This post takes a deep dive into the pathophysiology, presentation and management of electrical storm in the emergency department. The management focuses on patients with implantable cardioverter-defibrillators receiving multiple shocks, patients with polymorphic ventricular tachycardia and refractory ventricular fibrillation.
Electrical storm (ES) has varying definitions in the literature depending on whether or not the patient has an ICD (an implantable cardioverter-defibrillator). In patients without an ICD, electrical storm is most commonly defined as the occurrence of 3 or more episodes of sustained ventricular tachycardia (VT) or ventricular fibrillation (VF) over a 24 hour period, where sustained VT refers to VT lasting greater than 30 seconds or requiring termination due to hemodynamic compromise in less than 30 seconds.
In patients with an ICD, the definition of electrical storm is widely accepted as 3 appropriate detections of ventricular arrhythmia that lead to ICD therapies (either anti-tachycardia pacing or shock) in 24 hours. Of note this definition is not comprehensive as it does not account for VT that fails to terminate with appropriate ICD therapy and remains undetected by the patient, or VT slower than the programmed detection rate of the ICD.
Pathophysiology
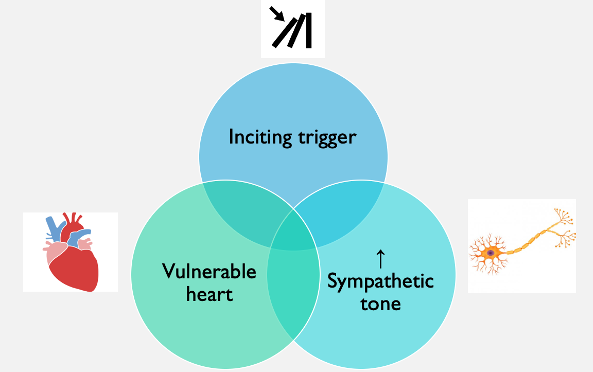
The underlying pathophysiology of ES is poorly characterized but the working theory is that 3 key factors are at play:
- An inciting event or trigger
- A vulnerable heart (the vast majority of patients who develop ES have underlying heart disease)
- A disruption in the sympathetic nervous system that results in enhanced sympathetic tone1
Arrhythmias Encountered in Electrical Storm
The relative distribution of arrhythmias encountered in ES is:
- Monomorphic VT- 86-97%
- VF- 1-21%
- Mixed VT/VF- 3-14%
- Polymorphic VT 2-8%
Clinical Presentation
There is a wide range of clinical presentations of ES. In the patient without an ICD, ES can include:
- Minor symptoms such as palpitations, presyncope or syncope
- Hemodynamically unstable ventricular arrhythmias
- Cardiac arrest
In the patient who has an ICD, they typically present complaining of multiple ICD discharges. However, ICD patients may have VT below detect may present similarly to the patient without an ICD.
What is VT below detect?
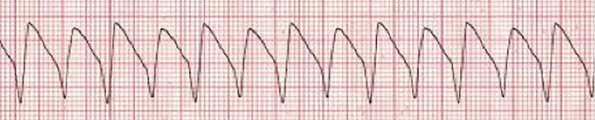
When a patient receives an ICD, it is programmed by the cardiologist or electrophysiologist to detect any rate greater than 175-180 beats per minute, and deliver a treatment (shock or anti-tachycardia pacing). This threshold is largely based on expert opinion. However, ICD patients may have VT at a rate less than this, for example at 160 beats per minute. The ICD doesn’t recognize this as VT and therefore will not shock the patient and terminate the VT
- This is what is known as VT below detect.
- It is important for us to recognize this because if we look up at the monitor and see that the patient is in VT, but the ICD is not discharging, this may not be due to malfunction of the ICD but rather the VT is below the level that the ICD is programmed to detect.
