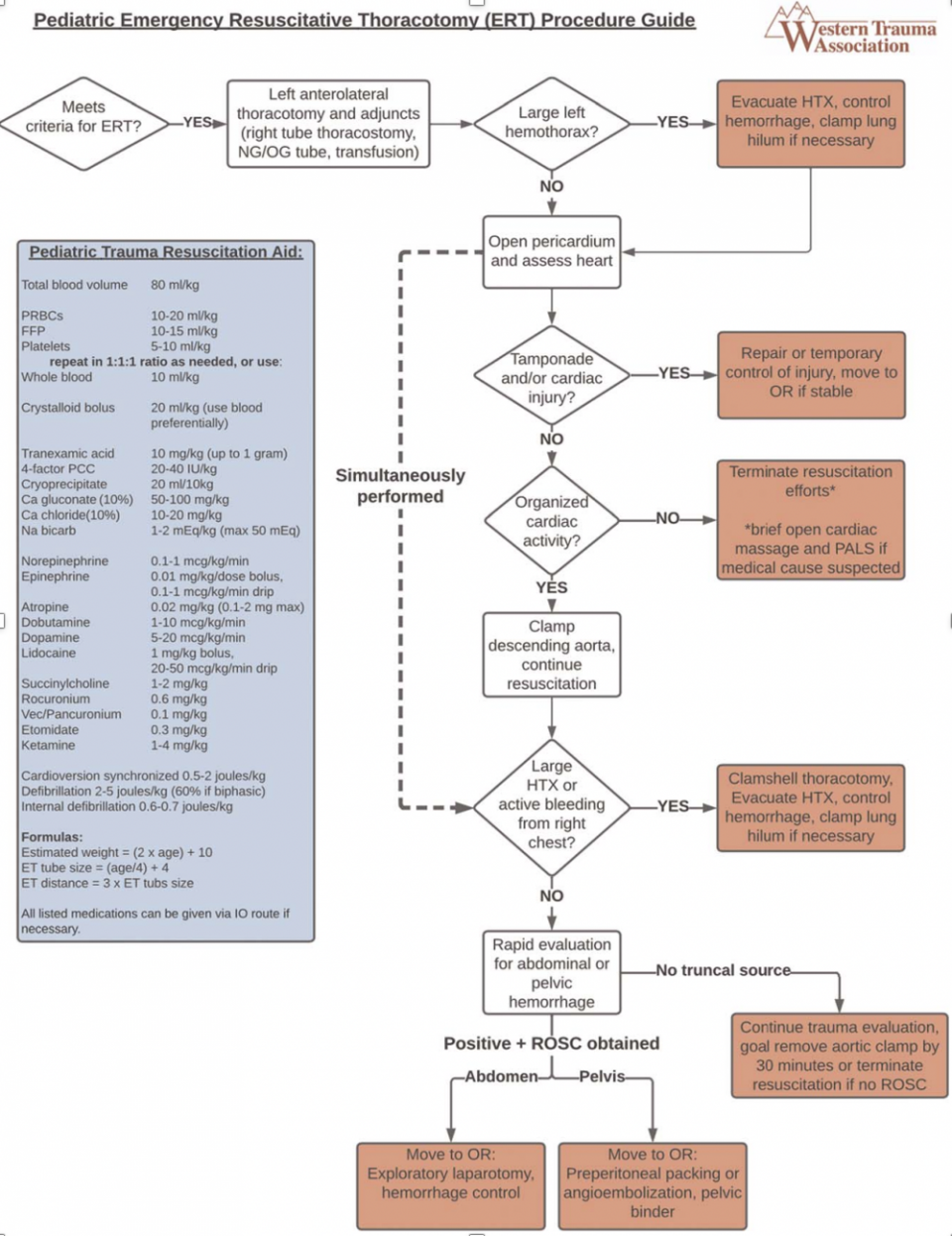
If a patient meets criteria for an ERT, the algorithm is shown here. Discussion on the intricacies of this algorithm is beyond the scope of this discussion. It is important to note that much of the presented algorithm is based heavily on surgeon preference and on a case-by-case basis, with many of these actions ongoing simultaneously (5).
