In this post, we will explore the current literature regarding common EM controversies, covering the nature and prevalence of penicillin allergies, the management of skin and soft tissue infections, and the role of NSAIDs for acute pain.
Penicillin allergies in the ED
- Penicillin (PCN) is a Beta-lactam antibiotic discovered in 1928. As of 2010, Beta-lactams (penicillins and cephalosporins) were the top 2 classes of antibacterial drugs sold in the US and 55% of global antibiotic drugs consumed [1].
- With time, patients became allergic to PCN and this relationship was studied by multiple researchers yielding variable results.
- What is the true rate of a PCN allergy?
- 1-4% of patients taking PCN develop a maculopapular rash, with up to 10% of patients reporting a non-life-threatening reaction (non-Type 1) [2]
- The accuracy of a reported clinical history of PCN allergy in predicting a positive skin test to PCN is low. Patients with remote or uncertain history of PCN allergy are equally likely to have a positive PCN skin test compared with patients without a history of PCN allergy (approximately 1.7%) [3].
- A positive skin test is associated with higher incidence of Type 1 allergic reactions (anaphylaxis – more clinically relevant). A negative skin test almost rules out a type 1 reaction (95-98% sensitive) [4]
- The true rate of PCN allergy was thought to be over-estimated due to mislabeling factors [2]:
- Prescribing PCNs for viral infections → viral exanthem → rash attributed to PCN rather than viral rash
- EBV maculopapular rash following amoxicillin prescription
- Other viral rashes (Cocksackie, Echovirus, HIV)
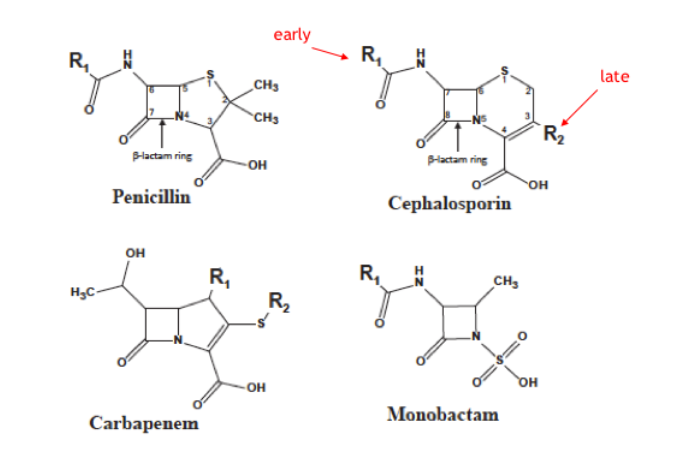
- The PCN molecule shares a Beta-lactam ring with other Beta-lactam antibiotics (cephalosporins, carbapenem, monobactam). What about cross-reactivity to other Beta-lactams?
- Cephalosporins
- Early studies (1960s) over-reported cross-reactivities due to contaminated models of cephalosporins (made from compounds contaminated with Benzylpenicillin) [6-9]
- Cross-reactivities ranged from 10-50% in some studies. [6-9]
- Cross-reactivity was thought to be due to the shared Beta-lactam ring. [6-9]
- Recent studies using synthetic “clean” cephalosporins show that cross-reactivity is much lower (<10%) and thought to be secondary to side-chain similarities (R1 and R2) [8,9]
- 1st Gen Cephalosporins have highest risk (0.5-5%) with minimal risk in 2nd/3rd Gen (0.1-2%) [8,9]
- Carbapenems
- Cross-reactivity is very low, some studies quoting it at 0.9% and overall <5.2% [6-9]
- Lowest risk of B-lactams due to difference in side-chain
- Cephalosporins
- Multiple implications from mislabeling PCN allergies and avoiding B-lactams in PCN allergic patients
- Increased failure rate and inadequate coverage
- Some studies show higher treatment failure rates in patients reporting PCN allergies who did not receive B-lactam antibiotics for their infections [10,11]
- Bacterial resistance
- PCN allergic patients associated with higher rates of C.diff (23%), VRE (30%) and MRSA (14%) thought to be related to antibiotic substitutions [11]
- Costs
- Canadian ICU study demonstrated that the use of alternative antibiotics in place of standard beta-lactams incurred an additional cost of $15,672 CAN [11]
- Study showing increased cost per prescription when substituting another antibiotic secondary to a reported PCN allergy (average 77$ difference) [12]
- Side effects
- Higher side effects with Beta-lactam alternatives (Vancomycin and acute kidney injuries, aminoglycosides, quinolones and Cdiff) [13]
- Increased failure rate and inadequate coverage
- How to approach a PCN allergy in the ED?
