Dr. Krishan Yadav is an FRCPC Emergency Medicine Physician with a Masters in Clinical Epidemiology, and here he helps to provide some great insights and opinions into the recently published CRASH-3 trial.
Tranexamic Acid (TXA)
TXA is an inexpensive and readily available medication that is a synthetic analog of lysine. It acts as a competitive inhibitor by binding to lysine receptors on plasminogen and reducing the formation of plasmin. This ultimately reduces the amount of plasmin available to degrade fibrin (clot) and thus decrease bleeding.
Rationale
In patients with traumatic brain injury (TBI), the CRASH-3 investigators hypothesized that early administration of TXA would reduce or prevent intracranial hemorrhage expansion and subsequently reduce mortality.
PICOS
Patients
- Adults with TBI within 3 hours of injury
- The clinician is substantially uncertain regarding the appropriateness of TXA
- GCS <12 or any intracranial bleeding on CT scan
- No major extracranial bleeding
Intervention
- TXA 1g IV over 10 minutes, followed by 1g IV over 8 hours
Comparator
- Placebo
Outcome
- Primary: head injury-related death in hospital within 28 days (for patients treated within 3 hours of injury)
- Secondary:
- Head-injury related death within 24 hours
- All cause-mortality
- Cause-specific mortality
- Disability
- MI, stroke, DVT, PE (vascular occlusive events)
- Seizures
- Neurosurgery
- Days in ICU
- Adverse events within 28 days
Setting
- 175 hospitals in 29 countries
- Canadian sites: 1
- US sites: 0
Results
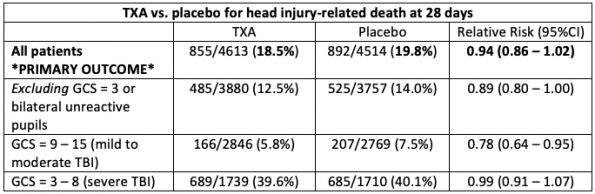
The relative risk (RR) is the patient’s risk of head injury-related death at 28 days if they had received TXA versus if they had received placebo. Focusing on the primary outcome (i.e. in all patients), we are thus 95% confident that the risk of head-injury related death at 28 days lies somewhere between 0.86 and 1.02. This confidence interval includes a RR = 1; in other words, no appreciable benefit with TXA relative to placebo. While there is a trend toward benefit, because the confidence interval includes 1, we cannot be certain of a benefit with TXA.
Data may be analyzed in several different ways – for example, via subgroups. Importantly, these were pre-specified. However, if you look at enough subgroups, eventually you may find a signal where the result is significant. This was the case in the subset of patients with mild to moderate TBI (RR 0.78; 0.64 – 0.95) and in head injury-related death within 24 hours (RR 0.81; 0.69 – 0.95). Subgroup analyses are hypothesis-generating, and should drive further research (they are not sufficient rationale to change clinical practice).
Importantly, there were no findings of increased adverse events in the TXA arm.
Caveats
- Primary outcome was changed to: head injury-related death within 28 days for patients treated within 3 hours of injury (was previously within 8 hours)
- The sample size was thus increased from 10,000 to 13,000 patients to ensure approximately 10,000 patients treated within 3 hours
- Ultimately there were 9,127 patients randomized within 3 hours of injury
Final thoughts (What does this mean for my practice?)
The CRASH-3 investigators conclude TXA is “safe in patients with TBI and that treatment within 3 hours of injury reduces head injury-related deaths.”
This conclusion is misleading. TXA reduces head-injury related mortality in a subgroup of patients: specifically, those with a GCS 9 – 15, and those at 24 hours.
For patients with TBI, I would focus on airway and increased intracranial pressure management. Following this, I would then administer TXA. The rationale is that it is safe, and that a certain subset of TBI patients may benefit from it (again: further research required). However, the rationale should not be “because CRASH-3 shows TXA reduces head injury-related deaths.”
In other words, I would consider administering TXA, but not for the wrong reasons.
Disclaimer: my wording has been intentionally careful – the above are recommendations for my own practice. Social media, podcasts, and so on are fantastic resources. However, if you come across something that may genuinely change your practice, then I strongly encourage you to read the article for yourself: https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(19)32233-0/fulltext