Treatment of Reversible Causes of Arrest: Evidence
Hemorrhage – Give blood early
The primary REVERSIBLE causes of pediatric traumatic cardiac arrest include hemorrhage, pneumothorax, hemothorax and cardiac tamponade (5). As such, treatment of these reversible causes of arrest is essential to ensure optimization of a patient prior to consideration for an ERT (5).
Control of external hemorrhage is achieved via direct pressure, hemostatic dressings, and tourniquets for external or extremity bleeding (5). Giving blood products is beneficial to trauma patients given the leading cause of death is exsanguination (4, 5). There is also good evidence to suggest that aggressive crystalloid administration to pediatric trauma patients increases risk of mechanical ventilation, longer ICU stays and traumatic coagulopathy (30-32). Interestingly, there is an increasing body of adult literature that also suggests shorter times to receiving packed red blood cells and the use of whole blood (IE all components) decreases risk of death and reduces the risk of traumatic coagulopathy (33, 34).
A retrospective analysis of over 10 years and 14000 pediatric trauma patients from the US found that those who received an early ED blood product or pre-hospital blood product had significantly higher 24-hour survival and reduced in hospital mortality compared to those who received it later in their ED course (35). In fact, those treated with pre-hospital transfusion had significant better 24-hour survival/ in-hospital mortality than those who received blood first in the ED with no change in complication rates (35). Unfortunately, the data for using whole blood as the hemostatic product of choice in pediatrics is mixed (36). One propensity matched study from the US suggested that the use of whole blood rather than individual component therapy in pediatric trauma mass transfusions resulted in decreased overall transfusion requirements and number of ventilated days without having any mortality benefit (36).
Hemorrhage – TXA
Tranexamic acid (TXA) is a synthetic inhibitor that helps stabilize the fibrin matrix of a blood clot (37). The CRASH-2 trial in adults was clear that TXA improved survival from hemorrhagic shock in trauma, including arrest if given within 3 hours (37). This occurred while having no increased risk of thromboembolic events (37). There are currently no RCTs in pediatrics to support the adult findings. Eckert et al performed a pediatric analysis on trauma patients from a combat zone in Afghanistan and noted that administration of TXA improved in-hospital mortality (PED-TRAX study) (38). A more recent prospective civilian sub-analysis of pediatric resuscitation in Canada, Italy and the US showed that those receiving TXA had improved survival and bleeding profile (39). Of note, only half the patients in this analysis were secondary to trauma (39).
Pneumothorax
The presence of a chest injury in pediatric trauma is estimated to increase the risk of arrest by up to 10-fold (40). The adult standard of care for tension pneumothorax, massive haemothorax and traumatic cardiac arrest is now thoracostomy given the concerns that needle decompression is associated with higher failure rates and significant morbidity such as the failure of the needle to penetrate the pleura, catheter dislodgment and occlusion (41, 42). In fact, in a recent Australian case series reviewing needle chest decompression, all children receiving it required a definitive thoracostomy to ensure adequate chest decompression (40). They concluded thoracostomy had a low complication rate and a reasonable safety profile (40). Unfortunately, there is no clear data suggesting thoracostomies improve survival from traumatic cardiac arrest, though this in inferred given it is a more common cause of death.
Debrief!
As these events can be extremely challenging for health care providers, a debrief is always important. Debriefing also helps with looking at team performance and system level areas for improvement. Research has shown that immediate and structured debrief improved provider recall of events, improved wellbeing, mitigated provider emotional trauma and helped better identify sources of error (43-46).
Some Controversy..
Pediatric Termination of Resuscitaiton
It is commonly held knowledge that the longer a resuscitation, the poorer the outcomes (5, 6). It is important to recognize that termination of resuscitation (TOR) in pediatric patients is both challenging and controversial. Adult TOR guidelines typically include consideration of time of resuscitation >20 minutes, no bystander CPR, if the event was witnessed, no shock was delivered and if there was no ROSC on transport (27). The most recent guidelines from the AAP released in 2014 recommended CONSIDERATION for TOR if the patient had a traumatic cardiac arrest, has received more than 30 minutes of resuscitation without a pulse and is >30 minutes from the nearest facility (27). Interestingly, a recent analysis out of Japan reviewed the length of CPR by which patients could have both survival and a favourable neurologic outcome falls below 1%. In Japan, their EMS teams cannot do out-of-hospital TOR. That time frame was just 15 minutes with a specificity and positive predictive value of 100% (47). Many studies reviewed by the AAP TOR guidelines suggested that CPR time <5 minutes was more likely to have favourable neurologic outcomes (27). Given that the research is suggestive of shorter intervals of time for favourable outcomes, it will be interesting to see how guidelines may change. Although the concern for family experience and emotional processing of events pre-hospital versus in the ED are valid, they have yet to be formally studied (27).