Appropriate vs Inappropriate ICD Shocks
When we think about ICD shocks, they can be divided into:
- Appropriate (70%)
- An ICD shock delivered for a malignant ventricular arrhythmia
- Ventricular fibrillation
- Monomorphic ventricular tachycardia
- Polymorphic ventricular tachycardia
- An ICD shock delivered for a malignant ventricular arrhythmia
- Inappropriate (30%)
- An ICD shock delivered in the absence of a malignant ventricular arrhythmia
- Causes of inappropriate shocks
- Non-malignant supraventricular arrhythmias- atrial fibrillation, sinus tachycardia etc.
- Electromagnetic interference
- Hardware malfunction- lead fracture, displacement
- T wave or atrial oversensing
- Muscle twitching 2
It is important to identify if inappropriate ICD shocks are occurring, as this will influence management. Moreover, while an appropriately functioning ICD will terminate life-threatening arrhythmias, it does not prevent them from recurring.
Causes of Electrical Storm
The causes of electrical storm can be classified into 2 categories:
- Scar-mediated reentry due to previous MI
- Reversible causes which including:
- Acute ischemia
- Acute decompensated heart failure
- Electrolyte abnormalities (primarily hypokalemia and hypomagnesemia)
- Drug toxicity or overdose, or recent changes to antiarrhythmic medication
- Sepsis
- Thyrotoxicosis
It should be noted an inciting factor is not identified in up to 66% of cases. 3
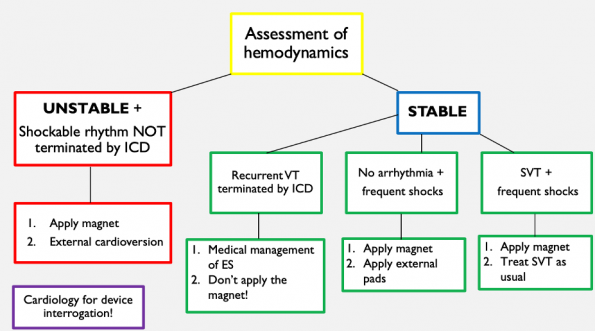
Approach to the ICD patient with Recurrent Shocks
After you perform a full history and physical examination and initiate investigations to assess for reversible causes, you should proceed with hemodynamic assessment of the patient.
- Hemodynamically unstable + shockable rhythm on the monitor that is not being terminated by the ICD requires external electrical cardioversion. The pads should ideally be placed in the A-P positioning. However, if you don’t have time to do this due to the emergent nature of the situation, place the pads on the chest and in the axilla 8-10cm away from the ICD to prevent ICD dysfunction.
If the patient is hemodynamically stable, have a look at the monitor and determine which of the three scenarios you find yourself in.
- Recurrent VT, appropriately terminated by the ICD- this means the device is providing appropriate shocks. Leave the ICD alone- it’s working properly! Proceed to the medical management of electrical storm.
- No arrhythmias + recurrent shocks – it is possible there is a lead fracture in the device. Apply the magnet, place external pads on the patient in the event they do have an arrhythmia that requires cardioversion.
- Arrhythmias other than VT (ex SVT or atrial fibrillation) + recurrent shocks- apply the magnet and treat the supraventricular tachyarrhythmia as you would normally.
In ALL cases, cardiology needs to be called for device interrogation.
Management of monomorphic VT storm in the ICD patient
Beta blockers
First line pharmacotherapy includes suppression of increased sympathetic tone encountered in ES. There is very little guidance from the literature as to which type of beta blocker to use.
Chazidou et al (2018) published a randomized trial of 60 ICD patients with hemodynamically stable ES who received either propranolol and amiodarone or metoprolol and amiodarone within 24 hours of ES presentation for 48 hours.
Findings:
- Patients who received propranolol had a significantly shorter time to termination of ventricular arrhythmias (3h in the propranolol group vs 18h in the metoprolol group) with a higher proportion of patients free of ventricular arrhythmias at 24 hours
- Patients who received propranolol also demonstrated lower rates of ventricular arrhythmias, fewer ICD discharge and a significantly shorter hospital stay compared to the metoprolol group. 4
Limitations
- One small, single centre trial with no patient centred outcomes
Experts in the field recommend any oral beta blocker is acceptable, as we have insufficient data to tell us that any one beta blocker is better than another when assessing long term outcomes. IV beta blockers should be avoided in the hemodynamically stable patient as this can cause significant myocardial suppression and hypotension in an already potentially sick heart (give oral beta blocker instead of IV). If the patient is hemodynamically unstable, you can consider using a short acting beta blocker such as esmolol.
