In this post, we cover the often seen, but poorly understood entity of Interstitial Lung Disease (ILD). We will focus our discussion on the key features and approaches that the front-line provider needs to know to feel confident managing the newly diagnosed ILD patient, the ILD flare, and the crashing ILD patient.
Objectives:
- Understand broad strokes of what ILD is, how to classify it, and how to discuss it
- Approach to the known ILD patient presenting with SOB/CP
- Provide information about new ILD medications and their subsequent complications
- Approach to the newly diagnosed ILD patient and special cases in which you should consider ILD
- Approach to the crashing ILD patient (Intubation, Goals of care, Transplant considerations)
ILD classification
- Interstitial Lung disease is defined as a “Heterogenous group of lung disorders that all share common symptomatic, radiographic, or histopathologic traits”
- This includes “a constellation of findings including dyspnea and diffuse bilateral infiltrates on imaging with a tissue diagnosis (when obtained)” (Antin-Ozerkis 2015; Herzog, Rubinowitz, and Gulati 2015)
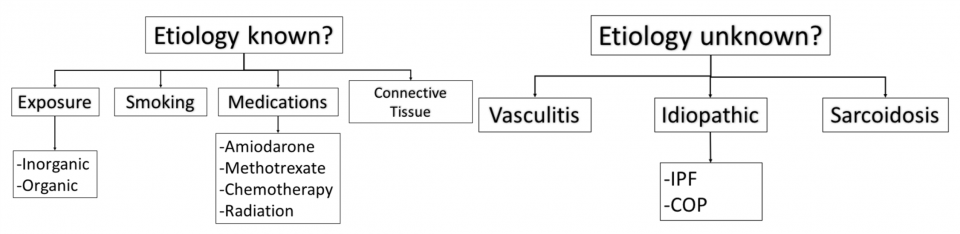
- ILD can be classified as either known or unknown etiology (Travis et al. 2013)
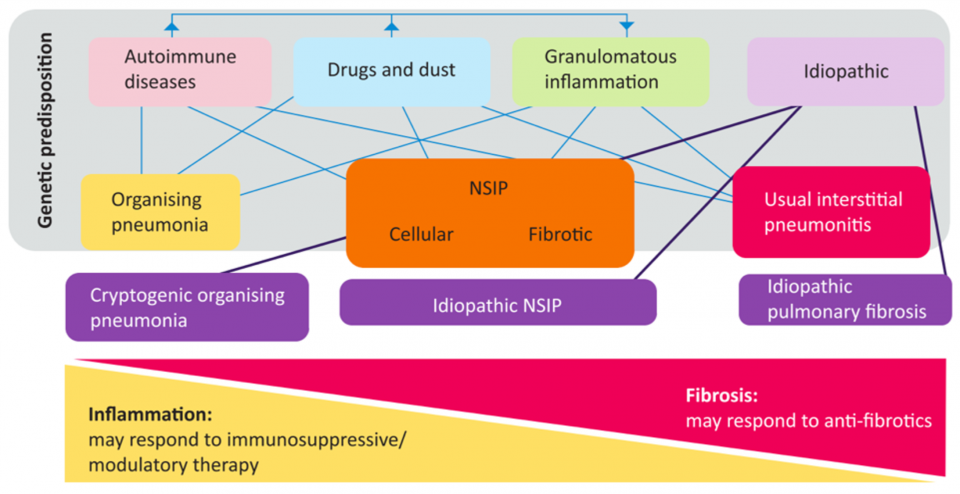
- A more helpful way to classify etiologies is on a spectrum of inflammation (i.e. Cryptogenic Organizing pneumonia) to fibrosis (i.e. Idiopathic pulmonary fibrosis)
(Mikolasch, Garthwaite, and Porter 2016)
Approach to the Known ILD patient with Shortness of Breath or Chest Pain
- “Shortness of Breath is NOT always an ILD Flare”
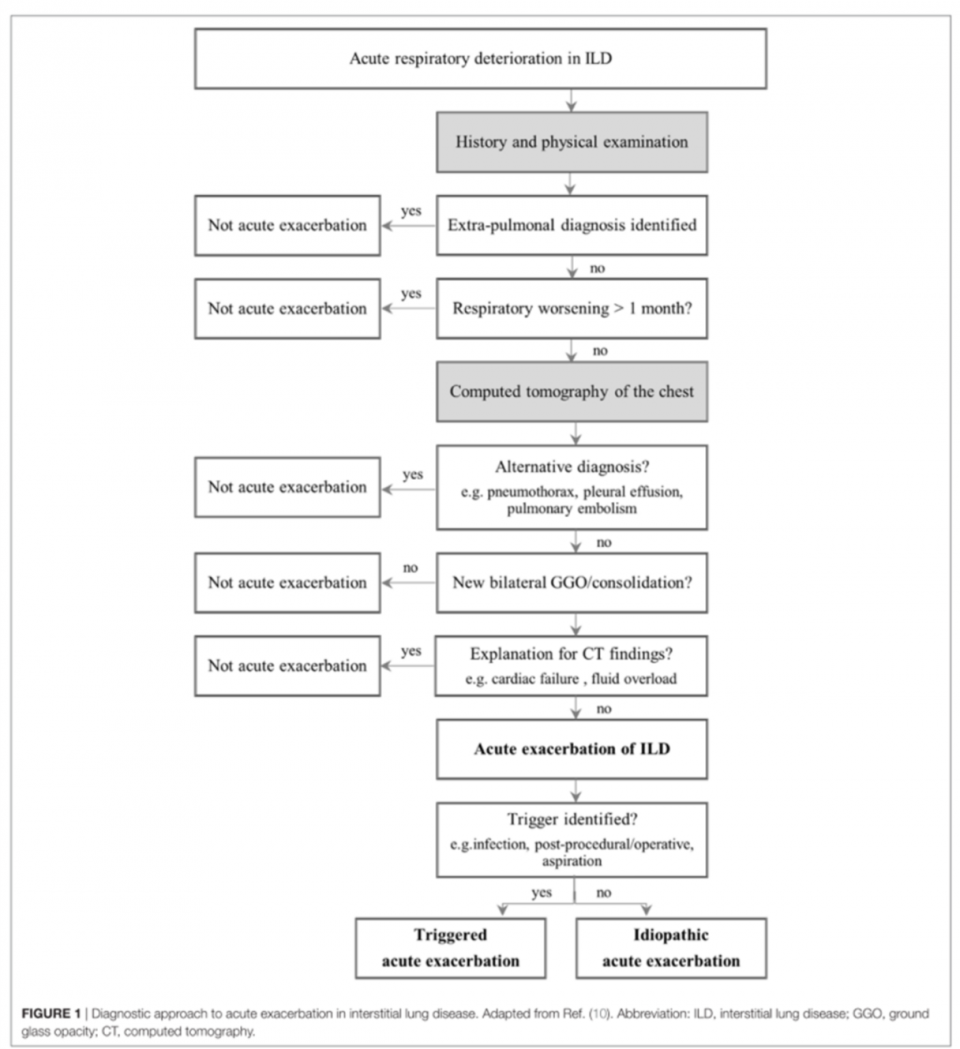
- An ILD flare is an “acute, clinically significant, respiratory deterioration of unidentifiable cause within the past month, and new bilateral opacities on CT” (Collard et al. 2016)
- It has many similarities to acute lung injury precipitants
- ILD flares have HIGH mortality associated with them (Leuschner and Behr 2017)
- One-month mortality: 33%
- Three-month mortality: 60%
