IV fluid is a drug
Intravenous fluid administration is the second most common medical intervention next to supplemental oxygen.1 Over 200 million liters of normal saline (NS) is used in the United States annually.2 Intravenous fluids, like any other drug, should be given with thought and knowledge about the effects and sequelae to your patient.
The origin of IV fluids
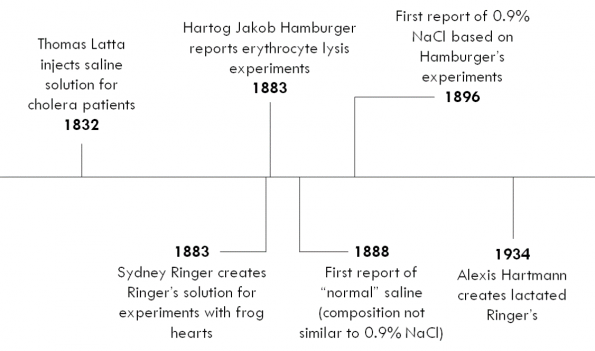
The earliest reported use of IV fluid dates back to 1832 during the cholera pandemic in England. A British physician named Thomas Latta noticed that “injecting a weak saline solution into the veins of the patient had the most wonderful and satisfactory effect.”3 He used a solution that was composed of “two drams of muriate of soda and two scruples of subcarbonate of soda in six pints of water.”4 Since 1832, many versions of salt solutions were created for intravenous therapy.
Where did normal saline come from?
The first reported use of the term “normal saline” was in 1888. The solution it was referring to did not resemble 0.9% saline. It is speculated that “normal saline” was used as colloquialism at the time with no scientific basis. The first reported use of 0.9% saline was in 1896. The justification for its use was based on experiments done by a Dutch physiologist named Hartog Jakob Hamburger. Hamburger did experiments examining the lysis of erythrocytes in solutions with different concentrations of saline. He found that a 0.9% saline solution resulted in the least amount of cell lysis, concluding that 0.9% saline was isotonic to human serum. The scientific evidence supporting the use of 0.9% saline in clinical practice seems to be based solely on this in vitro study.4
Physiologic effects of normal saline
To understand the effects of NS, we need to use the physicochemical theory of acid-base created by Canadian physiologist Peter Stewart.5,6 One of the underlying principles on which Stewart’s theory is based is electroneutrality, which means that positive and negative charges are always balanced.
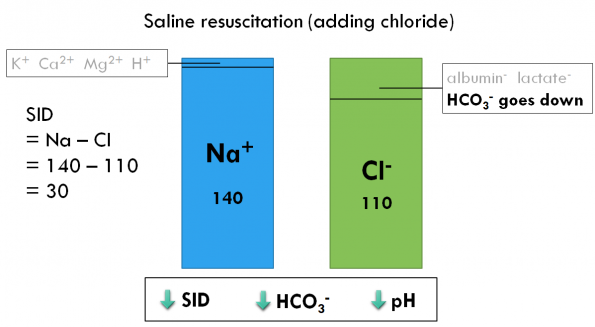
The predominant positive charge in our body is sodium, and the predominant negative charge is chloride. The difference between sodium and chloride is also known as the strong ion difference (SID). Strong ions are ions that dissolve readily in solution. A normal SID is about 40, and the SID is directly related to the acid-base status.
There are also small amounts of other positive charges such as potassium, calcium, magnesium, and hydrogen ions, as well as other negative charges such as albumin, lactate, and bicarbonate. Notably, Stewart’s theory states that hydrogen ions and bicarbonate ions are dependent variables, which means their concentrations vary depending on the concentrations of the other positive and negative charges. When we give normal saline, a disproportionate amount of chloride is added.
When the amount of chloride goes up, the amount of bicarbonate goes down, the SID goes down, and the pH also goes down, causing a hyperchloremic acidosis.
Why is hyperchloremic acidosis bad?
Numerous observational studies have shown that hyperchloremic acidosis is related to worse outcomes, including increased acute kidney injury and mortality.7–10 It has been shown that increased chloride concentrations in the renal tubules cause the release of adenosine from the macula densa, which causes afferent arteriolar resistance and reduced glomerular filtration.11
What is a balanced fluid?
