Aortic Stenosis is the most common valvular heart disease in Canada. In fact, after hypertension and coronary artery disease, it is the third most common cardiovascular condition. The prevalence of valvular heart disease increases sharply with age. Approximately 1 in 10 of your patients over the age of 75 has aortic stenosis.
The most common cause of AS in Canada is degenerative valve disease of advanced age, followed by congenital bicuspid aortic valve disease, and finally rheumatic heart disease. But worldwide, rheumatic heart disease remains the leading cause of AS, and is still the #1 cause of heart disease overall in children and young adults. It also remains prevalent among indigenous populations in resource-rich countries.
Why else should we care? In addition to its high prevalence world-wide, aortic stenosis is associated with high mortality. Despite a seemingly benign and asymptomatic latent period during which the disease progresses silently over 20-30 years, once symptoms develop, the prognosis changes dramatically.
Without prompt intervention in the form of aortic valve replacement, average survival is only 2-3 years, with a high risk of sudden death. Therefore, the development of symptoms is a critical point in the natural history of Aortic Stenosis.
Unfortunately, it is not uncommon for the early symptoms of AS to be falsely attributed to other causes, including the normal process of aging.
A simple example of this is Mrs. Smith, a 72F who has a hx of smoking. Her family physician prescribed her a puffer last year when she developed mild dyspnea. She presents to your ED with fatigue and breathlessness on exertion. Labs are normal, CXR is normal. Her lungs sound decreased throughout. The ED is loud, but you think you heard a murmur. You treat her for a COPD exacerbation (the same as on her last 2 visits), and you send her home. But what if you’ve missed the diagnosis of Severe Aortic Stenosis? The consequences of this could be fatal.
In this post we will focus on the physiologic and hemodynamic consequences of aortic stenosis, the management of AS, and the approach to patients with atrial fibrillation and those who require procedural sedation. Part two will emphasize the management of decompensated aortic stenosis and cardiogenic shock.
Definitions
For the remainder of this post, we will be focusing on severe aortic stenosis, which is defined as a cross-sectional aortic valve opening area that is less than 1 cm2. For reference, the normal adult aortic valve has an area of 3.0 to 4.0 cm2.
You may have heard of other terms, like “critical AS”, with varying cutoffs for the valve area, but for the purpose of this post, and per the current guidelines, we will use < 1 cm2 as the number to remember.
One final note on this: The diagnosis of severe aortic stenosis is nuanced. There are other measurements commonly used in the definition, including pressure gradients and peak aortic jet velocity, but it should be noted that, within the many subclassifications of AS, there is variation in those criteria. This is beyond the scope of today’s post.
But remember this: valve area does not always correlate with symptoms; although AS is an asymptomatic disease until it becomes severe, patients with severe stenosis can remain asymptomatic for some time.
In addition to this, severe AS does not always mean a reduced LV ejection fraction, though in the majority of patients with severe, symptomatic disease, this will be the case.
Consequences of Aortic Stenosis
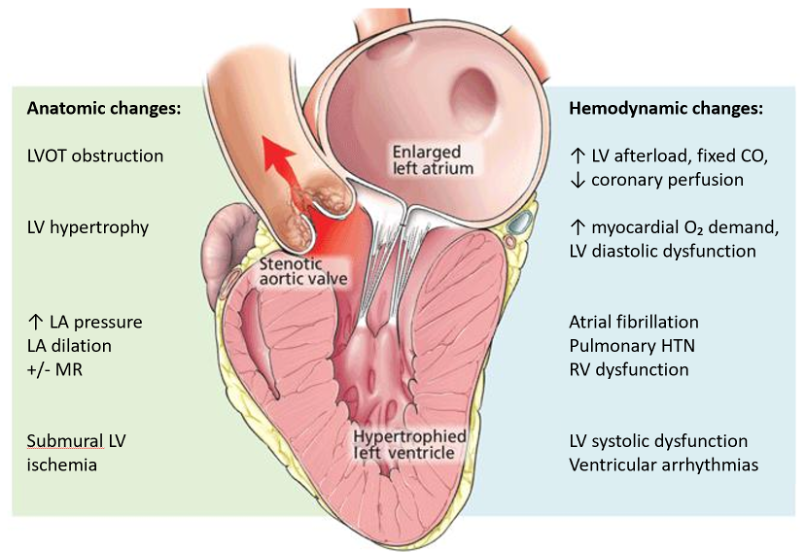
There are many anatomic and hemodynamic consequences of AS, but the illustration below highlights the key points relevant to us in the ED:
The classic triad of symptoms that represent severe aortic stenosis can be remembered by the SAD mnemonic – SYNCOPE, ANGINA, and DYSPNEA. These symptoms are almost always exertional, as the LVOT obstruction and relatively fixed cardiac output is essentially unable to compensate for any increased oxygen demand that comes from physical exertion.