- Taking a detailed history is key to de-labelling PCN allergic patients whenever possible
- Elements of a good allergy history [14]
- What exactly were the symptoms?
- Raised, red, itchy lesions < 24 hrs (hives/urticarial)
- Swelling of the mouth, eyes, lips or tongue (angioedema)
- Blisters, ulcers in mucosal areas, skin peeling (SJS, TEN)
- Respiratory or hemodynamic compromise (anaphylaxis)
- Joint pains (serum sickness)
- Other organs like kidneys, lungs, liver (DRESS, Type 4 severe reactions)
- Timing of the reaction after PCN (minutes, hours, days), was it after the first or multiple doses?
- How long ago did the reaction happen? (after 10 years of avoidance, only 20% of patients with IgE-mediated PCN allergy will still be allergic)
- How was the reaction treated? Any epinephrine required?
- Has patient tolerated similar medications (ampicillin, amoxicillin, cephalexin) since the PCN reaction?
- What exactly were the symptoms?
Algorithm on approaching penicillin allergies [14]:

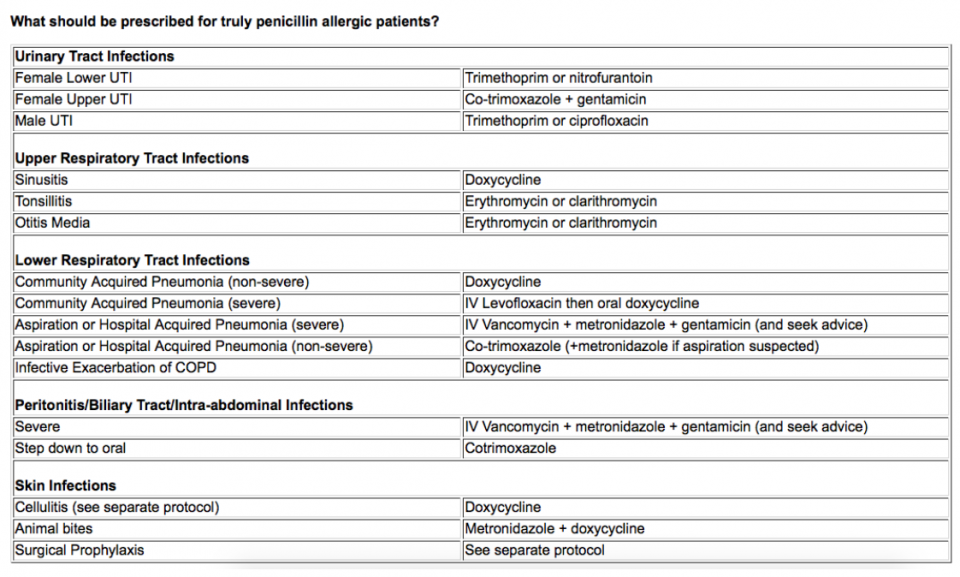
Alternatives to B-lactams in well-defined PCN allergic patients [15]
Take Home Points
- Cross reactivity from PCN to Cephalosporins is lower than previously published (1st > 2nd > 3rd GEN Cephalosporins)
- If no features of Type 1 or severe Type 4 reactions (anaphylaxis, angioedema, DRESS, SJS/TEN), can administer Cephalosporins with no expected significant reactions
- May consider doing a test dose with 10% of dose and close monitoring
- Adequate documentation and de-labelling false-allergies reduces costs for the system.
- Use clinical judgment/tools available (algorithms, pharmacists, allergists) to assess risk in patients reporting severe reactions to PCN.