- The approach to the ILD patient must include considerations of the following alternate pathologies:
- Pulmonary Embolism
- Pulmonary HTN
- CHF
- Infection
- Coronary artery disease
- Lung Cancer
- Diffuse Alveolar Hemorrhage
Pulmonary Embolism
- ILD patients are at higher risk for DVT and PE than Cancer patients (OR 1.54) (Sprunger et al. 2012)
Pulmonary HTN (Caminati, Cassandro, and Harari 2013)
- Group 3 cause of pulmonary hypertension
- Prevalence of pHTN amongst ILD patients is between 14% and 31%
- Systemic sclerosis is thought to be the cause of 30% of all pulmonary hypertension cases
- Causes pHTN “out of proportion” to what is expected in ILD alone
- 5x mortality risk compared to systemic sclerosis patients without lung involvement
Congestive Heart Failure (Agrawal et al. 2016; Panagiotou et al. 2017)
- Associated with pulmonary hypertension
- This can most easily be thought of as being due to the increased afterload seen by the right heart causing a vicious cycle of:
- Increased RV pressure causing over-dilatation
- Causing increased wall tension
- Resulting in impaired perfusion and
- Causing subsequent ischemia and further dilation and impairment
Infection (Meyer 2014)
- Many patients are on immunosuppressants
- As a potential precipitant of ILD flare as well as a clinical presentation that often consists of cough, shortness of breath, and fever, these patients should receive early empiric antibiotics
Coronary Artery Disease (Izbicki et al. 2009; Hubbard et al. 2008; Kizer et al. 2004)
- ILD patients are at higher risk for multi-vessel coronary artery disease
- OR 1.53 – 4.16
Lung Cancer (G Raghu, Nyberg, and Morgan 2004)
- ILD patients have a RR of 7 to 14 in developing Lung Cancer
- In multiple cohort studies of cancer free ILD patients, at 10 years between 4% and 15% had developed Lung Cancer
Diffuse Alveolar Hemorrhage (Lara and Schwarz 2010)
- This is usually the result of a small vessel vasculitis that presents with the constellation of hemoptysis, anemia, diffuse pulmonary infiltrates, and hypoxemic respiratory failure
- Up to 1/3 of patients can have no hemoptysis initially in the ED