CPR and Epinephrine: are they harmful?
CPR and epinephrine administration are both recommended as part of the ATLS algorithm for cardiac arrest (48). The research surrounding the role of CPR and epinephrine in pediatric traumatic cardiac arrest is conflicting. Physiologically speaking, forcing the heart to pump without blood to pump does not seem as though it would prove helpful. Despite CPR and epinephrine provided in the pre-hospital setting, traumatic arrest survival is poor as previously mentioned. One recent study showed that although epinephrine did increase the rate of ROSC/ post-ROSC BP and HR, it had no impact on long-term survival or favourable neurologic outcome in pediatric traumatic cardiac arrest patients (49). In fact, it was a risk factor for mortality (48). Another recent analysis also showed that those who survived a pediatric traumatic arrest received less doses of epinephrine while having a longer CPR time as well as having no effect on favourable neurologic outcomes (50). Although this was noted here, it is a general literary consensus that shorter resuscitation time itself typical improves survival outcomes. A recent literature review to assess if CPR was helpful in pediatric traumatic cardiac arrest revealed no trials to support or deny its use (51). Although there appears to be some ambiguity, the evidence for both CPR and epi for non-traumatic cardiac arrest is clear and as such it is still the recommended path for trauma pending further research.
Societal Cost
Lastly, I wanted to briefly highly another point of view. Given the low yield of ERT in pediatrics, the question of its societal utility and resource allocation can be made. One study noted 51% of ERTs completed were deemed unnecessary (no survivors or organ donation) resulting in considerable resource use (335 units pRBCs and OR visits) (52). There is also an increased incidence of needle stick injuries by up to 7.2% in health care providers during an ERT (53). Given the ongoing personnel and blood shortages, the use of this technique needs to be extremely selective.
Summary
Pediatric traumatic cardiac arrest is a trying issue. The events are low in frequency yet have high acuity. The use of emergency resuscitative thoracotomy in pediatrics is more selective to those patients with signs of life and truncal trauma in comparison to adults. As such, the algorithms used for these cases involves usual PALS cardiac arrest algorithms with a focus on treating reversible causes of arrest in trauma including hemorrhage, pneumothorax, hemothorax and cardiac tamponade. The evidence for many of these recommendations is poor. Therefore, a guideline is just a recommendation, and each patient must be looked at with a case-by-case basis. Further research needs to be conducted surrounding the use of many of our interventions in the pediatric setting to best provide care and improve outcomes.
References
- Cunningham RM, Walton MA, Carter PM. The major causes of death in children and adolescents in the United States. N Engl J Med. 2018;379(25):2468–75.
- Centers for Disease Control and Prevention, National Center for Health Statistics. Underlying Cause of Death 1999–2020 on CDC WONDER Online Database, released in 2021. Data are from the Multiple Cause of Death Files, 1999–2020, as compiled from data provided by the 57 vital statistics jurisdictions through the Vital Statistics Cooperative Program. Accessed at http://wonder.cdc. gov/ucd-icd10.html. Accessed date 17 Oct 2023.
- Hewes HA, Ely M, Richards R, Shah MI, Busch S, Pilkey D et al. Ready for children: assessing pediatric care coordination and psychomotor skills evaluation in the prehospital setting. Prehospital emergency care. 2019; 23: 510-8.
- Jeruzal JN, Boland LL, Frazer MS, Kamrud JW, Myers RN Lick CJ et al. Emergency medical services providers perspectives on pediatric calls: A qualitative study. Prehospital emergency care [Internet]. 2019; 23(4): 501-9.
- Martin MJ, Brasel KJ, Brown CVR, Hartwell JL, de Moya M, Inaba K et al. Pediatric emergency resuscitative thoracotomy: A Western Trauma Association, Pediatric Trauma Society, and Eastern Association for the Surgery of Trauma collaborative critical decisions algorithm. J Trauma Acute Care Surg. 2023;95(4):583-591. doi: 10.1097/TA.0000000000004055. Epub 2023. PMID: 37337331.