Skin and Soft Tissue Infections (SSTIs)
- SSTIs and abscesses are among the top presentations to the ED with over 3 million visits annually in the US [16]
- Incision and drainage (I+D) is mainstay of treatment for abscesses as per the 2014 IDSA guidelines below [17]
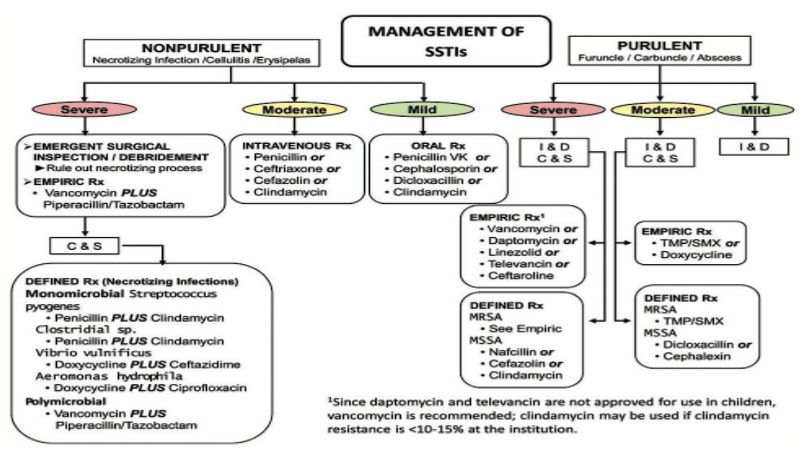
- Review of multiple ED resources and IDSA guidelines show differing approaches with respect to abscess management:
- Antibiotics
- Most resources do not recommend routine antibiotic prescription for mild abscesses [17, 18]
- Antibiotics are recommended under certain circumstances [17]:
- Severe or extensive disease (using clinical judgment)
- Rapid progression in presence of associated signs/symptoms of systemic illness (SIRS)
- Associated comorbidities or immunosuppression
- Extremes of age
- Abscess in area difficult to drain (face, hand, genitalia)
- Associated septic phlebitis
- Lack of response to I+D alone
- If treating with Abx, course of 5-10 days with an agent that has MRSA coverage (doxycycline, clindamycin, Septra); if high risk of MRSA [17]
- Incision + Drainage
- Conventional straight line technique vs. Loop (novel technique using two small incisions at edge of abscess and a loop to maintain drainage, repositioned BID and self-removed by patient within 10-14 days)
- Conventional I+D is quick, easy, and traditionally taught in medical training (including straight line, ellipse, etc).
- Some studies support the LOOP technique as having improved cosmesis, decreased need for wound care/follow-ups, decreased costs to system, less painful, and more tolerated by pediatric population [19-23]
- Link to LOOP technique video: https://www.youtube.com/watch?v=gw7tA1B9Aos)
- Few RCTs comparing conventional vs. Loop I+D techniques showed non-inferiority in adults and pediatric populations. However, data is limited to single-centre/small sample size. Loop technique is promising but stronger data is required before making a recommendation for it [19-23]
- Conventional straight line technique vs. Loop (novel technique using two small incisions at edge of abscess and a loop to maintain drainage, repositioned BID and self-removed by patient within 10-14 days)
- Irrigation
- Most emergency physicians irrigate abscesses although not much data is available to guide this practice [24]
- One study showed similar failure rates between irrigation (up to 100cc of NS) vs. non-irrigation group of adults with similar-size abscesses [24]
- Overall, consensus is to irrigate, but to what extent is unclear
- Packing
- Traditionally done and thought to prevent premature closure of the wound and abscess recurrence
- New studies show that packing may be painful and unnecessary [25-27]
- Pediatric ED population RCT comparing packed vs. non-packed showed similar failure rates [25]
- Adult ED population RCT comparing packed vs. non-packed showed similar failure rates but higher pain and higher narcotic use in the packed group [26]
- Cochrane review (2 RCTs) comparing packing vs. non-packing in perianal abscesses showed no difference in wound healing time or pain as well as no influence in secondary outcomes (recurrence, incontinence, return to work, resource use) [27]
- Antibiotics
Take Home Points
- Mainstay of abscess management remains an I+D (use technique that you prefer, make a large incision – i.e: “don’t skimp on it”)
- Irrigation may or may not make a difference
- Packing is not always necessary, especially for smaller abscesses
- Prescribe antibiotics only in high risk cases (5-7 days is usually sufficient)
NSAIDs for acute pain
- NSAIDs are among the top analgesic choice for treating moderate-severe acute pain in the ED [28-29]
- Oral (Ibuprofen, Naproxen) vs. parenteral (IV/IM Toradol) NSAIDs debate ongoing for decades [28]
- Evidence overwhelmingly shows no difference in analgesic effect between the two routes. However, many emergency physicians continue to use parenteral NSAIDs with the misconception that it is more “powerful and effective” than their oral counterparts [29,30]
- The idea that administering a drug parenterally infers an additional placebo “needle effect” is a myth that has been disproven by multiple RCTs [30-32]
- Providing parenteral (IM/IV) NSAIDs when not indicated is potentially harmful (more painful IM route) with no added benefits to oral NSAIDs [32]
- Reserve parenteral NSAIDs to cases where oral administration is contraindicated (i.e: NPO, decreased LOC, nausea/vomiting, ileus)
- Why care about parenteral vs. oral in the ED?