Summary Approach to known ILD patient with shortness of breath
(Leuschner and Behr 2017)
Medications: Old and New Kids on the Block
- While the evidence for the management of ILD flares is weak and conflicting for most treatments, early empiric antibiotics has been unanimously agreed upon (Maher et al. 2015)
- Steroid use is complicated, with variable evidence on efficacy
- Bottom Line: Administer steroids in consultation with your Respirology consultants
- There are two new anti-fibrotic medications recently approved in Canada
- Pirfenidone (Esbriet): At one year, increased the number of patients with no decline in their pulmonary function by 59% (Noble et al. 2016)
- Nintedinab (Ofev): Similarly slowed disease progression (Richeldi et al. 2014)
- The most common side effect for both medications is nausea, vomiting, and diarrhea
- Rarer but more serious complications include:
- SJS and DRESS
- Hepatotoxicity
- Rarer but more serious complications include:
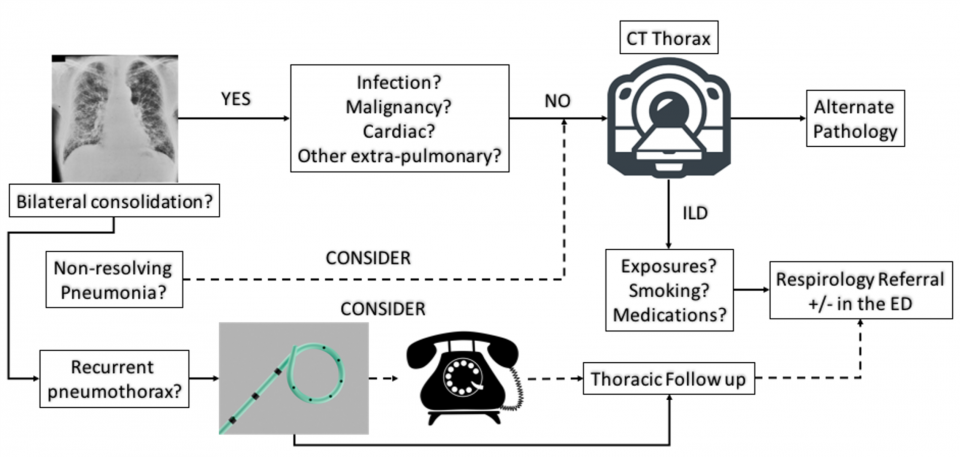
Approach to the newly suspected ILD patient
- If a patient has a suggestive CXR for ILD and no previous history, many patients should undergo a CT thorax (+/- PE study) for the following reasons:
- High risk for alternate pathology
- High risk for comorbid illnesses (ie. CHF)
- CT can be diagnostic for certain ILD
- May affect timeliness of management (can consider earlier consultation for steroid responsive etiologies like cryptogenic organizing pneumonias)
- Special cases to consider ILD:
- Recurrent, non-resolving pneumonia
- Consider cryptogenic organizing pneumonia
- Recurrent pneumothorax in a young woman
- Consider Lymphangioleiomyomatosis (LAM): a vascular malformation disorder that results in multiple cystic formations in the lung (Gupta et al. 2017)
- Recurrent, non-resolving pneumonia
Common Approach to ED Work Up
Approach to the Crashing ILD Patient
*Disclaimer: ILD is a variable entity with multiple different diseases under the term. As such, it is difficult to generalize across all entities. This section will discuss Idiopathic Pulmonary Fibrosis.