Syncope is caused by low cardiac output or episodes of arrhythmia, chest pain results from low cardiac output, compounded by exertional tachycardia, while dyspnea is multifactorial: a result of inadequate forward flow, low coronary perfusion and possible pulmonary congestion.
But it is important to note that patients may not present with the classic triad of symptoms, particularly early in the symptomatic phase. Instead, vague exercise intolerance and exertional dizziness is often described. Atrial fibrillation is common, and we will chat more about its significance in a bit.
Rarely, patients may present with mucosal, GI, and skin bleeding, which is a result of Heyde syndrome – an acquired vWF deficiency from mechanical destruction at the stenotic valve.
At the end of the spectrum, patients may present in acutely decompensated heart failure or cardiogenic shock if myocardial ischemia and dysfunction trigger a downward hemodynamic spiral, leading to severe pump failure with impaired forward flow. Sudden death can occur, with an annual incidence estimated as high as 34%.
Management of Aortic Stenosis
Unfortunately, no medical therapies influence the natural history of aortic stenosis. Aortic valve replacement (AVR), remains, to date, the only therapeutic option to improve the survival of patients with symptomatic severe AS.
Luckily, we have made incredible advances in this area in the last 2 decades, with the first TAVI (transcatheter aortic valve implantation) performed in 2002, revolutionizing the treatment of AS and providing a minimally invasive option for patients who may not have otherwise been candidates for traditional surgery.
Like the definition of severe AS, the choice and timing of intervention is nuanced. However, per the practice guidelines on the management of valvular heart disease from the American Heart Association and the European Society of Cardiology, the general indication for AVR is severe AS that is either symptomatic and/or associated with left ventricular (LV) systolic dysfunction (1,2).
Aortic valve replacement, either by surgical or transcatheter approach, is an excellent option for many patients that develop severe AS, and following these definitive interventions, age-corrected survival returns to near-normal.
Despite the advances in invasive management, however, there have been no strides made in the medical management of aortic stenosis. So, let’s take a deep dive together into what we can do for these patients when they present to the Emergency Department.