- Side effects
- Parenteral Toradol has higher COX-1 selectivity (coefficient of 2.5 vs. 0.5 in Naproxen/Advil) → more GI side effects [33]
- Risk of GI side effects (including hemorrhage) proportional to dose given [29-33]
- Single dose of parenteral Toradol interferes with platelet function (worsens bleeding in post-op patients) [29-33]
- Costs
- As an example, at The Ottawa Hospital, a parenteral dose of Toradol vs. oral equivalent (Advil/Naproxen) is 18-28 x more expensive [34]
- Side effects
- Ketorolac and ceiling effect?
- IV: Post-op studies showing IV Ketorolac at low doses (7.5 mg – 10 mg) was as effective as larger doses (15-30 mg) at providing analgesia [35, 36]
- IM: Cancer-pain studies showing 10 mg similar to 30 mg IM in relief of pain [37,38]
- Emergency Department study in 2017 [39]
- IV Ketorolac 10 mg vs. 15 mg vs. 30 mg were all equally effective in acute pain reduction at 30 mins
Take Home points
- Unless contraindicated, using oral NSAIDs is recommended over parenteral NSAIDs. Advil or Naproxen are adequate starting options.
- If prescribing NSAIDs, ensure no contraindications and do not prescribe for a long duration. Consider prophylactic PPIs for patients with high risk or history of peptic ulcer disease / GI side effects. Avoid NSAIDs in the following population:
- Over age 65
- History of peptic ulcer disease
- Taking corticosteroids, anticoagulants, or ASA
- If using parenteral (IV/IM) NSAIDs, consider starting with lower doses (i.e: Ketorolac 10-15 mg/dose) especially in the elderly (age >65)
References
- Pichicero, M and Zagursky R (2014). Penicillin and Cephalosporin allergy. Ann Allergy Asthma Immunol.112. 404e412
- Salkind, A. R., Cuddy, P. G., & Foxworth, J. W. (2001). Is this patient allergic to penicillin?: an evidence-based analysis of the likelihood of penicillin allergy. Jama, 285(19), 2498-2505.
- Gadde J et al (1993). Clinical experience with pencillin skin testing in a large inner-city STD clinic. JAMA. 270:2456-2463.
- Trubiano, J. A., Adkinson, N. F., & Phillips, E. J. (2017). Penicillin allergy is not necessarily forever. Jama, 318(1), 82-83.
- Pichichero, M. E., & Casey, J. R. (2007). Safe use of selected cephalosporins in penicillin-allergic patients: a meta-analysis. Otolaryngology-Head and Neck Surgery, 136(3), 340-347.
- Pichichero, M. E. (2007). Use of selected cephalosporins in penicillin-allergic patients: a paradigm shift. Diagnostic microbiology and infectious disease, 57(3), S13-S18.
- Pichichero, M. E. (2005). A review of evidence supporting the American Academy of Pediatrics recommendation for prescribing cephalosporin antibiotics for penicillin-allergic patients. Pediatrics, 115(4), 1048-1057.