- IPF can have variable disease progression, as seen below:
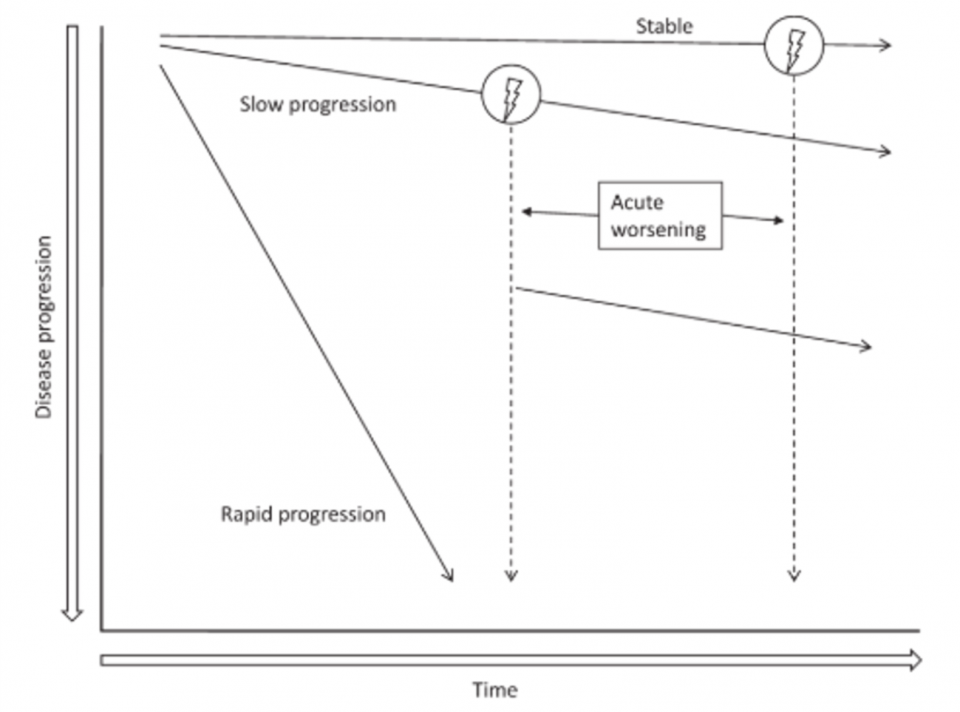 (Ganesh Raghu et al. 2011)
(Ganesh Raghu et al. 2011)
- Unfortunately, on the whole, these patients have a very poor prognosis if they need to be intubated:
- Maher et. al (2015) state that “Intubation and mechanical ventilation are NOT part of standard of care”
- The British Interstitial Lung Disease Guidelines (2008) state that there is a “near 100% in hospital mortality” for IPF patients intubated in the ICU
- Bottom Line: The prognosis is guarded for these IPF patients. A recent Pulmonary Function Test result is more useful in helping prognosticate.
- If the patient is on a transplant list, involve Respirology early in their care
- In the sick ILD patient in respiratory failure, provide the following management:
- Supplemental O2 with BiPAP
- Broad spectrum empiric antibiotics
- IV corticosteroids
- Acknowledging that, while steroids may not help, they likely won’t make things significantly worse and you are counting on them reversing whatever inflammation (if any) that is exacerbating the problem
- These patients are physiologically difficult intubations and should be assumed to also have a component of pulmonary hypertension
- If the patient requires intubation, follow these 4 steps to optimize success:
- Preoxygenate them with either two handed BVM or BiPAP
- Optimize their preload with a fluid bolus (500cc to 1L)
- Start early vasopressor infusions prior to intubation
- Consider half dose Ketamine induction
- +/- full Succinylcholine or Rocuronium for RSI
For a review of the Physiologically Difficult Airway, see Dr. Lauren Lacroix’s previous Grand Rounds post
Summary
- Shortness of Breath is not always an ILD flare
- Consider PE, CHF, Infection, CAD, Lung Cancer, and Diffuse Alveolar Hemorrhage
- Medications Old and New
- Antibiotics are a cornerstone to ILD flare management
- Consider steroid use in consultation with your local Respirologist
- Anti-fibrotic medications are providing new hope to ILD patients
- Consider hepatotoxicity and nausea/vomiting/diarrhea as potential side effects
- Approach to New ILD in the ED
- Consider obtaining a CT thorax to further delineate disease process and exclude dangerous alternate pathologies
- Consider ILD in your non-resolving pneumonia patients and recurrent pneumothorax patient
- Considerations in the Crashing ILD patient
- Physiologically difficult airways with hypoxemia and often pulmonary hypertension
- Optimize their hemodynamics pre-intubation and consider a reduced dose induction agent with full dose paralytics
References
Agrawal, Abhinav, Isha Verma, Varun Shah, Abhishek Agarwal, and Rutuja R Sikachi. 2016. “Cardiac Manifestations of Idiopathic Pulmonary Fibrosis.” Intractable & Rare Diseases Research 5 (2). doi:10.5582/irdr.2016.01023.