Antiarrhythmics- Amiodarone
Amiodarone is a class III antiarrhythmic medication with potassium channel blocking effects, which results in lengthening the cardiac action potential and increased refractoriness of the cardiac myocytes. To a lesser extent, amiodarone also displays features of other antiarrhythmic classes such as sodium channel blockade, beta blockade and calcium channel blockade. Because of its multiple mechanisms of action, particularly the sympathetic blockade effects, amiodarone theoretically is felt to be superior to other antiarrhythmics for the treatment of ES.
The evidence for amiodarone in refractory VF and VT comes from 3 trails conducted in the mid 1990s by the IV Amiodarone Multicentre Trial Group. They found that in patients who had hemodynamically destabilizing ventricular arrhythmias refractory to procainamide, bretylium and lidocaine, amiodarone resulted in significant reductions in recurrence of ventricular arrhythmias, particularly in the higher dosing ranges. 5-7
Amiodarone is considered the standard of care and antiarrhythmic choice for monomorphic VT storm for the ICD patient proposed in the literature and by expert opinion.
Neuraxial Modulation- Stellate Ganglion Block
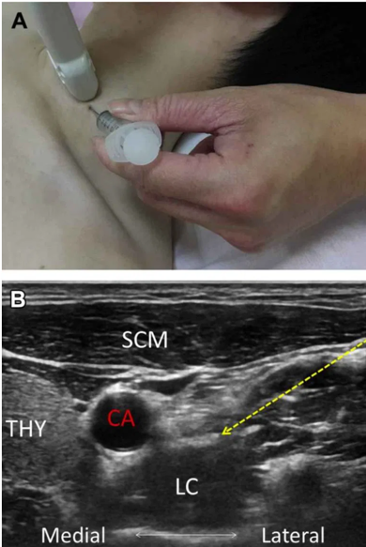
Modulating the autonomic nervous system for the purposes of preventing arrhythmias is an emerging therapeutic modality in electrical storm. Stellate ganglia are a collection of nerves in the sympathetic nervous system; they are found on both sides of the neck and provide sympathetic outflow to the heart. A stellate ganglion block is a percutaneous nerve block procedure done at the bedside which blocks sympathetic outflow to the heart by blocking the stellate ganglion with local anesthetic.
The procedure is commonly performed by anesthesiologists for complex regional pain syndrome and has also been reported to treat refractory angina, phantom limb pain, vascular insufficiency and hyperhidrosis. Several different approaches to the stellate ganglion block including the landmark approach, US guided and under fluoroscopy.
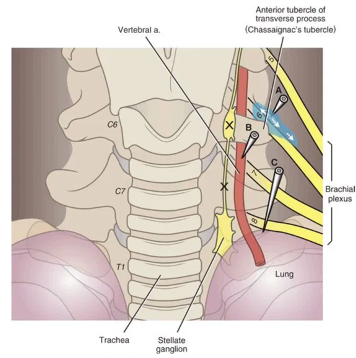
Meng et al (2017) published a systematic review examining the efficacy of stellate ganglion blocks in managing electrical storm. It included 38 patients with electrical storm who received a SGB.
Findings:
- Significant reduction in number of episodes of ventricular arrhythmias from 12.4 to 1.1 episodes/ day
- Significant reduction in number of external or ICD shocks from 10 to 0.05 shocks/ day
- Following SGB 80.6% of patients survived to hospital discharge 8
Management of Polymorphic Ventricular Tachycardia (PMVT)
PMVT is ventricular tachycardia with varying QRS morphology from beat to beat. PMVT can be further subdivided into PMVT with long QTc (Torsades de pointes), which may either be acquired or congenital in nature, or normal QTc PMVT.