- Herbert, M. E., Brewster, G. S., & Lanctot-Herbert, M. (2000). Ten percent of patients who are allergic to penicillin will have serious reactions if exposed to cephalosporins. The Western journal of medicine, 172(5), 341.
- Campagna, J. D., Bond, M. C., Schabelman, E., & Hayes, B. D. (2012). The use of cephalosporins in penicillin-allergic patients: a literature review. The Journal of emergency medicine, 42(5), 612-620.
- Meghan, N. J et al. (2016). Consequences of avoiding B-lactams in patients with B-lactam allergies. Journal of Allergy Clinical Immunology. 137:1148-53.
- Picard, M., Bégin, P., Bouchard, H., Cloutier, J., Lacombe-Barrios, J., Paradis, J., … & Paradis, L. (2013). Treatment of patients with a history of penicillin allergy in a large tertiary-care academic hospital. The Journal of Allergy and Clinical Immunology: In Practice, 1(3), 252-257.
- Raja, A. S., Lindsell, C. J., Bernstein, J. A., Codispoti, C. D., & Moellman, J. J. (2009). The use of penicillin skin testing to assess the prevalence of penicillin allergy in an emergency department setting. Annals of emergency medicine, 54(1), 72-77
- Macy, E., & Contreras, R. (2014). Health care use and serious infection prevalence associated with penicillin “allergy” in hospitalized patients: a cohort study. Journal of Allergy and Clinical Immunology, 133(3), 790-796.
- Blumenthal, K. and Solensky, R. (2018). Allergy evaluation for immediate penicillin allergy: Skin test-based diagnostic strategies and cross-reactivity with other beta-lactam antibiotics: ALGORITHM – Approach to the patient with a past penicillin reaction who requires antibiotics. UpToDate. Accessed December 14, 2017.
- Pegler, S., & Healy, B. (2007). In patients allergic to penicillin, consider second and third generation cephalosporins for life threatening infections. BMJ : British Medical Journal, 335(7627), 991. http://doi.org/10.1136/bmj.39372.829676.47
- May, L., Harter, K., Yadav, K., Strauss, R., Abualenain, J., Keim, A., & Schmitz, G. (2012). Practice patterns and management strategies for purulent skin and soft-tissue infections in an urban academic ED. The American journal of emergency medicine, 30(2), 302-310.
- Dennis L. Stevens, Alan L. Bisno, Henry F. Chambers, E. Patchen Dellinger, Ellie J. C. Goldstein, Sherwood L. Gorbach, Jan V. Hirschmann, Sheldon L. Kaplan, Jose G. Montoya, James C. Wade; Practice Guidelines for the Diagnosis and Management of Skin and Soft Tissue Infections: 2014 Update by the Infectious Diseases Society of America, Clinical Infectious Diseases, Volume 59, Issue 2, 15 July 2014, Pages e10–e52, https://doi.org/10.1093/cid/ciu296. Accessed November 30th, 2017.
- Prusakowski, M. K., & Kuehl, D. R. (2015). Trends in emergency department management of skin abscesses. American journal of infection control, 43(4), 336-340.
- Özturan, İ. U., Doğan, N. Ö., Karakayalı, O., Özbek, A. E., Yılmaz, S., Pekdemir, M., & Suner, S. (2017). Comparison of loop and primary incision & drainage techniques in adult patients with cutaneous abscess: A preliminary, randomized clinical trial. The American Journal of Emergency Medicine.
- Gaszynski, R., Punch, G., & Verschuer, K. (2016). Loop and drain technique for subcutaneous abscess: a safe minimally invasive procedure in an adult population. ANZ journal of surgery.
- Ladde, J. G., Baker, S., Rodgers, C. N., & Papa, L. (2015). The LOOP technique: a novel incision and drainage technique in the treatment of skin abscesses in a pediatric ED. The American journal of emergency medicine, 33(2), 271-276.
- Gottlieb, M., & Peksa, G. D. (2017). Comparison of the loop technique with incision and drainage for soft tissue abscesses: A systematic review and meta-analysis. The American journal of emergency medicine.