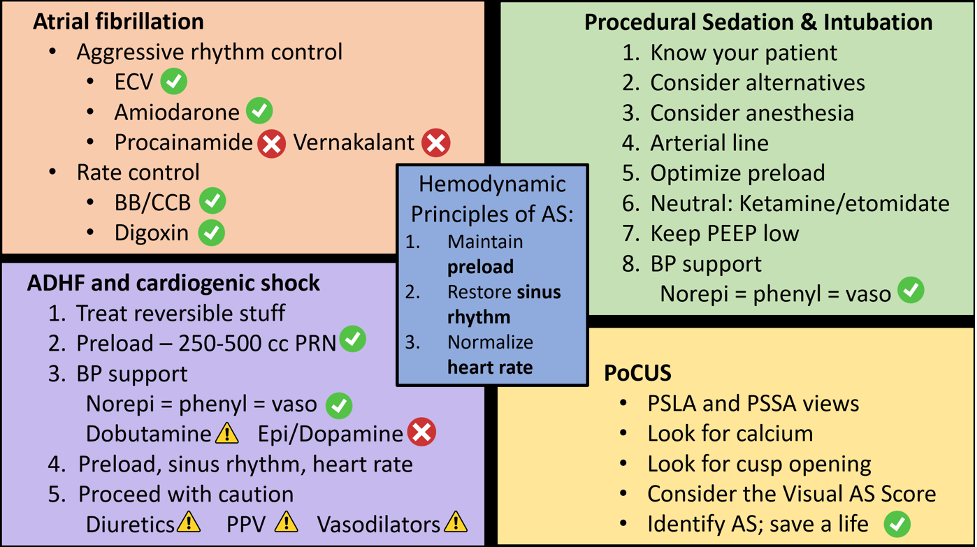
Hemodynamic Principles of AS Management
Let’s start with a review of the three golden rules of AS management, which represent our cornerstones of resuscitation:
- Maintain adequate preload: Patients with severe AS are preload dependent. Due to LVH and impaired diastolic function, they require higher preload to fill the non-compliant left ventricle. Remember, hypotension is very dangerous here!
- Restore sinus rhythm: For adequate filling, the non-compliant LV relies heavily on atrial kick, which accounts for 20-40% of cardiac output. In any rhythm other than a sinus rhythm, loss of the atrial kick will mean a significant drop in LV preload, which, as we’ve already learned, can be detrimental in AS.
- Ensure a normal heart rate: Excessive tachycardia not only promotes ischemia in the big LV that is already prone to mismatch in O2 supply and demand, but it also reduces time spent in diastole, translating once again to a reduction in LV preload and coronary artery perfusion. On the other hand, significant bradycardia may also be poorly tolerated as it causes a drop in cardiac output.
Let’s now talk about the management of AS in a few common ED scenarios:
- Atrial fibrillation
- Procedural sedation and intubation
Aortic Stenosis and Atrial Fibrillation
Atrial fibrillation is both common and potentially dangerous in patients with aortic stenosis. In a study out of Switzerland (3) that looked at patients scheduled to undergo a TAVI procedure, 34% of patients with AS experienced a.fib, either paroxysmal or permanent. In comparison, the estimated global prevalence of a.fib is 1-2% of the general population, increasing to 10% of those over the age of 80. The first episode of A Fib in a patient with aortic stenosis may be a sign of progression to severe, symptomatic disease, therefore making it a significant turning point in the natural history of AS.
Current guidelines fail to address the significant relationship between arrhythmias and valvular heart disease (4).
In addition, there is very limited information on aortic stenosis in the publications that guide our ED management of arrhythmias, including the CCS and CAEP guidelines on the acute management of afib and flutter (5,6). (NOTE: As a reminder – Aortic Stenosis is NOT included in the term “valvular heart disease” as it pertains to the CAEP Afib/flutter Guidelines; this is an easy point of confusion].
A 2012 review by Darby and DiMarco (7) is one of the only publications we currently have on the management of a.fib in valvular heart disease, specifically AS. They emphasize that patients with severe AS are vulnerable and benefit from early restoration of sinus rhythm, β-Blockers and calcium channel blockers are first line for rate control, but amiodarone is the preferred agent in left ventricular hypertrophy. Finally, they reiterate that catheter or surgical ablation are important options if antiarrhythmics fail.
Picture this clinical scenario: Mrs. Fields is a 78 year old female who presents to your ED in a.fib with RVR. Her HR is 145 and her BP is 90/50. She looks unwell. On history, you discover she is known for aortic stenosis, currently being assessed for a TAVI procedure. She is also known for paroxysmal atrial fibrillation, and her smart watch alerted her today that she was fast and irregular once again. She is on a DOAC. On ROS, you identify that she has been unwell for the past week with a viral-sounding illness, with GI upset and low PO intake. On exam, she appears hypovolemic.
