At this month’s PTM Journal Club, two topics were covered; the use of intranasal (IN) glucagon for hypoglycemia and preshospital triage of chest pain patients. Haamid et al. (2023) conducted a retrospective chart review to determine if the use of intranasal glucagon could be used as a substitute for intramuscular glucagon, and/or oral or intravenous dextrose. While in the ARTICA trial, Aarts et al., aimed to determine if prehospital triage of chest pain patients using the HEART score and troponin measurement was a viable diversion alternative to the overcrowding currently experienced in North American healthcare systems.
As a side note, check-out the fantastic critical commentary by Dr. James Worrall’s on the HEART Score by clicking here.
Pre-Hospital Intranasal Glucagon for Hypoglycemia
Background and Objectives
Around 3 million people are living with diabetes in Canada. Hypoglycemia ranges from 0.8 to 3.2 events per person-year, and annual prevalence ranges from 10% to 53% for type 1 diabetes and from 4.4% to 6% for type 2 diabetes. In a pre-hospital setting, hypoglycemia is treated with oral or intravenous dextrose and intramuscular glucagon is administered without vascular access. However, occupational needle-stick injury remains a significant risk for paramedics. Thus, the authors sought to determine if intranasal glucagon effectively treats hypoglycemia.
Methods
This study conducted a retrospective chart review of 44 cases from January 2015 through December 2020 of adults >18 years old where paramedics solely administered intranasal glucagon in a pre-hospital setting for hypoglycemia. The primary outcome was the clinical response to IN glucagon, which the lead paramedic documented as “no improvement”, “slight improvement”, or “substantial improvement” in mental status. Secondary outcomes included pre- and post-administration blood glucose values. Below, you will see the results:
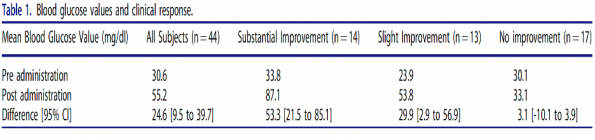
Authors’ Conclusions
Intranasal glucagon for hypoglycemia resulted in a clinically significant improvement in mental status and a corresponding increase in blood glucose levels in select pre-hospital settings.
Limitations
This study was a retrospective chart review with a small sample size. Furthermore, the primary outcome of clinical improvement in mental status was poorly defined, leaving room for personal interpretation. Despite their conclusions of clinical improvement in mental status, there was still a large proportion of patients who made only a slight vs no improvement in their clinical response. The limited improvement in mental status is concerning. I suspect these individuals had limited hepatic glycogen stores as a possible explanation for their level of improvement. This is why understanding the subtypes of patient populations or presentations who benefited from glucagon administration would likely have yielded stronger conclusions; which unfortunately was not reported in this study.
Bottom Line
Intranasal glucagon is effective in patients with adequate hepatic glycogen stores. Consider first-line in patients with hypoglycemia to limit needle-stick injuries, but administering dextrose must be considered if there is no clinical improvement.
Citation
Rule-out of non-ST-segment elevation acute coronary syndrome by a single, pre-hospital troponin measurement: a randomized trial
Background and Objectives
Overcrowding in the emergency department is an ongoing issues, and patients with chest pain are often transferred to the ED after initial assessment by paramedics. However, current guidelines suggest that around 80-90% of patients do not have acute coronary syndrome. These visits include costly additional testing and often result in prolonged in-hospital stays. Thus, the authors sought to determine if pre-hospital risk stratification of chest pain using the HEART score and single troponin measurement would reduce healthcare costs and major adverse cardiac events (MACE).
Methods
The objective of the ARTICA study was to determine the 30-day healthcare costs and incidence of major adverse cardiac events of pre-hospital rule-out of non-ST-segment elevation acute coronary syndrome (NSTE-ACS) with a single point-of-care troponin in low-risk patients (HEART score ≤3 with at least 2 hours of chest pain), as compared to transfer to the emergency department (ED). From March 2019 to May 2022, 866 participants were randomized to either the pre-hospital or ED group. The primary outcome was health care costs at 30 days, and the secondary outcome was the incidence of MACE at 30 days. Below, you will see a summary of the methods and results:
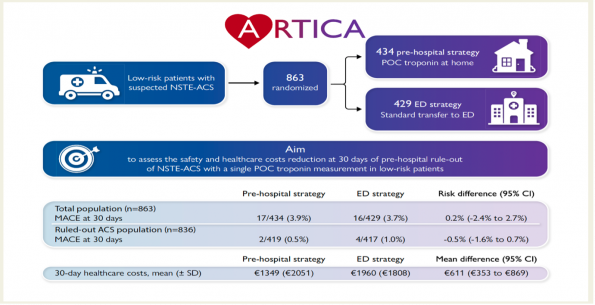
Authors’ Conclusions
Pre-hospital rule-out of ACS with a POC troponin measurement in low-risk patients significantly reduces healthcare costs, while the overall incidence of MACE was low in both control and experimental groups.
Limitations
Although the study was well-conducted, it unfortunately was underpowered to draw the conclusion that a pre-hospital rule-out strategy is non-inferior then transfer to the ED, based on safety. The authors suggest needing around 18,000 people to be adequately powered to assess for MACE. another important difference to consider is that this study was performed in the Netherlands; where EMS providers are critical care nurses, differing wildly from Canadian EMS providers. It is also important to consider that chest pain is more than ACS, and emergency providers can refer patients for appropriate outpatient testing, which may provide long-term benefits. Lastly, troponin testing in the pre-hospital setting is less precise and cannot detect small concentrations of troponin compared to the analyzers used within the hospital.
Bottom Line
Point of care (POC) troponin testing in patients with low-risk chest pain reduces healthcare costs, and although underpowered, the 30-day MACE was low ([0.5% (2/419) vs 1.0% (4/417)], with a risk difference of -0.5% (95% CI – 1.6%-0.7%; P=0.41). With paramedicine changing in Canada, I believe there is a utility for POC troponin testing for low-risk patients in the future. However, further studies assessing for MACE are required.
Citation