PMVT + long QTc (Torsades de Pointes)
Acquired
- Majority of adult cases of torsades are acquired and torsades is precipitated by bradycardia.
- 3 main causes of acquired long QTc: ischemia, medications and electrolyte abnormalities.
- Treatment:
- Cardioversion if hemodynamically unstable
- Magnesium
- Replacement of electrolytes
- Stop offending agents
- Avoidance of antiarrhythmics as these may prolong the QTc further
- Isoproterenol (beta agonist, increases HR and shortens ventricular repolarization)
- Overdrive pacing
Congenital
- Much less commonly, long QTc may be congenital in nature due to loss of function of potassium channels or gain of function of sodium channels and in these instances, PMVT is precipitated by tachycardia
- Treatment
- Cardioversion if hemodynamically unstable
- Magnesium
- Beta blockers such as esmolol to slow down the heart rate
PMVT + normal QTc
PMVT and a normal QTc at baseline, the underlying etiology in the vast majority of these cases is myocardial ischemia. Treatment includes:
- Cardioversion if hemodynamically unstable
- Usual treatment of their ischemic event and emergency revascularization therapy
- Consideration of amiodarone or lidocaine
- Beta blocker once stabilized (often started in the CCU)
Summary: Algorithm for the Management of Ventricular Tachycardia Electrical Storm
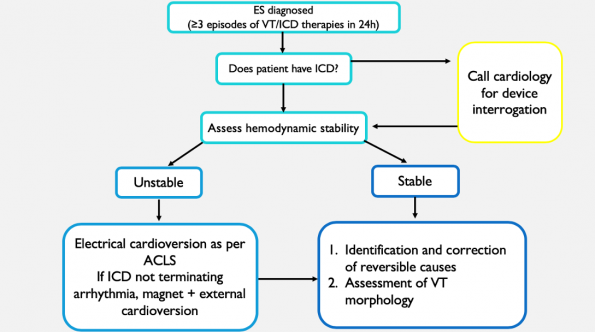
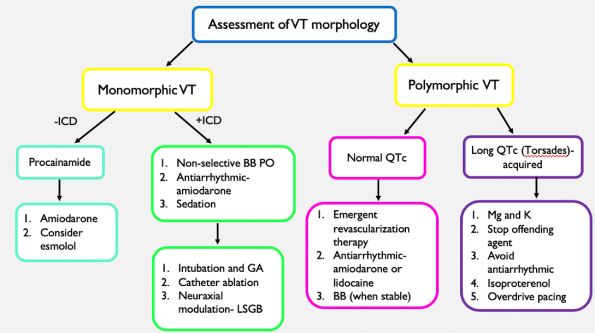
Management of Shock Refractory VF
Shock refractory ventricular fibrillation (VF) is VF that is resistant to at least three defibrillation attempts, 300 mg of amiodarone, and 3 rounds of epinephrine with no return of spontaneous circulation (ROSC) after > 10 min of CPR. The management of shock refractory VF goes beyond ACLS guidelines and unfortunately, there is no established evidence-based standard of care for these patients.
Several strategies have been proposed that aim to convert shock refractory VF to a perfusing rhythm:
- Beta blockade with esmolol
- Withholding epinephrine
- Dual sequential external defibrillation
Beta Blocker (esmolol)
- In shock refractory VF, there is a huge increase in sympathetic tone that results in increased myocardial O2 demand, exacerbation of myocardial ischemia and depression of the VF threshold.
- Moreover, the increase in sympathetic in drive is partially driven by giving more epinephrine as part of standard ACLS care.
- Esmolol is a fast onset, short-acting, cardio-selective beta blocker that aims to attenuate the sympathetic drive and increase the fibrillation threshold.
Driver et al (2014) published a single centre, retrospective study in the US comparing 6 patients who got esmolol after usual ACLS care to 19 controls who just got usual ACLS care.