As Emergency physicians, we know that many patients present with atrial fibrillation that is secondary to an underlying illness. In these patients, cardioversion is usually avoided due to the potential for harm, and aggressive rate control may compromise the patient’s compensatory abilities for increasing cardiac output (8). So, in most patients presenting with secondary a.fib, we would probably agree to treat the underlying cause rather than the arrhythmia, reaching for IV fluids or antibiotics rather than setting up for cardioversion.
But does that concept of compensatory tachycardia still hold true for patients like Mrs. Fields, who have severe AS? What if her dehydration is only mild, but the real problem is that her tachycardia and atrial fibrillation are tipping her AS hemodynamics over the edge? Certainly, reversible contributing factors should be identified and addressed, but what about rhythm control?
In keeping with the second hemodynamic principle of aortic stenosis (“maintain sinus rhythm”), expert consensus here is yes, rhythm control should be strongly considered in severe aortic stenosis. This is particularly true if there is hemodynamic instability despite the correction of reversible factors.
So how do we do this?
In the unstable patient, electrical cardioversion is always an option and should be considered. However, options for safe chemical cardioversion in severe aortic stenosis are limited. Procainamide and vernakalant are contraindicated, given their negative inotropy and vasodilator properties that often lead to hypotension, which would be dangerous here. Given that most patients with severe AS will have some degree of LV systolic dysfunction, amiodarone is the only agent that is universally appropriate for rhythm control in severe AS (7).
So, for rhythm control options in AS, consider electrical cardioversion, or reach for Amiodarone.
Finally, what about in the patient who is not a candidate for rhythm control due to the duration of their arrhythmia or lack of anticoagulation, or if rhythm control is unsuccessful?
There are no tricks here – follow your usual rate control algorithm:
- If there is normal LV systolic function and no acute HF, beta blockers and calcium channel blockers are both safe options
- If there is evidence of reduced LVEF or signs of acute heart failure, Digoxin is your best bet
Procedural Sedation and Intubation in Severe AS
These two procedures are well within the wheelhouse of emergency medicine, but things get complicated when severe AS is added to the mix.
In review of the cardiology and anesthesiology literature on general anesthesia and surgery in AS, combined with the ED and anesthesia literature on intubation and procedural sedation, a few themes emerge:
- Most medical interventions that alter hemodynamic conditions carry the risk of destabilizing the patient with AS
- Most adverse events occur because the diagnosis of AS is not known to the team carrying out the procedure
- Anesthesia consultation should be considered for procedures involving severe AS
- And finally, this last point is a big one: the degree of risk increases significantly when patients with AS are symptomatic of their disease. As such, the AHA currently recommends that all patients with symptomatic severe AS have all elective procedures postponed until AVR can first be performed (1).
But, in patients with asymptomatic severe AS, the guidelines are much less clear. It is difficult to predict which patients with asymptomatic disease will experience clinical deterioration in these scenarios. But at the very least, this condition should make us pause.
Now, we don’t do many elective procedures in the ED. Instead, we are often forced to act, even when conditions are less than ideal. So, with all of that said, here is an approach that may help you when making nerve-wracking decisions around procedural sedation and intubation in the context of AS in the ED…
- Most importantly: Know your patient
- Review their recent Echo, if they have one, paying particular attention to the severity of their stenosis and their LV systolic function
- Ask the patient about their symptoms – again, symptomatic stenosis carries a greater risk of complications than asymptomatic disease
- What are their comorbidities? Any additional cardio-pulmonary disease is an added risk factor for destabilization
- Finally, perform an exam. This is particularly important if you are not sure about the severity of the patient’s AS
- Assess the murmur and palpate the carotid – remembering the clinical features that suggest severe stenosis (radiation to the neck, loss of the 2nd heart sound, and a weak carotid pulse)
- Assess the patient’s volume status
- Consider performing a bedside ultrasound (we’ll come back to that in a bit)
- Consider alternatives. Ask yourself: is this procedure necessary in the ED today? Is there an appropriate alternate approach? Examples of this could include a peripheral nerve block or hematoma block instead of procedural sedation, or a trial of NIPPV or an awake intubation, instead of an RSI.
