The diagnosis of subarachnoid hemorrhage (SAH) carries with it remarkable morbidity and mortality.
- Case fatality rates approach 50% 1,2
- Of those who survive, half will experience chronic disability and a significant reduction in health-related quality of life1,2
Given this high rate of negative sequelae, making an accurate and timely diagnosis is of distinct importance. The issue is that subarachnoid hemorrhage represents a true ‘needle in a haystack’ diagnosis. While the chief complaint of headache accounts for 2% of all ED visits,3,4 subarachnoid hemorrhage occurs in 1 out of every 100 headaches.4,5 For the average emergency physician, this translates to diagnosing less than 50 subarachnoid hemorrhages over the course of a career!
Presentation
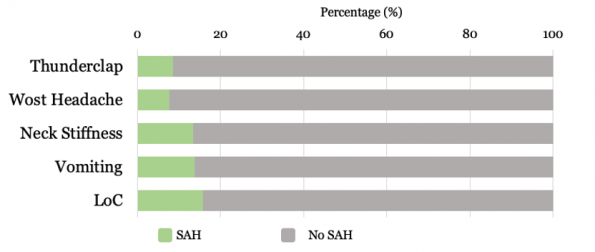
Like most conditions, subarachnoid hemorrhage has a spectrum of presentations. Some present dramatically with focal neurological deficits and an altered level of consciousness, but these are not the patients we are at risk of missing in the ED. It is the neurologically intact patient with a normal GCS that poses the greatest diagnostic challenge. Among these patients, the clinical features that are more common, on a percentage basis, in patients with subarachnoid hemorrhage than without include:
- Thunderclap headache – 83%6,7
- Worst headache of life – 95%6–8
- Neck stiffness – 74%6–8
- Vomiting – 62%6–8
- Loss of consciousness – 13%6–8
The problem is that subarachnoid hemorrhage is rare. Although the majority of patients with SAH present with one or more of the above features, it turns out that in isolation, they aren’t overly predictive. If we look at all comers presenting to the ED with acute headache peaking within one hour, any one of these features is more likely to be present in patients without SAH than with!
The poor predictive value of ‘thunderclap’ seems particularly surprising. In part, this may relate to how we are asking about this in our history. Neurosurgeons place a lot of weight on how a patient presents, but when they describe the classic aneurysmal subarachnoid hemorrhage presentation, they don’t refer to the official definition of thunderclap headache which allows for a 60-second crescendo. Instead, they refer to a 10/10 headache that is instantly peaking. Snap of the fingers. Nothing, and then baseball bat to the head. Using descriptive terms like this should result in more predictive value.
Combining features can also be helpful. As turns out, combining the classic instantly peaking headache described above with one of; neck stiffness, vomiting, or a loss of consciousness increases your pre-test probability to approximately 25%. It is in these patients where I truly worry about the diagnosis of subarachnoid hemorrhage. Alternatively, patients that present with a moderate to severe headache peaking within one hour with a normal neurological exam and none of the above features have a pre-test probability in the neighbourhood of 7%.6–9 This is the minimum criteria I use to consider the diagnosis. This one-hour time frame is quite conservative and works to ensure we are not missing any potential subarachnoid cases.
Clinical Feature Take-Aways
- Subarachnoid hemorrhage is an uncommon but deadly diagnosis.
- The average emergency clinician will see less than 50 cases over the course of their career
- When to consider the diagnosis:
- Moderate to severe headache
- Peak within 1 hour
- No clear alternative cause
- When to worry:
- Instantly peaking 10/10 headache
- Any of vomiting, neck stiffness, loss of consciousness
Non-Contrast CT: Within 6 hours
Up until 2011, available research suggested that non-contrast CT (NCCT) was effective at identifying subarachnoid blood and that it did so with greater sensitivity the closer we were to the time of symptom onset.10,11 The problem was that available literature was largely retrospective and often used outdated technology which made it challenging to draw any firm conclusions.
