Posterior circulation ischemia accounts for approximately 20-25% of all ischemic strokes and is a significant cause of patient disability. The diagnosis can be extremely challenging as findings are often not typically focal. Posterior strokes are misdiagnosed more than 3x more often than anterior circulation strokes.1 Similar to anterior circulation strokes, posterior strokes are most commonly from:
- Arterial embolism (most common)
- Atherosclerotic disease
- Arterial dissection
This blog post will review the following topics:
- Review the presentation of posterior circulation strokes
- Posterior Stroke Code: nuances around when to activate the code in the 0-6h and 6-24h time frames
- Workup and management of posterior stroke in the context of acute vestibular syndrome
- Briefly address the workup and management of posterior TIA
Disclaimer #1: Information gathered for this blog includes some expert opinions, local stroke protocols, and published evidence. Recommendations may not be 100% applicable to your center. I recommend you discuss nuances with your local stroke experts to ensure consistent practice among your ED and stroke physicians.
Disclaimer #2: This will not be a comprehensive review of vertigo in the emergency department. For this, I recommend visiting Dr. Peter Johns YouTube channel or the podcasts/show notes for episodes 316 and 358 of EMCrit.
Anatomy
The posterior circulation includes the vertebrobasilar artery system, which supplies the brainstem, cerebellum, occipital lobes, and thalami. The vertebrobasilar artery system includes the two vertebral arteries (which arise from the subclavian vessels) that converge to form the basilar artery. From the vertebral and basilar artery, there are three paired cerebellar arteries (the AICA, PICA and SCA), and then the basilar artery ends in a bifurcation, creating the posterior cerebral arteries (PCAs). Due to the complexity of the affected areas of the brain, the presentation of posterior stroke can mimic the anterior circulation but doesn’t always, and there is significant variability in deficits due to collateral flow.
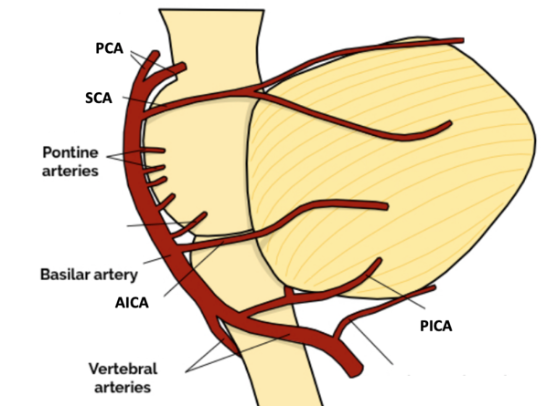
When talking about the classic posterior stroke red flag symptoms, you’ve probably learned about them as either the “Deadly Ds” (or terrible Ds) or in the “Dizziness +1” framework. The Deadly D’s refer to cranial nerve involvement and cerebellar dysfunction, resulting in
- Diplopia
- Dysarthria
- Dysphonia
- Dysphagia
- Dysmetria
- Dysdiadochokinesia
Interestingly, isolated vertigo is an uncommon presentation of posterior circulation stroke. Most cases will have another associated feature. The dizzy +1 framework is helpful as it emphasizes everything you need for a thorough yet targeted history and physical exam to help you identify worrisome findings. Dizziness in addition to any of the following should raise your suspicion:
- Vascular risk factors
- Neck pain
- Occipital headache
- Altered level of consciousness
- Ataxia (gait or limb)
- Nystagmus
- Cranial nerve dysfunction
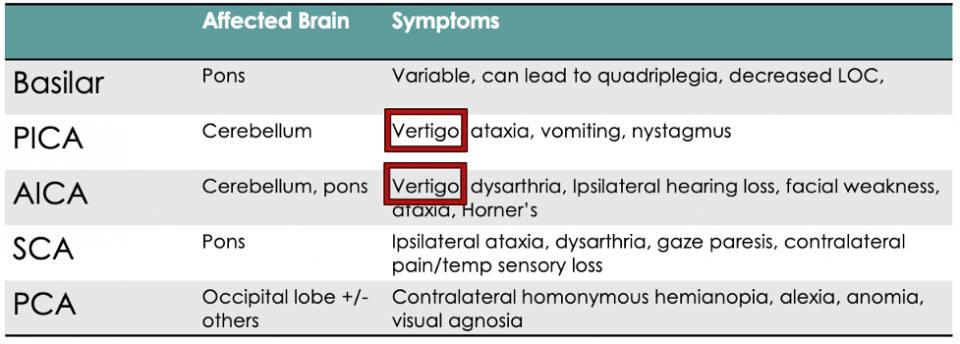
On the other hand, some posterior stroke syndromes present WITHOUT vertigo. The following table shows a simplified version of some posterior stroke syndromes, and you’ll notice that vertigo is on some, but not all, of these lists.2
Part 2: The Posterior Circulation Stroke Code
Be sure to know your local stroke codes and protocols. To briefly review our local practice:
Stroke code activations are split based on time from last seen well. If they were last well within 6h, and the patient has an NIHSS of 4 or more or a disabling deficit, a stroke code is activated.