- Tawil CE, Leblanc PA, Beno S, Nemeth J. Traumatic cardiac arrest: unique considerations or the pediatric patient. Can J Emerg Med. 2022;24:457-458.
- Allen CJ, Valle EJ, Thorson CM, Hogan AR, Perez EA, Namias N, et al. Pediatric emergency department thoracotomy: a large case series and systematic review. J Pediatr Surg. 2015;50(1):177–181.
- Moore HB, Moore EE, Bensard DD. Pediatric emergency department thoracotomy: a 40-year review. J Pediatr Surg. 2016;51(2):315–3.
- Prieto JM, Van Gent JM, Calvo RY, Rooney AS, Martin MJ, Sise MJ, et al. Nationwide analysis of resuscitative thoracotomy in pediatric trauma: time to differentiate from adult guidelines? J Trauma Acute Care Surg. 2020;89(4): 686–690.
- Hanafi M, Al-Sarraf N, Sharaf H, Abdelaziz A. Pattern and presentation of blunt chest trauma among different age groups. Asian Cardiovasc Thorac Ann. 2011;19(1):48–51.
- Mollberg NM, Tabachnick D, Lin FJ, Merlotti GJ, Varghese TK, Arensman RM, et al. Age-associated impact on presentation and outcome for penetrating thoracic trauma in the adult and pediatric patient populations. J Trauma Acute Care Surg. 2014;76(2):273–277; discussion 7-8.
- Swendiman RA, Sharoky CE, Russell KW, Goldshore MA, Blinman TA, Nance ML. Life-saving interventions in pediatric trauma: a National Trauma Data Bank experience. J Trauma Acute Care Surg. 2019;87(6):1321–1327.
- Moskowitz EE, Burlew CC, Kulungowski AM, Bensard DD. Survival after emergency department thoracotomy in the pediatric trauma population: a review of published data. Pediatr Surg Int. 2018;34(8):857–860
- Flynn-O’Brien KT, Stewart BT, Fallat ME, Maier RV, Arbabi S, Rivara FP, et al. Mortality after emergency department thoracotomy for pediatric blunt trauma: analysis of the National Trauma Data Bank 2007-2012. J Pediatr Surg. 2016;51(1):163–167.
- Kissoon N, Dreyer J, Walia M. Pediatric trauma: differences in pathophysiology, injury patterns and treatment compared with adult trauma. CMAJ. 1990; 142(1):27–34
- McLaughlin C, Zagory JA, Fenlon M, Park C, Lane CJ, Meeker D, et al. Timing of mortality in pediatric trauma patients: a National Trauma Data Bank analysis. J Pediatr Surg. 2018;53(2):344–351.
- Brenner M, Zakhary B, Coimbra R, Morrison J, Scalea T, Moore LJ, et al. Resuscitative endovascular balloon occlusion of the aorta (REBOA) may be superior to resuscitative thoracotomy (RT) in patients with traumatic brain injury (TBI). Trauma Surg Acute Care Open. 2022;7(1):e000715.
- Theodorou CM, Trappey AF, Beyer CA, Yamashiro KJ, Hirose S, Galante JM, et al. Quantifying the need for pediatric REBOA: a gap analysis. J Pediatr Surg. 2021;56(8):1395–1400.
- Campagna GA, Cunningham ME, Hernandez JA, Chau A, Vogel AM, NaikMathuria BJ. The utility and promise of resuscitative endovascular balloon occlusion of the aorta (REBOA) in the pediatric population: an evidencebased review. J Pediatr Surg. 2020;55(10):2128–2133.\
- Power A, Parekh A, Scallan O, Smith S, Novick T, Parry N, et al. Size matters: first-in-human study of a novel 4 French REBOA device. Trauma Surg Acute Care Open. 2021;6(1):e000617.
- Harris M, Crowe RP, Anders J, D’Acunto S, Adelgais KM, Fishe JN. Identification of factors associated with return of spontaneous circulation after pediatric out-of-hospital cardiac arrest using natural language processing. Prehosp Emerg Care. 2022;1–8.
- Rickard AC, Vassallo J, Nutbeam T, Lyttle MD, Maconochie IK, Enki DG, et al. Paediatric traumatic cardiac arrest: a Delphi study to establish consensus on definition and management. Emerg Med J. 2018;35(7):434–439.