Findings:
- In the esmolol group, all 6 patents achieved ROSC, and 50% of these patients survived to discharge with good neurologic outcome despite prolonged CPR times of ~1 hour.
- In the usual ACLS group, only 11% had survival to discharge good neurologic outcome9
Lee et al (2016) reported a single centre, retrospective study conducted in Korea that compared 16 patients with OHCA in shock refractory VF who got usual ACLS care and esmolol to 25 patients who just received usual ACLS care.
Findings
- Significantly improved rates of sustained ROSC with 56% in the esmolol group compared to 16% of patients in the usual ACLS group
- Survival with good neurological outcomes at 30 days, 3 months and at 6 months were >2-fold better in the esmolol group, compared to the control group, although these increases were not statistically significant. 10
Dosing of esmolol used in these initial studies was 500mcg/kg bolus followed by an infusion at a maximum rate of 50-100mcg/kg/min and this is what I would recommend for our patients. Remember that esmolol has a very short half life of only 9 minutes, so it should be given as a bolus followed by an infusion! Additionally, in cases of shock refractory VF, I would give a maximum of 3 rounds of epinephrine as epinephrine will exacerbate the increased sympathetic drive and perpetuate further VF.
Dual Sequential External Defibrillation
- Dual external defibrillation refers to the use of 2 separate defibrillators to deliver 2 electrical currents through the heart nearly simultaneously and it has been postulated as a method for improving treatment of patients who are in shock refractory VF.
- Dual defibrillation comes in 2 flavours:
- Simultaneous defibrillation whereby the shock button on both defibrillators is pressed at the same time
- Sequential defibrillation whereby the shock button is pressed in sequential manner
- There have been isolated case reports that if you perform dual defibrillation simultaneously right down to the millisecond, you can damage the motherboard of the defibrillators. Experts in this field recommend dual sequential defibrillation or DSED.
Why is Dual Defibrillation Effective?
There are several theories as to why subsequent shocks are failing to terminate VF in shock refractory VF:
- As VF persists, the energy required to defibrillate increases as a function of time.
- There is insufficient energy supplied to terminate VF.
- As the resuscitation progresses you get more hypoxia, acidosis and catecholamine surges which increase myocardial O2 consumption making it harder to successfully defibrillate the ventricle.
There are 3 theories as to why dual external defibrillation may be effective in shock refractory VF
- Setting up theory- that the first shock lowers the defibrillation threshold, increasing the likelihood of success of the second shock in terminating VF.
- More power theory- that higher energy is required and used to terminate VF.
- Multiple vector theory- that by adding a second defibrillator, you are increasing the number of vectors that the electricity uses to reach the heart muscle, increasing the chance that VF is terminated.
Cheskes et al (2019) recently published a retrospective observation study of 252 out of hospital cardiac arrest patients with refractory VF in 4 Canadian EMS agencies. They sought to explore the relationship between type of defibrillation (standard vs duel sequential), the number of defibrillation attempts and VF termination. VF termination was assessed in two ways: either any form of termination (ROSC, PEA or asystole) and VF termination specifically to ROSC.
Findings:
- VF termination (to either ROSC, PEA or asystole) and VF termination to ROSC was similar between standard defibrillation and dual sequential.
- When dual sequential terminated VF to ROSC, it did so with a single DSED attempt in 66.7% of cases.
- When early defibrillation attempts were considered (so attempts 4-8), VF termination and VF termination to ROSC was higher in the dual sequential group.
- When later defibrillation attempts were considered (so attempts 9-17) VF termination was higher in the dual sequential group but ROSC was rare in either strategy with only one person from either group actually achieving ROSC. 11
While this is only observational data and doesn’t take into account any patient-centered outcomes, the finding of improved VF termination to ROSC with fewer preceding standard shocks suggests that successful resuscitation with dual sequential defibrillation may be a time-dependent variable with greater success EARLIER on in the resuscitation.