In the 6-24h time frame, a stroke code is called if the FAST ED score is 4 or more, or if there is severe weakness and aphasia or neglect (positive ACT-FAST screen). These are our triggers to identify a large vessel occlusion which might be amenable to endovascular therapy up to 24 hours following symptom onset.
The goal of the stroke code is to make a quick decision around the administration of high-risk reperfusion therapies, including thrombolysis or EVT. The issue with our current stroke assessment scales is that they are more geared towards anterior circulation deficits. In both anterior and posterior strokes the NIH scale may not reflect the severity of the deficit.
This is why stroke protocols have an emphasis on a disabling deficit. A low NIH with a disabling deficit, although it can occur with anterior or posterior strokes, is more likely to occur in the posterior circulation. For example, a patient with vertigo, ataxia and dysmetria in 2 limbs will score an NIH of 2. However, if the patient can’t walk due to ataxia, this is life-changing. These patients would be considered for thrombolysis and a stroke code should be activated.
From reading and speaking with some of our local stroke neurologists, I recommend stroke code in the context of posterior circulation strokes in the following scenarios:
- Severe ataxia: Inability to ambulate
- Hemianopsia: Hemi-field visual deficit
- Severe dysarthria (debatable, discuss with your local stroke team)
- Lateral Medullary Syndrome
Remember that this is in the context of the patient’s baseline functional status. Mild dysarthria in an elderly multimorbid patient is a different story than a 45-year-old radio host, who is now completely incomprehensible. As a reminder, the above list references some disabling deficits that may be unique to the posterior circulation. Unilateral leg weakness and aphasia or other language deficits are commonly seen in the anterior circulation and do also result in low NIH, but are significantly disabling deficits.
When your initial stroke screen is fairly unremarkable but the patient still has a flavour of possible acute posterior stroke, I encourage you to move through a more comprehensive NIH to look for dysmetria, sensory deficits, and visual field cuts, quickly do a HINTS exam and WALK the patient. If the patient cannot ambulate from ataxia (and they were ambulatory before), or they have a visual field deficit, activate your local stroke team for consideration of advanced therapies.
Isolated Vertigo
Locally, isolated vertigo is often not treated with thrombolysis (and more certainly not with thrombectomy), but may be severe enough to require admission. The natural history of vertigo from posterior stroke is that these patients actually do quite well without treatment. However, it’s usually not our job in the emergency department to decide on advanced therapies, so if the patient has acute vertigo that is clearly central in nature, impairs their ambulation, or has other associated neuro findings on exam, I do recommend activating the stroke team, understanding that they may not receive thrombolysis depending on the neurologist’s assessment of their objective impairment.
Lateral Medullary Syndrome
Lateral medullary syndrome is one of the most missed serious posterior stroke presentations. These patients present with significant vertigo, nausea/vomiting, will score low on an NIH, and further deficits require a thoughtful exam. They will have more subtle neuro findings including crossed-body sensory deficits and Horner’s syndrome.2 The ataxia is often not appreciated initially as they have such significant vomiting that they aren’t getting up and walking on your initial assessment. They will not have motor deficits or facial droop.