Antin-Ozerkis, Danielle. 2015. “Interstitial Lung Disease: A Clinical Overview and General Approach.” In Fishman’s Pulmonary Diseases and Disorders, 5e, edited by Michael A Grippi, Jack A Elias, Jay A Fishman, Robert M Kotloff, Allan I Pack, Robert M Senior, and Mark D Siegel. New York, NY: McGraw-Hill Education. accessmedicine.mhmedical.com/content.aspx?aid=1122360443.
Caminati, Antonella, Roberto Cassandro, and Sergio Harari. 2013. “Pulmonary Hypertension in Chronic Interstitial Lung Diseases.” European Respiratory Review 22 (129): 292–301. doi:10.1183/09059180.00002713.
Collard, Harold R, Christopher J Ryerson, Tamera J Corte, Gisli Jenkins, Yasuhiro Kondoh, David J Lederer, Joyce S Lee, et al. 2016. “Acute Exacerbation of Idiopathic Pulmonary Fibrosis. An International Working Group Report.” American Journal of Respiratory and Critical Care Medicine 194 (3): 265–75. doi:10.1164/rccm.201604-0801CI.
Gupta, Nishant, Geraldine A. Finlay, Robert M. Kotloff, Charlie Strange, Kevin C. Wilson, Lisa R. Young, Angelo M. Taveira-DaSilva, et al. 2017. “Lymphangioleiomyomatosis Diagnosis and Management: High-Resolution Chest Computed Tomography, Transbronchial Lung Biopsy, and Pleural Disease Management an Official American Thoracic Society/Japanese Respiratory Society Clinical Practice Guideline.” American Journal of Respiratory and Critical Care Medicine 196 (10): 1337–48. doi:10.1164/rccm.201709-1965ST.
Herzog, Erica L, Ami N Rubinowitz, and Mridu Gulati. 2015. “Idiopathic Interstitial Pneumonias Other Than Idiopathic Pulmonary Fibrosis Nonspecific Interstitial Pneumonia.” In Fishman’s Pulmonary Diseases and Disorders, edited by Michael A Grippi, Jack A Elias, Jay A Fishman, Robert M Kotloff, Allan I Pack, Robert M Senior, and Mark D Siegel. New York, NY: McGraw-Hill Education. accessmedicine.mhmedical.com/content.aspx?aid=1122360915.
Hubbard, Richard B., Chris Smith, Ivan Le Jeune, Jonathan Gribbin, and Andrew W. Fogarty. 2008. “The Association between Idiopathic Pulmonary Fibrosis and Vascular Disease: A Population-Based Study.” American Journal of Respiratory and Critical Care Medicine 178 (12): 1257–61. doi:10.1164/rccm.200805-725OC.
Izbicki, Gabriel, Itsik Ben-Dor, David Shitrit, Danielle Bendayan, Thomas K. Aldrich, Ran Kornowski, and Mordechai R. Kramer. 2009. “The Prevalence of Coronary Artery Disease in End-Stage Pulmonary Disease: Is Pulmonary Fibrosis a Risk Factor?” Respiratory Medicine 103 (9): 1346–49. doi:10.1016/j.rmed.2009.03.012.