The Double Sequential External Defibrillation for Refractory VF (DOSEVF) trial is an ongoing pre-hospital, multicentre cluster RCT comparing dual sequential external defibrillation, vector change external defibrillation and standard defibrillation in patients with OCHA presenting in shock refractory VF. The primary outcome of this study will be survival to hospital discharge. The hope is that this trial will give us some further guidance as to whether these alternate defibrillation strategies for shock refractory will impact patient-centred, clinical outcomes.
Summary: Algorithm for Shock Refractory VF
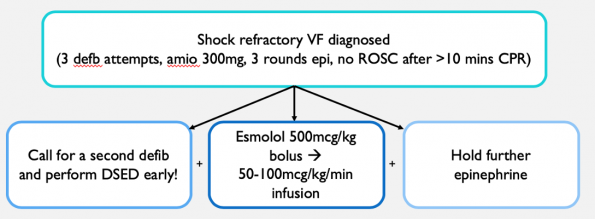
Citations
- Geraghty, L., Santangeli, P., Tedrow, U. B., Shivkumar, K., & Kumar, S. (2019). Contemporary management of electrical storm. Heart, Lung and Circulation, 28(1), 123-133.
- Iftikhar, S., Mattu, A., & Brady, W. (2016). ED evaluation and management of implantable cardiac defibrillator electrical shocks. The American journal of emergency medicine, 34(6), 1140-1147.
- Grieco, D., Borrelli, A., de Ruvo, E., Scara’, A., Sciarra, L., Della Bona, R., … & Calo’, L. (2019). Treatment of the arrhythmic storm. European Heart Journal Supplements, 21(Supplement_B), B23-B24.
- Chatzidou, S., Kontogiannis, C., Tsilimigras, D. I., Georgiopoulos, G., Kosmopoulos, M., Papadopoulou, E., … & Rokas, S. (2018). Propranolol versus metoprolol for treatment of electrical storm in patients with implantable cardioverter-defibrillator. Journal of the American College of Cardiology, 71(17), 1897-1906.
- Kowey, P. R., Levine, J. H., Herre, J. M., Pacifico, A., Lindsay, B. D., Plumb, V. J., … & Scheinman, M. M. (1995). Randomized, double-blind comparison of intravenous amiodarone and bretylium in the treatment of patients with recurrent, hemodynamically destabilizing ventricular tachycardia or fibrillation. Circulation, 92(11), 3255-3263.
- Levine, J. H., Massumi, A., Scheinman, M. M., Winkle, R. A., Platia, E. V., Chilson, D. A., … & Intravenous Amiodarone Multicenter Trial Group. (1996). Intravenous amiodarone for recurrent sustained hypotensive ventricular tachyarrhythmias. Journal of the American College of Cardiology, 27(1), 67-75.
- Scheinman, M. M., Levine, J. H., Cannom, D. S., Friehling, T., Kopelman, H. A., Chilson, D. A., … & Kowey, P. R. (1995). Dose-ranging study of intravenous amiodarone in patients with life-threatening ventricular tachyarrhythmias. Circulation, 92(11), 3264-3272.
- Meng, L., Tseng, C. H., Shivkumar, K., & Ajijola, O. (2017). Efficacy of stellate ganglion blockade in managing electrical storm: a systematic review. JACC: Clinical Electrophysiology, 3(9), 942-949.
- Driver, B. E., Debaty, G., Plummer, D. W., & Smith, S. W. (2014). Use of esmolol after failure of standard cardiopulmonary resuscitation to treat patients with refractory ventricular fibrillation. Resuscitation, 85(10), 1337-1341.
- Lee, Y. H., Lee, K. J., Min, Y. H., Ahn, H. C., Sohn, Y. D., Lee, W. W., … & Park, S. O. (2016). Refractory ventricular fibrillation treated with esmolol. Resuscitation, 107, 150-155.
- Cheskes, S., Wudwud, A., Turner, L., McLeod, S., Summers, J., Morrison, L. J., & Verbeek, P. R. (2019). The impact of double sequential external defibrillation on termination of refractory ventricular fibrillation during out-of-hospital cardiac arrest. Resuscitation, 139, 275-281.