Lateral medullary syndrome is extremely important to identify because of significant associated morbidity. Patients have profound dysphagia and frequently suffer from life-threatening aspiration events. Their dysphagia is very slow to improve and often these patients will have PEG tubes for prolonged periods due to the dysphagia. These patients are often treated with thrombolysis as they have poor outcomes. Interestingly, patients with lateral medullary syndrome often have hiccups associated with their nausea and vomiting due to the affected area of the brain. Keep this diagnosis in mind next time your vomiting patient also has hiccups.
Posterior Circulation Stroke Code 0-6 Hours, Take Home Points:
- For all strokes, not just posterior, be sure your assessment is emphasizing disabling deficits.
- When your initial stroke screen is negative, but the initial presenting complaint could still represent stroke (dizziness, resolving dysarthria, vague sensory changes), take a moment to do a slightly more thorough NIH- including dysmetria and visual field deficits.
- Stand the patient up and make sure they can walk. An acutely ataxic patient, visual field deficit or lateral medullary syndrome may be treated with advanced stroke therapies such as thrombolysis.
Posterior Circulation Strokes: 6-24 Hour Time Frame
There have been 4 clinical trials that looked at EVT for basilar artery strokes. BEST3 and Basics4 were the first two, which did not show statistically significant improvement in outcomes compared to medical therapy. These had some methodologic flaws including significant crossover and also only looked at presentations within the first 6-8 hours from symptom onset. BAOCHE5 and ATTENTION6 are the two more recent trials, which did show improved outcomes with EVT compared to medical therapy within 24 and 12h from symptom onset respectively.
While these studies are interesting and represent big advancements in the world of EVT, we aren’t going to go through them in significant detail today because they don’t actually change our practice in the emergency department.
These studies all focused on basilar artery strokes, which is a big artery resulting in large territories of ischemic brain. These strokes will be captured in our current large vessel occlusion screen. These studies required moderate to high NIHSS to even be included. Some of the other syndromes we have talked about in this article, like isolated dizziness/ataxia and lateral medullary syndrome are usually vertebral artery occlusions, so they are not currently EVT targets. Basilar artery strokes with low NIH scores are generally not treated with EVT as there is a significant risk of further ischemic or hemorrhagic injury. Thrombolysis actually appears to be safer in the posterior circulation than the anterior, with hemorrhage rates around 4%. EVT has poor outcomes in around 10% of cases where the posterior circulation is involved.
So for the 6-24h time frame, stick to our current LVO screening with the FAST-ED score of 4 or higher.7 This screening tool will still capture posterior large vessel occlusions that are potentially amenable to EVT.
Part 3: Acute Vestibular Syndrome
In the emergency department, we spend a lot of time trying to figure out which dizzy patients are having a possible posterior circulation stroke. The updated GRACE 3 guidelines were published in March of 2023, and they divide vertigo into three main categories:
- Acute vestibular syndrome: the acute onset of continuous, persistent dizziness or vertigo lasting 24 hours or longer. The main items on this differential are vestibular neuritis and stroke.
- Spontaneous episodic vestibular syndrome: the differential is broad and includes vestibular migraine, posterior TIA, and underlying medical etiologies, among others.