This was until Perry et al., published their landmark study “Sensitivity of CT performed within six hours of onset of headache for diagnosis of subarachnoid hemorrhage”. This large, prospective, multi-center study demonstrated a 100% sensitivity for NCCT at identifying subarachnoid blood when performed within 6 hours of headache onset and interpreted by an experienced radiologist. For many, this changed the way subarachnoid hemorrhage was worked-up in the ED. Before this study, CT followed by a lumbar puncture was the consensus gold standard. After, many clinicians were stopping after NCCT if they could get the scan performed within this 6-hour window.
Unfortunately, this sensitivity of 100% turned out to be a little too good to be true. In 2020, Perry et al. published a follow-up study in Stroke that looked to validate the 6-hour CT rule. This time around, they found NCCT performed within 6 hours of headache onset to be only 95.5% sensitive.12 Of the 111 patients with subarachnoid hemorrhage in their study, 5 were missed by NCCT.

Of these, two were false positives caused by a traumatic LP and the aneurysms identified on CTA were ultimately deemed to be incidental by neurosurgery. One was a radiology miss where a blinded re-interpretation of the scan did show a subarachnoid hemorrhage. The final two reflected true misses. The first was a small non-aneurysmal bleed caused by a rare dural venous fistula. The second was in a patient with sickle cell anemia and a hemoglobin of 63 g/L. This raises an important caveat that is explored in the radiology literature that blood appears isodense when the hemoglobin concentration falls below 100 g/L13. It is important to keep this concept in mind when evaluating patients with a worrisome headache and anemia. Further diagnostic testing may be warranted.
If we remove only the false positive cases, and leave the radiology miss as this represents human error that will surely continue to exist, we are left with a sensitivity of NCCT performed within 6 hours of headache onset of 97.3%. By combining Dr. Perry’s two studies, we get a value closer to 98%. Our ultimate conclusion, however, remains the same. Although NCCT is very good a detecting subarachnoid blood within 6 hours of headache onset, it is not perfect. There are important caveats to consider such as anemia and there will always be human error.
This leaves us with the important question of whether NCCT performed within 6 hours is good enough to be a stand-alone rule-out test. To answer this, it is important to remember just how rare the diagnosis of subarachnoid hemorrhage is. On average, an emergency physician will see 1.2 SAH cases per year. This translates to 1 miss every 40 years using NCCT alone! For many, this miss rate is acceptable. If this isn’t the case for you, it can be further lowered by adding either an LP or a CTA in those select patients who present with that classic, high-risk presentation discussed earlier.
Non-Contrast CT: Beyond 6 hours
As it turns out, there are only two studies that directly look at the sensitivity of NCCT beyond 6 hours. The first was the original study by Perry et al., discussed earlier, that prospectively enrolled a total of 2179 acute headache patients presenting beyond 6 hours. They found a relatively weak sensitivity of 85.7%.9 The second study, published by Backes et al. in 2012, was a retrospective analysis of 113 patients with SAH that presented after 6 hours from headache onset. Here, they found a slightly improved sensitivity of 92.3% when all scans were interpreted by a neuroradiologist.14 In 2016, Carpenter et al. performed a meta-analysis that pooled the data of these two studies and found an overall sensitivity of 89% for NCCT performed beyond 6 hours.3 Not overly impressive.
Fortunately, the story doesn’t quite end here. If we look closer at the study by Perry et al., we notice a much lower prevalence of SAH in the beyond 6-hour cohort:

This seems to check-out intuitively. Patients who feel like they took a baseball bat to the head will likely present to the hospital quickly. In general, patients that present later are going to have a lower pre-test probability of disease. Obviously, the clinical history still prevails. If a patient provides that classic story and says that they were suffering miserably at home for 6 hours before arriving to the ED, you should still be highly concerned. But this is likely the exception.