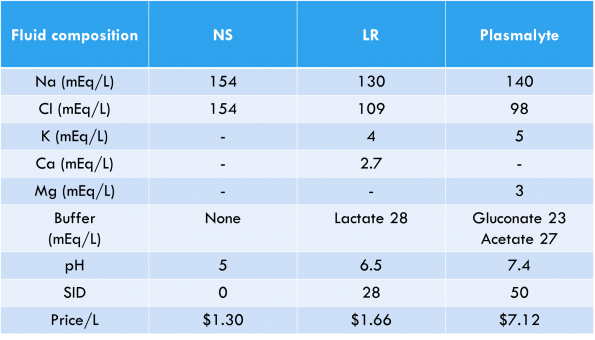
A balanced fluid is one that will not disrupt the body’s normal pH. A balanced solution has a SID that is similar to the physiologic SID of 40. This type of fluid is created by adding a buffer such as lactate in lactated Ringer’s (LR), or gluconate and acetate in Plasmalyte. Therefore, LR and Plasmalyte are balanced solutions whereas normal saline is not. The composition, properties, and pricing for NS, LR, and Plasmalyte are shown below.1,3,12
Why don’t we just use Lactated Ringer’s?
Two common reasons clinicians may hesitate to use LR are hyperkalemia and hyperlactatemia. These common misconceptions are clarified below.
Hyperkalemia
No study on this topic has ever demonstrated that LR significantly increases serum potassium. This makes sense for two reasons. First, the amount of potassium in LR (4 mEq/L) is negligible when distributed in the body. Second, 98% of total body potassium is stored inside cells. This means that the shift of potassium from intracellular to extracellular makes a much bigger impact on serum potassium than the 4 mEq/L of potassium in LR. Normal saline, which we established as consistently causing acidosis, is actually worse in the setting of hyperkalemia. The acidosis caused by NS will actually cause potassium to shift out of cells and cause an increase in serum potassium. This is supported by randomized studies in the renal transplant literature, which consistently demonstrate higher serum potassium levels in patients after receiving NS compared to LR.13–16
High lactate
Studies on this topic have shown that LR does increase serum lactate slightly.17 However, this increase in lactate is not clinically significant because it is not associated with acidosis.18 The lactate that we consider to be harmful is lactic acid, which dissociates into lactate and hydrogen ions, causing an acidosis. The lactate in LR is sodium lactate, which dissociates into lactate and sodium, which means it will never cause an acidosis. Lactate itself is not known to be clinically harmful. In fact, lactate is metabolized by the liver and used by the body as an energy source. Sodium lactate has been shown to increase cardiac output.19 Many clinicians use lactate to guide resuscitation and may be concerned that LR may have the potential to confound serum lactate levels. Unless the patient has profound hepatic dysfunction, lactate is quickly metabolized by the liver and the increases in lactate caused by LR should be negligible. Furthermore, the utility of lactate as a resuscitation marker is debatable and, ultimately, lactate should be just one of many data points a clinician uses to guide resuscitation.
When does fluid choice actually matter?
SALT-ED trial
In a recent large randomized trial of noncritically ill emergency department patients, there was no difference in the primary outcome of hospital-free days between saline and balanced crystalloids.2 There was a statistically significant difference in the secondary composite outcome of major adverse kidney events within 30 days (MAKE-30) in favor of balanced crystalloids. The composite outcome of MAKE-30 was composed of death, new renal-replacement therapy, and final creatinine ≥ 200% of baseline. The difference in the composite outcome, however, was driven by doubling of the creatinine – making the clinical significance of this difference debatable. While increases in creatinine are not desirable, the results of the SALT-ED trial suggest that for your average, non-critically ill patient who is getting just a liter of fluids, the fluid choice probably doesn’t matter that much.
SMART trial
In another recent large trial, critically ill patients admitted to the intensive care unit were randomized to saline and balanced crystalloids.29 There was a statistically significant difference in the primary outcome of MAKE-30 in favor or balanced crystalloids, and this difference was driven by mortality. Also, subgroup analysis showed that patients with sepsis and previous renal-replacement therapy and patients without traumatic brain injury benefited more from balanced crystalloids. Recognizing that the results of subgroup analyses must be interpreted with caution, these findings are consistent with physiology, previous observational data, and a recent meta-analysis.30 Overall, this study provides the best evidence to date to suggest that balanced crystalloids are better for critically-ill patients.
Brain-injury
Brain-injured patients (including traumatic brain injury, subarachnoid hemorrhage, subdural hemorrhage, intraparenchymal hemorrhage) should not receive hypotonic fluids because of the risk of cerebral edema. Therefore, saline (or Plasmalyte if available) would be preferable over lactated Ringer’s for brain-injured patients.
Metabolic alkalosis
Patients with metabolic alkalosis from vomiting (such as infants with pyloric stenosis and patients with bulimia or other hyperemesis syndromes) should receive saline to replenish their chloride and counteract the alkalosis.
Sepsis
Patients with sepsis are often acidotic and receive large volumes of fluid resuscitation. The results of the SMART trial combined with previous observational evidence suggest that patients with sepsis should receive balanced fluids.20,21 This group of critically-ill patients may stand to benefit the most from resuscitation with balanced fluids.