- Triggered (positional) episodic vestibular syndrome (most commonly BPPV)
Pertinent Updates from the GRACE-3 Guidelines
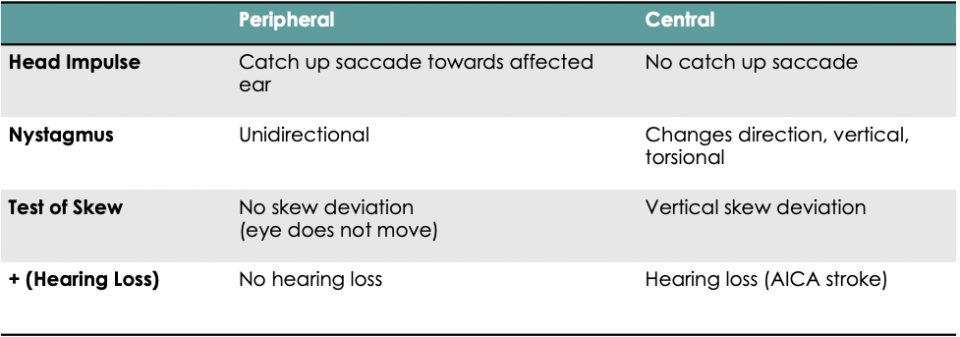
Recommendation 2: In adult ED patients with AVS with nystagmus, we recommend routine use of the three-component head impulse, nystagmus, test of SKEW (HINTS) examination for clinicians trained in its use* to distinguish between central (stroke) and peripheral (inner ear, usually vestibular neuritis) diagnoses.
Recommendation 3: In adult ED patients with AVS with nystagmus, we suggest assessing hearing at the bedside by finger rub to identify new unilateral hearing loss as an additional criterion to aid in the identification of stroke, even if the three-component HINTS examination results suggest a peripheral vestibular diagnosis.
Many emergency physicians have already incorporated the HINTS+ exam into their practice, however over recent years there’s been quite a lot of debate over the emergency department use and reliability of the HINTS+ exam. There’s also no actual definition on what it means “to be HINTS+ trained,” as recommendation #2 references. Aside from the proof I see in our local ED, there is actually evidence that emergency physicians can use this test appropriately and effectively. As a reminder, the HINTS+ exam is used only in cases of acute vestibular syndrome with associated nystagmus on exam.
A study by Gerlier et al. found the HINTS exam to be 96.7% sensitive and 67% specific when used in the emergency setting.9 It was a prospective study where emergency physicians underwent 4 hours of lectures and 2 hours of workshops to learn three different tests used in differentiating various causes of vertigo, including the HINTS exam. They then prospectively enrolled 300 patients with vertigo, and the primary outcome was the diagnostic accuracy of the HINTS exam. The reference test was diffusion weighed MRI done at least 48 hours after onset of symptoms.
I spoke with one of our local experts, Dr. Peter Johns, to obtain some advice on how to become “HINTS competent” He recommends the following:
- Be able to outline the HINTS exam components and what each of the findings means. As in, which findings indicate a peripheral cause, and which ones indicate a central cause.
- Have someone who is comfortable with the exam watch you do it a few times.
- Have your interpretation of the findings confirmed by a physician comfortable with the test. I suggest filming the patient’s eyes during your exam (with the patient’s consent) so that you can go back and watch it again in case you aren’t sure what you saw, and it allows you to show another physician and confirm how you are interpreting the test.
I also highly recommend Dr. John’s Youtube videos as a place to start if you are uncomfortable with the exam.
Recommendation 7: in adult ED patients with acute vestibular syndrome and central or equivocal HINTS results, we recommend the use of stroke protocol MRI (with DWI and MRA) to further help distinguish between central (stroke) and peripheral diagnoses.
This is one of the recommendations that is not necessarily applicable to the Canadian healthcare system. There are places in Canada where it’s becoming standard of care to have this MRI for this scenario done within 1 week of presentation, but many centers do not have this type of access. Timing of MRI will require coordination with your neurology colleagues and outpatient stroke follow-up clinics, and may not happen until weeks later in cases of mild symptoms not requiring hospital admission.
Of note, the GRACE 3 guidelines specifically recommend against the use of CT/CTA to distinguish between central and peripheral causes. Given we cannot get same-day MRI for these cases, it is still likely that we will be obtaining CT/CTA to assess for any obvious stroke or bleed, understanding that this test is not sufficient to completely visualize the posterior system, and choosing to treat with dual antiplatelet and arrange appropriate neurology follow up (inpatient vs outpatient depending on safety for discharge) will be based on our clinical assessment (history and physical exam).