Kizer, Jorge R., David A. Zisman, Nancy P. Blumenthal, Robert M. Kotloff, Stephen E. Kimmel, Robert M. Strieter, Selim M. Arcasoy, Victor A. Ferrari, and John Hansen-Flaschen. 2004. “Association between Pulmonary Fibrosis and Coronary Artery Disease.” Archives of Internal Medicine 164 (5): 551–56. doi:10.1001/archinte.164.5.551.
Lara, Abigail R., and Marvin I. Schwarz. 2010. “Diffuse Alveolar Hemorrhage.” Chest 137 (5): 1164–71. doi:10.1378/chest.08-2084.
Leuschner, Gabriela, and Jürgen Behr. 2017. “Acute Exacerbation in Interstitial Lung Disease.” Frontiers in Medicine 4: 176. doi:10.3389/fmed.2017.00176.
Maher, Toby M., Moira K. B. Whyte, Rachel K. Hoyles, Helen Parfrey, Yuuki Ochiai, Nicky Mathieson, Alice Turnbull, Nicola Williamson, and Bryan M. Bennett. 2015. “Development of a Consensus Statement for the Definition, Diagnosis, and Treatment of Acute Exacerbations of Idiopathic Pulmonary Fibrosis Using the Delphi Technique.” Advances in Therapy 32 (10): 929–43. doi:10.1007/s12325-015-0249-6.
Meyer, Keith C. 2014. “Immunosuppressive Agents and Interstitial Lung Disease: What Are the Risks?” Expert Review of Respiratory Medicine 8 (3): 263–66. doi:10.1586/17476348.2014.880054.
Mikolasch, T. A., H. S. Garthwaite, and J. C. Porter. 2016. “Update in Diagnosis and Management of Interstitial Lung Disease.” Clinical Medicine. Vol. 16. doi:10.7861/clinmedicine.16-6-s71.
Noble, Paul W, Carlo Albera, Williamson Z Bradford, Ulrich Costabel, Roland M Du Bois, Elizabeth A Fagan, Robert S Fishman, et al. 2016. “Pirfenidone for Idiopathic Pulmonary Fibrosis: Analysis of Pooled Data from Three Multinational Phase 3 Trials.” Eur Respir J 47: 243–53. doi:10.1183/13993003.00026-2015.
Panagiotou, Marios, Alistair C. Church, Martin K. Johnson, and Andrew J. Peacock. 2017. “Pulmonary Vascular and Cardiac Impairment in Interstitial Lung Disease.” European Respiratory Review 26 (143). doi:10.1183/16000617.0053-2016.
Raghu, G, F Nyberg, and G Morgan. 2004. “The Epidemiology of Interstitial Lung Disease and Its Association with Lung Cancer.” British Journal of Cancer, no. 2: 3–10. doi:10.1038/sj.bjc.6602061.
Raghu, Ganesh, Harold R. Collard, Jim J. Egan, Fernando J. Martinez, Juergen Behr, Kevin K. Brown, Thomas V. Colby, et al. 2011. “An Official ATS/ERS/JRS/ALAT Statement: Idiopathic Pulmonary Fibrosis: Evidence-Based Guidelines for Diagnosis and Management.” American Journal of Respiratory and Critical Care Medicine 183 (6): 788–824. doi:10.1164/rccm.2009-040GL.
Richeldi, Luca, Roland M Du Bois, Ganesh Raghu, Arata Azuma, Kevin K Brown, Ulrich Costabel, Vincent Cottin, et al. 2014. “Efficacy and Safety of Nintedanib in Idiopathic Pulmonary Fibrosis.” N Engl j Med 22: 2071–82. doi:10.1056/NEJMoa1402584.
Sprunger, D. B., A. L. Olson, T. J. Huie, E. R. Fernandez-Perez, A. Fischer, J. J. Solomon, K. K. Brown, and J. J. Swigris. 2012. “Pulmonary Fibrosis Is Associated with an Elevated Risk of Thromboembolic Disease.” European Respiratory Journal 39 (1): 125–32. doi:10.1183/09031936.00041411.
Travis, William D., Ulrich Costabel, David M. Hansell, Talmadge E. King, David A. Lynch, Andrew G. Nicholson, Christopher J. Ryerson, et al. 2013. “An Official American Thoracic Society/European Respiratory Society Statement: Update of the International Multidisciplinary Classification of the Idiopathic Interstitial Pneumonias.” American Journal of Respiratory and Critical Care Medicine 188 (6): 733–48. doi:10.1164/rccm.201308-1483ST.