For a more in-depth review on the topic of Sepsis, please see Dr. Shannon Fernando’s Grand Rounds on the topic.
Diabetic ketoacidosis (DKA)
While the evidence for balanced fluids for patients with diabetic ketoacidosis has not shown differences in patient-oriented outcomes, it makes sense physiologically that they should receive balanced fluids. Small studies have shown faster resolution of acidosis and less hyperchloremia for balanced fluids compared to saline.22–24
Pancreatitis
Studies have shown that patients with pancreatitis have faster resolution of SIRS at 24 hours and decreased mortality when they received balanced fluids compared to saline.25–27 The American College of Gastroenterology Guidelines also recommends LR as the preferred fluid for acute pancreatitis.28
Take home points
- IV fluid is a drug; your choice of fluid should be tailored to the physiology of the patient.
- Normal saline causes a hyperchloremic acidosis, which is associated with AKI and increased mortality.
- High lactate and hyperkalemia are not valid reasons to avoid Ringer’s lactate.
- Avoid hypotonic solutions for brain-injured patients.
- Use balanced fluids for patients with sepsis, DKA, pancreatitis, renal failure, and any critical illness requiring large-volume (>2L) fluid resuscitation.
References
- McIntyre L, Taljaard M, McArdle T, et al. FLUID trial: A protocol for a hospital-wide open-label cluster crossover pragmatic comparative effectiveness randomised pilot trial. BMJ Open. 2018;8(8):1-11. doi:10.1136/bmjopen-2018-022780
- Self WH, Semler MW, Wanderer JP, Wang L, Byrne DW. Balanced Crystalloids versus Saline in Noncritically Ill Adults. N Engl J Med. 2018;378(9):819-828. doi:10.1056/NEJMoa1711584
- Finfer S, Myburgh J, Bellomo R. Intravenous fluid therapy in critically ill adults. Nat Rev Nephrol. 2018;14(9):541-557. doi:10.1038/s41581-018-0044-0
- Awad S, Allison SP, Lobo DN. The history of 0.9% saline. Clin Nutr. 2008;27(2):179-188. doi:10.1016/j.clnu.2008.01.008
- Burdett E, Roche A, MG M. Hyperchloremic Acidosis: Pathophysiology and Clinical Impact. Transfus Altern Transfus Med. 2008;5(4):424-430. doi:10.1111/j.1778-428x.2003.tb00297.x
- Kaplan L, Frangos S. Clinical review: Acid-base abnormalities in the intensive care unit. Crit care. 2005;9(2):198-203. doi:10.1186/cc2912
- Yunos NM, Hegarty C, Story D, Ho L, Bailey M, Bellomo R. Association Between a Chloride-Liberal vs Chloride-Restrictive Intravenous Fluid Administration Strategy and Kidney Injury. JAMA. 2012;308(15):1566. doi:10.1001/jama.2012.13356
- Shaw AD, Raghunathan K, Peyerl FW, Munson SH, Paluszkiewicz SM, Schermer CR. Association between intravenous chloride load during resuscitation and in-hospital mortality among patients with SIRS. Intensive Care Med. 2014;40(12):1897-1905. doi:10.1007/s00134-014-3505-3
- Sen A, Keener CM, Sileanu FE, et al. Chloride content of fluids used for large-volume resuscitation is associated with reduced survival. Crit Care Med. 2017;45(2):e146-e153. doi:10.1097/CCM.0000000000002063
- Mccluskey SA, Karkouti K, Wijeysundera D, Minkovich L, Tait G, Beattie WS. Hyperchloremia after noncardiac surgery is independently associated with increased morbidity and mortality: A propensity-matched cohort study. Anesth Analg. 2013;117(2):412-421. doi:10.1213/ANE.0b013e318293d81e
- Chowdhury AH, Cox EF, Francis ST, Lobo DN. A randomized, controlled, double-blind crossover study on the effects of 2-L infusions of 0.9% saline and plasma-lyte® 148 on renal blood flow velocity and renal cortical tissue perfusion in healthy volunteers. Ann Surg. 2012;256(1):18-24. doi:10.1097/SLA.0b013e318256be72
- Smith CA, Duby JJ, Utter GH, Galante JM, Scherer LA, Schermer CR. Cost-minimization analysis of two fluid products for resuscitation of critically injured trauma patients. Am J Heal Pharm. 2014;71(6):470-475. doi:10.2146/ajhp130295
- Weinberg L, Harris L, Bellomo R, et al. Effects of intraoperative and early postoperative normal saline or Plasma-Lyte 148 ® on hyperkalaemia in deceased donor renal transplantation: A double-blind randomized trial. Br J Anaesth. 2017;119(4):606-615. doi:10.1093/bja/aex163
- Modi MP, Vora KS, Parikh GP, Shah VR. A Comparative Study of Impact of Infusion of Ringer ’ s Lactate Solution Versus Normal Saline on Acid – Base Balance and Serum Electrolytes. Saudi J Kidney Dis Transplant. 2012;23(1):135-137.