- Consider anesthesia consultation. Our number one priority is patient safety, so should we be the ones doing this procedure? Should we call for help?
- Consider invasive monitoring. An arterial line will provide real-time BP monitoring in a patient who is particularly vulnerable to hypotension.
- Optimize preload. Going back to our golden rule, if the patient does not appear volume overloaded, always consider a pre-induction bolus of IV fluids.
- Choose hemodynamically stable agents for sedation and induction. Ketamine and etomidate are quoted most often in the literature.
- If giving PPV, keep the PEEP low, if possible.
- Be ready to support the blood pressure.
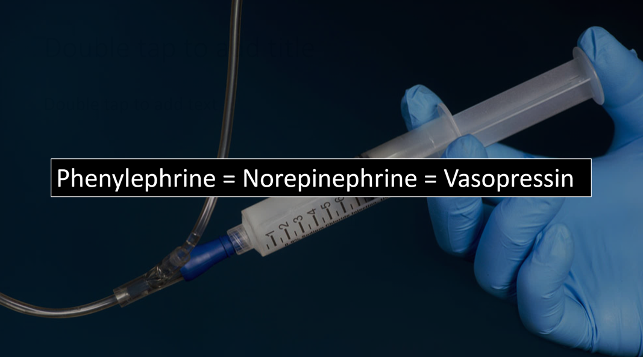
- NOTE: The AHA currently suggests phenylephrine or vasopressin as first-line vasopressors in aortic stenosis, but they do not reference any literature supporting this (9).
- One potential advantage of phenylephrine over other agents is that it may decrease HR, which as you recall, is helpful in AS. It is also particularly useful in push-dose form for transient BP support, such as when counteracting the vasodilatory effects of sedation.
- However, other expert consensus proposes that norepinephrine is preferred (10). Until we have evidence declaring a clear winner, all 3 of these are likely reasonable choices.
- Whatever you choose, have it on hand, and consider running it in line prior to the start of the procedure.
For more on the intricacies of managing decompensated aortic stenosis with cardiogenic shock and the approach to PoCUS, stay tuned for part 2.
References
- Otto CM, Nishimura RA, Bonow RO, Carabello BA, Erwin JP, Gentile F, et al. 2020 ACC/AHA Guideline for the Management of Patients With Valvular Heart Disease: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2021 Feb 2;143(5):e72–227.
- Vahanian A, Beyersdorf F, Praz F, Milojevic M, Baldus S, Bauersachs J, et al. 2021 ESC/EACTS Guidelines for the management of valvular heart disease: Developed by the Task Force for the management of valvular heart disease of the European Society of Cardiology (ESC) and the European Association for Cardio-Thoracic Surgery (EACTS). Eur Heart J. 2022 Feb 14;43(7):561–632.
- Stortecky S, Buellesfeld L, Wenaweser P, Heg D, Pilgrim T, Khattab AA, et al. Atrial Fibrillation and Aortic Stenosis. Circ Cardiovasc Interv. 2013 Feb;6(1):77–84.
- Kubala M, de Chillou C, Bohbot Y, Lancellotti P, Enriquez-Sarano M, Tribouilloy C. Arrhythmias in Patients With Valvular Heart Disease: Gaps in Knowledge and the Way Forward. Front Cardiovasc Med [Internet]. 2022 [cited 2023 Nov 10];9. Available from: https://www.frontiersin.org/articles/10.3389/fcvm.2022.792559
- Stiell IG, de Wit K, Scheuermeyer FX, Vadeboncoeur A, Angaran P, Eagles D, et al. 2021 CAEP Acute Atrial Fibrillation/Flutter Best Practices Checklist. Can J Emerg Med. 2021 Sep 1;23(5):604–10.