- Wyrick DL, Dassinger MS, Bozeman AP, Porter A, Maxson RT. Hemodynamic variables predict outcome of emergency thoracotomy in the pediatric trauma population. J Pediatr Surg. 2014;49(9):1382–1384.
- Seamon MJ, Haut ER, Van Arendonk K, Barbosa RR, Chiu WC, Dente CJ, et al. An evidence-based approach to patient selection for emergency department thoracotomy: a practice management guideline from the Eastern Association for the Surgery of Trauma. J Trauma Acute Care Surg. 2015;79(1): 159–173.
- Lin YR, Wu HP, Huang CY, Chang YJ, Lin CY, Chou CC. Significant factors in predicting sustained ROSC in paediatric patients with traumatic out-of-hospital cardiac arrest admitted to the emergency department. Resuscitation.2007;74:83–89.
- Calkins CM, Bensard DD, Partrick DA, Karrer FM. A critical analysis of outcome for children sustaining cardiac arrest after blunt trauma. J Pediatr Surg.2002;37:180–184.
- Oruc M, Ulku R. Evaluation of factors affecting prognosis in penetrating thoracic injuries. Turk Gogus Kalp Damar Cerrahisi Derg. 2018;26(4):598–605.
- Duron V, Burke RV, Bliss D, Ford HR, Upperman JS. Survival of pediatric blunt trauma patients presenting with no signs of life in the field. J Trauma Acute Care Surg. 2014;77(3):422–426.
- Withholding or termination of resuscitation in pediatric out-of-hospital traumatic cardiopulmonary arrest. American College of Surgeons Committee on Trauma; American College of Emergency Physicians Pediatric Emergency Medicine Committee; National Association of Ems Physicians; American Academy of Pediatrics Committee on Pediatric Emergency Medicine, , Fallat ME. Withholding or termination of resuscitation in pediatric out-of-hospital traumatic cardiopulmonary arrest. Pediatrics. 2014;133(4):e1104–e1116.
- Polites SF, Moody S, Williams RF, et al. Timing and volume of crystalloid and blood products in pediatric trauma: an Eastern Association for the Surgery of Trauma multicenter prospective observational study.J Trauma Acute Care Surg. 2020;89(1):36-42. doi:10.1097/TA. 0000000000002702
- Polites SF, Nygaard RM, Reddy PN, et al. Multicenter study of crystalloid boluses and transfusion in pediatric trauma—when to go to blood? J Trauma Acute Care Surg. 2018;85(1):108- 112. doi:10.1097/TA.0000000000001897
- Schauer SG, April MD, Becker TE, Cap AP, Borgman MA. High crystalloid volumes negate benefit of hemostatic resuscitation in pediatric wartime trauma casualties. J Trauma Acute Care Surg. 2020;89(2S)(suppl 2):S185-S191. doi:10.1097/ TA.0000000000002590
- Powell EK, Hinckley WR, Gottula A, Hart KW, Lindsell CJ, McMullan JT. Shorter times to packed red blood cell transfusion are associated with decreased risk of death in traumatically injured patients. J Trauma Acute Care Surg. 2016;81(3):458-462. doi:1097/TA.0000000000001078
- Sheppard FR, Schaub LJ, Cap AP, et al. Whole blood mitigates the acute coagulopathy of trauma and avoids the coagulopathy of crystalloid resuscitation. J Trauma Acute Care Surg. 2018;85(6):1055-1062. doi:1097/TA.0000000000002046
- Morgan KM, Abou-Khalil E, Strotmeyer S, Richardson WM, Gaines BA, Leeper CM. Association of prehospital transfusion with mortality in pediatric trauma.JAMA Pediatr. Published online May 22, 2023. doi:10.1001/jamapediatrics.2023.1291
- Anand T, Obaid O, Nelson A, Chehab M, Ditillo M, Hammad A, Douglas M, Bible L, Joseph B. Whole blood hemostatic resuscitation in pediatric trauma: A nationwide propensity-matched analysis. J Trauma Acute Care Surg. 2021 Oct 1;91(4):573-578. doi: 10.1097/TA.0000000000003306. PMID: 34086658.