- Thompson, D. O. (2014). Loop drainage of cutaneous abscesses using a modified sterile glove: a promising technique. The Journal of emergency medicine, 47(2), 188-191.
- Chinnock, B., & Hendey, G. W. (2016). Irrigation of cutaneous abscesses does not improve treatment success. Annals of emergency medicine, 67(3), 379-383.
- Kessler, D. O., Krantz, A., & Mojica, M. (2012). Randomized trial comparing wound packing to no wound packing following incision and drainage of superficial skin abscesses in the pediatric emergency department. Pediatric emergency care, 28(6), 514-517.
- O’Malley, G. F., Dominici, P., Giraldo, P., Aguilera, E., Verma, M., Lares, C., … & Williams, E. (2009). Routine packing of simple cutaneous abscesses is painful and probably unnecessary. Academic Emergency Medicine, 16(5), 470-473.
- Smith, S. R., Pearce, L. E., Newton, K., Dumville, J. C., Smith, J. A., Barrow, P. J., … & Hill, J. (2014). Internal dressings for healing perianal abscess cavities. Cochrane Database of Systematic Reviews, (7).
- Arora, S., Wagner, J. G., & Herbert, M. (2007). Myth: parenteral ketorolac provides more effective analgesia than oral ibuprofen. Canadian Journal of Emergency Medicine, 9(1), 30-32.
- Wright, J. M., Price, S. D., & Watson, W. A. (1994). NSAID use and efficacy in the emergency department: single doses of oral ibuprofen versus intramuscular ketorolac.
- Neighbor, M. L., & Puntillo, K. A. (1998). Intramuscular ketorolac vs oral ibuprofen in emergency department patients with acute pain. Academic emergency medicine, 5(2), 118-122.
- Schwartz, N. A., Turturro, M. A., Istvan, D. J., & Larkin, G. L. (2000). Patients’ Perceptions of Route of Nonsteroidal Anti‐inflammatory Drug Administration and Its Effect on Analgesia. Academic Emergency Medicine, 7(8), 857-861.
- Braaten, K. P., Hurwitz, S., Fortin, J., & Goldberg, A. B. (2014). Intramuscular ketorolac versus oral ibuprofen for pain relief in first-trimester surgical abortion: a randomized clinical trial. Contraception, 89(2), 116-121.
- Danelich, I. M., Wright, S. S., Lose, J. M., Tefft, B. J., Cicci, J. D., & Reed, B. N. (2015). Safety of nonsteroidal antiinflammatory drugs in patients with cardiovascular disease. Pharmacotherapy: The Journal of Human Pharmacology and Drug Therapy, 35(5), 520-535.
- Calculations as per TOH pharmacist scenario.
- Reuben, S. S., Connelly, N. R., Lurie, S., Klatt, M., & Gibson, C. S. (1998). Dose-response of ketorolac as an adjunct to patient-controlled analgesia morphine in patients after spinal fusion surgery. Anesthesia & Analgesia, 87(1), 98-102.
- Brown, C. R., Moodie, J. E., Wild, V. M., & Bynum, L. J. (1990). Comparison of intravenous ketorolac tromethamine and morphine sulfate in the treatment of postoperative pain. Pharmacotherapy: The Journal of Human Pharmacology and Drug Therapy, 10(6P2).
- Staquet, M. J. (1989). A Double‐Blind Study with Placebo Control of Intramuscular Ketorolac Tromethamine in the Treatment of Cancer Pain. The Journal of Clinical Pharmacology, 29(11), 1031-1036.
- Minotti, V., Betti, M., Ciccarese, G., Fumi, G., Tonato, M., & Favero, A. (1998). A Double‐Blind Study Comparing Two Single‐Dose Regimens of Ketorolac with Diclofenac in Pain Due to Cancer. Pharmacotherapy: The Journal of Human Pharmacology and Drug Therapy, 18(3), 504-508.
- Motov, S., Yasavolian, M., Likourezos, A., Pushkar, I., Hossain, R., Drapkin, J., … & Rockoff, B. (2017). Comparison of intravenous ketorolac at three single-dose regimens for treating acute pain in the emergency department: A randomized controlled trial. Annals of emergency medicine, 70(2), 177-184.