- Andrade JG, Aguilar M, Atzema C, Bell A, Cairns JA, Cheung CC, et al. The 2020 Canadian Cardiovascular Society/Canadian Heart Rhythm Society Comprehensive Guidelines for the Management of Atrial Fibrillation. Can J Cardiol. 2020 Dec;36(12):1847–948.
- Darby AE, DiMarco JP. Management of Atrial Fibrillation in Patients With Structural Heart Disease. Circulation. 2012 Feb 21;125(7):945–57.
- Pascale King AM. Atrial Fibrillation/Flutter Best Practice Checklist [Internet]. EMOttawa Blog. 2022 [cited 2023 Nov 10]. Available from: https://emottawablog.com/2022/04/atrial-fibrillation-flutter-best-practice-checklist/
- van Diepen S, Katz JN, Albert NM, Henry TD, Jacobs AK, Kapur NK, et al. Contemporary Management of Cardiogenic Shock: A Scientific Statement From the American Heart Association. Circulation. 2017 Oct 17;136(16):e232–68.
- Jentzer JC, Ternus B, Eleid M, Rihal C. Structural Heart Disease Emergencies. J Intensive Care Med. 2021 Sep 1;36(9):975–88.
- Buchwald AB, Meyer T, Scholz K, Unterberg C, Schorn B. Efficacy of balloon valvuloplasty in patients with critical aortic stenosis and cardiogenic shock—the role of shock duration. Clin Cardiol. 2001;24(3):214–8.
- Brunauer A, Koköfer A, Bataar O, Gradwohl-Matis I, Dankl D, Dünser MW. The arterial blood pressure associated with terminal cardiovascular collapse in critically ill patients: a retrospective cohort study. Crit Care. 2014;18(6):719.
- Khot UN, Novaro GM, Popović ZB, Mills RM, Thomas JD, Tuzcu EM, et al. Nitroprusside in Critically Ill Patients with Left Ventricular Dysfunction and Aortic Stenosis. N Engl J Med. 2003 May 1;348(18):1756–63.
- Eleid MF, Nishimura RA, Sorajja P, Borlaug BA. Systemic Hypertension in Low-Gradient Severe Aortic Stenosis With Preserved Ejection Fraction. Circulation. 2013 Sep 17;128(12):1349–53.
- Lloyd JW, Nishimura RA, Borlaug BA, Eleid MF. Hemodynamic Response to Nitroprusside in Patients With Low-Gradient Severe Aortic Stenosis and Preserved Ejection Fraction. J Am Coll Cardiol. 2017 Sep 12;70(11):1339–48.
- Claveau D, Piha-Gossack A, Friedland SN, Afilalo J, Rudski L. Complications Associated With Nitrate Use in Patients Presenting With Acute Pulmonary Edema and Concomitant Moderate or Severe Aortic Stenosis. Ann Emerg Med. 2015 Oct 1;66(4):355-362.e1.
- Alzahrani H, Woo MY, Johnson C, Pageau P, Millington S, Thiruganasambandamoorthy V. Can severe aortic stenosis be identified by emergency physicians when interpreting a simplified two-view echocardiogram obtained by trained echocardiographers? Crit Ultrasound J. 2015 Apr 18;7:5.
- Abe Y, Ito M, Tanaka C, Ito K, Naruko T, Itoh A, et al. A Novel and Simple Method Using Pocket-Sized Echocardiography to Screen for Aortic Stenosis. J Am Soc Echocardiogr. 2013 Jun;26(6):589–96.
- Furukawa A, Abe Y, Ito M, Tanaka C, Ito K, Komatsu R, et al. Prediction of aortic stenosis-related events in patients with systolic ejection murmur using pocket-sized echocardiography. J Cardiol. 2017 Jan;69(1):189–94.
- Furukawa A, Abe Y, Morizane A, Miyaji T, Hosogi S, Ito H. Simple echocardiographic scoring in screening aortic stenosis with focused cardiac ultrasonography in the emergency department. J Cardiol. 2021 Jun;77(6):613–9.





Excellent Review