- Shakur H, Roberts I, Bautista R, Caballero J, Coats T, CRASH-2_Trial_Collaborators, , et al. Effects of tranexamic acid on death, vascular occlusive events, and blood transfusion in trauma patients with significant haemorrhage (CRASH-2): a randomised, placebo-controlled trial. Lancet 2010; 376(9734):23–32.
- Eckert MJ, Wertin TM, Tyner SD, Nelson DW, Izenberg S, Martin MJ. Tranexamic acid administration to pediatric trauma patients in a combat setting: the pediatric trauma and tranexamic acid study (PED-TRAX). J Trauma Acute Care Surg. 2014;77(6):852–858 discussion 8.
- Leonard JC, Josephson CD, Luther JF, Wisniewski SR, Allen C, Chiusolo F, et al. Life-threatening bleeding in children: a prospective observational study. Crit Care Med. 2021;49(11):1943–1954.
- Quinn N, Palmer CS, Bernard S, Noonan M, Teague WJ. Thoracostomy in children with severe trauma: An overview of the paediatric experience in Victoria, Australia. Emerg Med Australas. 2020 Feb;32(1):117-126. doi: 10.1111/1742-6723.13392. Epub 2019 Sep 18. PMID: 31531952.
- Kaserer A, Stein P, Simmen HP, Spahn DR, Neuhaus V. Failure rate of prehospital chest decompression after severe thoracic trauma. Am. J. Emerg. Med. 2017; 35: 469–74.
- Laan DV, Vu TD, Thiels CA et al. Chest wall thickness and decompression failure: a systematic review and meta-analysis comparing anatomic locations in needle thoracostomy. Injury 2016; 47: 797–804.
- Mullan PC, Cochrane NH, Chamberlain JM, Burd RS, Brown FD, Zinns LE, et al. Accuracy of postresuscitation team debriefings in a pediatric emergency department. Ann Emerg Med. 2017;70(3):311–319.
- Slater PJ, Edwards RM, Badat AA. Evaluation of a staff well-being program in a pediatric oncology, hematology, and palliative care services group. J Healthc Leadersh. 2018;10:67–85.
- Ugwu CV, Medows M, Don-Pedro D, Chan J. Critical event debriefing in a community hospital. Cureus. 2020;12(6):e8822.
- Zhang YH, De Silva MWS, Allen JC Jr., Lateef F, Omar EB. End-of-life communication in the emergency department: the emergency physicians’ perspectives. J Emerg Trauma Shock. 2022;15(1):29–34.
- Shibahashi K, Sugiyama K, Hamabe Y. Pediatric out-of-hospital traumatic cardiopulmonary arrest after traffic accidents and termination of resuscitation. Ann Emerg Med. 2020;75(1): 57-65.
- Galvagno SM Jr, Nahmias JT, Young DA. Advanced Trauma Life Support Update 2019: Management and Applications for Adults and Special Populations. Anesthesiol Clin. 2019;37(1):13-32. doi: 10.1016/j.anclin.2018.09.009. Epub 2018 Dec 27. PMID: 30711226.
- Lin YR, Wu MH, Chen TY, et al. Time to epinephrine treatment is associated with the risk of mortality in children who achieve sustained ROSC after traumatic out-of-hospital cardiac arrest. Crit Care. 2019;23(1):101.
- Lelak KA, Arora R, Mowbray FI, Arkatkar A, Krouse C, Cloutier D et al. Cardiopulmonary resuscitation and epinephrine use in pediatric traumatic cardiac arrest. Am Surg. 2023;89(6):2965-8.
- Bowles, Fiona, and Kat Rawlinson. “BET 3: The Efficacy of Chest Compressions in Paediatric Traumatic Arrest.” Emergency Medicine Journal : EMJ, vol. 33, no. 5, 2016, pp. 368–368, https://doi.org/10.1136/emermed-2016-205807.3.
- Passos EM, Engels PT, Doyle JD, Beckett A, Nascimento B Jr., Rizoli SB, et al. Societal costs of inappropriate emergency department thoracotomy. J Am Coll Surg. 2012;214(1):18–25.
- Nunn A, Prakash P, Inaba K, Escalante A, Maher Z, Yamaguchi S, et al. Occupational exposure during emergency department thoracotomy: a prospective, multi-institution study. J Trauma Acute Care Surg. 2018; 85(1):78–84.