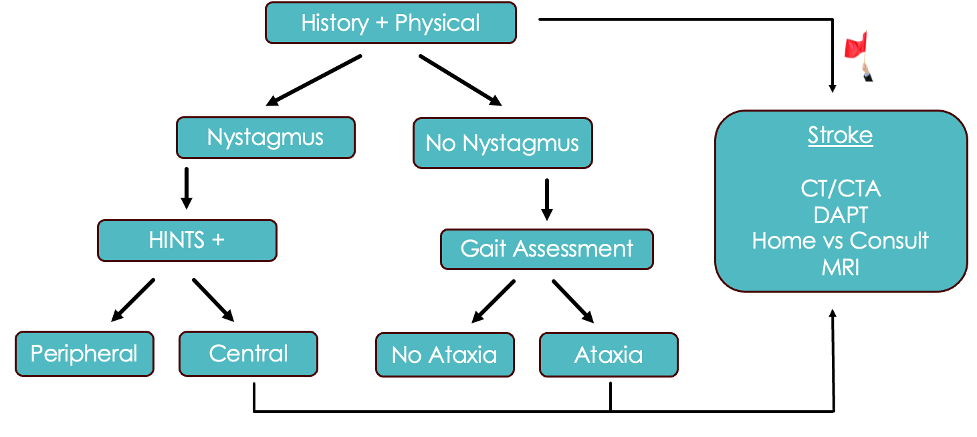
Below is an algorithm that takes into account everything we have just discussed for Acute Vestibular Syndrome:
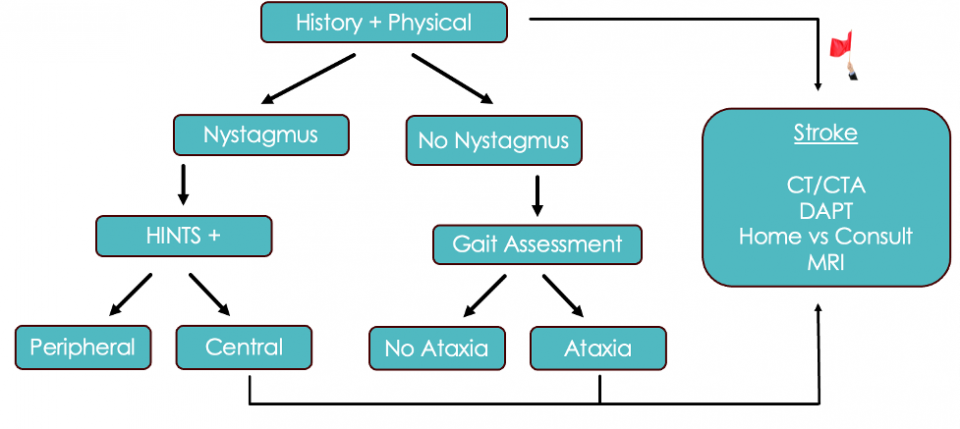
- First, we are going to do a thorough history and physical. If they have any of our red flag symptoms or signs on exam, such as dysmetria, diplopia, visual field deficits, dysphagia, or dysarthria, we are going to pursue a presumptive stroke diagnosis.
- If not, identify whether the patient has nystagmus
- If there is nystagmus, we are going to do the HINTS + exam and then pursue our stroke pathway if the HINTS exam suggests a central cause
- Now, if there’s no nystagmus, these cases get more challenging. GRACE 3 recommends that the next steps be based on a gait assessment. If the patient is ataxic, we are going to pursue the stroke pathway.
- If they have no nystagmus and no ataxia, we are really entering an evidence-free zone here. In speaking with experts, this appears to be a very low-risk category of patients, but we don’t really have many studies to back that up.
- Patients falling into the suspected stroke category should undergo CT/CTA in the department since we don’t have immediate access to the definitive test of MRI in this moment.