- Khajavi MR, Etezadi F, Moharari RS, et al. Effects of normal saline vs. lactated ringer’s during renal transplantation. Ren Fail. 2008;30(5):535-539. doi:10.1080/08860220802064770
- O’Malley CMN, Frumento RJ, Hardy MA, et al. A randomized, double-blind comparison of lactated ringer’s solution and 0.9% NaCl during renal transplantation. Anesth Analg. 2005;100(5):1518-1524. doi:10.1213/01.ANE.0000150939.28904.81
- Zitek T, Skaggs ZD, Rahbar A, Patel J, Khan M. Does Intravenous Lactated Ringer’s Solution Raise Serum Lactate? J Emerg Med. 2018;55(3):313-318. doi:10.1016/j.jemermed.2018.05.031
- Ross SW, Christmas AB, Fischer PE, et al. Impact of common crystalloid solutions on resuscitation markers following Class I hemorrhage: A randomized control trial. J Trauma Acute Care Surg. 2015;79(5):732-740. doi:10.1097/TA.0000000000000833
- Ichai C, Orban JC, Fontaine E. Sodium lactate for fluid resuscitation: The preferred solution for the coming decades? Crit Care. 2014;18(4):12-13. doi:10.1186/cc13973
- Raghunathan K, Shaw A, Nathanson B, et al. Association between the choice of IV crystalloid and in-hospital mortality among critically Ill adults with sepsis. Crit Care Med. 2014;42(7):1585-1591. doi:10.1097/CCM.0000000000000305
- Emrath ET, Fortenberry JD, Travers C, McCracken CE, Hebbar KB. Resuscitation with Balanced Fluids Is Associated with Improved Survival in Pediatric Severe Sepsis. Crit Care Med. 2017;45(7):1177-1183. doi:10.1097/CCM.0000000000002365
- Chua HR, Venkatesh B, Stachowski E, et al. Plasma-Lyte 148 vs 0.9% saline for fluid resuscitation in diabetic ketoacidosis. J Crit Care. 2012;27(2):138-145. doi:10.1016/j.jcrc.2012.01.007
- Mahler SA, Conrad SA, Wang H, Arnold TC. Resuscitation with balanced electrolyte solution prevents hyperchloremic metabolic acidosis in patients with diabetic ketoacidosis. Am J Emerg Med. 2011;29(6):670-674. doi:10.1016/j.ajem.2010.02.004
- Van Zyl DG, Rheeder P, Delport E. Fluid management in diabetic-acidosis-Ringer’s lactate versus normal saline: A randomized controlled trial. Q J Med. 2012;105(4):337-343. doi:10.1093/qjmed/hcr226
- Aboelsoud MM, Siddique O, Morales A, Seol Y, Al-Qadi MO. Fluid Choice Matters in Critically-ill Patients with Acute Pancreatitis: Lactated Ringer’s vs. Isotonic Saline. R I Med J. 2016;99(10):39-42.
- Choosakul S, Harinwan K, Chirapongsathorn S, et al. Comparison of normal saline versus Lactated Ringer’s solution for fluid resuscitation in patients with mild acute pancreatitis, A randomized controlled trial. Pancreatology. 2018;18(5):507-512. doi:10.1016/j.pan.2018.04.016
- Iqbal U, Anwar H, Scribani M. Ringer’s lactate versus normal saline in acute pancreatitis: A systematic review and meta-analysis. J Dig Dis. 2018;19(6):335-341. doi:10.1111/1751-2980.12606
- Tenner S, Baillie J, Dewitt J, Vege SS. American college of gastroenterology guideline: Management of acute pancreatitis. Am J Gastroenterol. 2013;108(9):1400-1415. doi:10.1038/ajg.2013.218
- Semler MW, Self WH, Wanderer JP, et al. Balanced Crystalloids versus Saline in Critically Ill Adults. N Engl J Med. 2018;378(9):829-839. doi:10.1056/NEJMoa1711584
- Xue M, Zhang X, Liu F, et al. Effects of chloride content of intravenous crystalloid solutions in critically ill adult patients: a meta-analysis with trial sequential analysis of randomized trials. Ann Intensive Care. 2019;9(1). doi:10.1186/s13613-019-0506-y