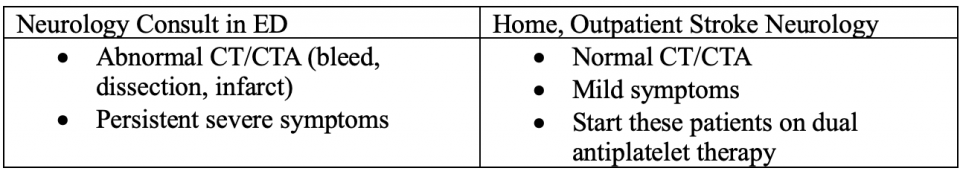
I recommend we talk to neuro in the department for anyone with an abnormal scan (so obviously if they have a bleed or dissection) but also for cases with an established infarct. Neurology will at minimum want to look at the scan and based on the size of the infarct, decide if it’s something that should be monitored in the hospital or not. However, we know that many of these patients will have a normal CT/CTA given sensitivity of CT/CTA for posterior infarct is only 10-41%. What do we do with these ones? If they have severe persistent symptoms and can’t go home, then neurology should be consulted for admission.
If their symptoms are extremely mild, (they are ambulating well and able to swallow) they can be started on DAPT and sent to outpatient rapid stroke follow-up. They will get an MRI as an outpatient via the stroke pathway. Some of you may feel most comfortable calling neurology even if you think they are dischargeable, which is completely reasonable. I recommend discussing some of these scenarios with your local stroke physicians to get a sense of your local stroke expert practices.
Take-home point: Make your clinical impression before you obtain your CT/CTA. Your decision to send someone down the posterior stroke pathway with dual antiplatelet should be guided by your history and physical exam, not your CT/CTA results! These patients will need confirmatory MRI testing when possible based on your local resources.
References
- Costa A, Miranda O, Cerqueira A, et al. (January 21, 2022) A Patient With (Initially) Non-Persistent Vertigo – A Posterior Circulation Stroke Case. Cureus 14(1): e21468. DOI 10.7759/cureus.21468
- Amin, H. & Schindler, J. Vascular neurology board review- An essential study guide. 2nd Springer Cham. 2022.
- Liu X, Dai Q, Ye R, Zi W, Liu Y, Wang H, et al. Endovascular treatment versus standard medical treatment for vertebrobasilar artery occlusion (BEST): an open-label, randomised controlled trial. Lancet Neurol. 2020;19:115–122.
- Langezaal L, van der Hoeven E, Mont’Alverne F, de Carvalho J, Lima FO, Dippel D, et al. Endovascular therapy for stroke due to basilar-artery occlusion. N Engl J Med. 2021;384:1910–1920.
- Jovin TG, Li C, Wu L, Wu C, Chen J, Jiang C, Shi Z, Gao Z, Song C, Chen W, et al. Trial of thrombectomy 6 to 24 Hours after stroke due to basilar-artery occlusion.N Engl J Med. 2022; 387:1373–1384. doi: 10.1056/nejmoa2207576
- Tao C, Li R, Zhu Y, Qun S, Xu P, Wang L, et al. Endovascular treatment for acute basilar artery occlusion: a multicenter randomized controlled trial (ATTENTION) Int J Stroke. 2022 Feb 22.
- Lima F O, Silva G S, Furie K L. Field Assessment Stroke Triage for Emergency Destination: A Simple and Accurate Prehospital Scale to Detect Large Vessel Occlusion Strokes. 2016;47(08):1997–2002.
- Edlow et al. Guidelines for reasonable and appropriate care in the emergency department 3 (GRACE-3): Acute dizziness and vertigo in the emergency department. Acad Emerg Med. 2023;30:442-486.
- Gerlier et al. Differentiating central from peripheral causes of acute vertigo in an emergency setting with the HINTS, STANDING, and ABCD2 tests: A diagnostic cohort study. Acad Emerg Med. 2021;28(12):1368-1378.

Excellent review. Thank you.
I just think that this subject has been made more difficult by not defining terms very clearly, particularly for branch points.
Can I just clarify to see if you agree?
– Nystagmus = Spontaneous nystagmus = Nystagmus in neutral gaze (ie not gaze evoked or provoked). Can use Frenzel or white paper, or can just look closely without fixing gaze.
– Ataxia = unable to walk independently.
Thanks
Scott