Using a sensitivity value of 89%, a negative likelihood ratio of 0.11, and a pre-test probability of 5.4% for the beyond 6-hour group, we get a post-test probability for subarachnoid hemorrhage of 0.6% following a negative NCCT. This means that if you used NCCT alone beyond 6 hours, you would miss one subarachnoid hemorrhage out of every 166 patients that you work-up.
Moreover, this is almost certainly conservative. A major issue with this data is that both studies created a dichotomy at a 6-hour time point. Beyond 6 hours, all patients were lumped into one massive heterogeneous category. In the study by Perry et al. the cases of SAH that were missed on NCCT ranged from 8 hours to 8 days after the onset of headache. In the study by Backes et al., ALL cases of SAH that were missed on NCCT came beyond 24 hours! It is likely a safe assumption that the sensitivity of NCCT performed at 8 hours is markedly different than the sensitivity at 8 days, yet in the data presented above, both are combined into one catch-all group.
Based on this, the sensitivity of NCCT performed within the first 24 hours is probably well into the 90% range. What we are still missing, however, is the inflection point. Where does the sensitivity of NCCT really start to decline? It is important to remember that the 6-hour time window was selected at random by a group of 5 emergency physicians and one neurosurgeon. There is nothing scientific about this number. Based on current data, the inflection point for NCCT probably lies somewhere much closer to the 24-hour mark, but to know this definitively, further prospective study is needed. Until then, it is important to know that if your suspicion is low, NCCT is likely sufficient well beyond 6 hours. This said, in the context of a worrisome presentation, further workup is likely warranted.
NCCT Take-Aways:
- The sensitivity of NCCT performed within 6 hours of headache onset approaches but does not equal 100%.
- Human error exists
- Anemia makes subarachnoid blood more difficult to identify
- The inflection point for when the sensitivity of NCCT begins to decline remains unknown
- 6 hours was selected based on no solid evidence
- The sensitivity of NCCT up to 24 hours from headache onset is likely well above 90%
Lumbar Puncture
The pathognomonic finding for SAH on lumbar puncture is xanthochromia. We know that the formation of xanthochromia takes time as RBCs are gradually broken down into oxyhemoglobin and then bilirubin which both contribute to the discolouration of CSF.15 An often-quoted time for how long this process takes is 12 hours, with some proponents recommending that LPs be delayed until at least this long to ensure an accurate result. As it turns out, however, this recommendation is not based on sound evidence. A 1989 study by Vermeulen et al. found that between 12 hours and 3 days from the onset of headache, 100% of patients with confirmed SAH on CT had xanthochromia present.16 The problem was that they used spectrophotometry for xanthochromia detection which is not available in a vast majority of North American hospitals, TOH included.17 Here, visual inspection is the preferred method, and not surprisingly, this demonstrates lower sensitivity.15
What about RBCs? Surprisingly, despite lumbar puncture being a mainstay in the work-up of subarachnoid hemorrhage for decades, there is no widely accepted definition for what makes an elevated RBC count. A major barrier to assessing for RBCs in the CSF when working up SAH is the traumatic tap, which occurs in anywhere from 10-30% of lumbar punctures depending on the cut-off used.18 A commonly held misconception is that you can safely label an LP result as a ‘traumatic tap’ if the RBC count declines by 25% from tube 1 to tube 4. The flaw in this logic is that it is possible to have a traumatic tap AND a subarachnoid hemorrhage. Because of this, observing a decline is not enough. There needs to be a firm cut-off.
Perry et al. attempted to answer this question with their 2015 study published in BMJ, “Differentiation between traumatic tap and aneurysmal subarachnoid hemorrhage”. Of the 1739 patients enrolled, 36% had an abnormal lumbar puncture based on an RBC cut-off of 1 x 106/L.18 98% of these were false positives.18 In total, only 15 patients had a true aneurysmal subarachnoid hemorrhage.18 Looking at these 15 patients, only 7 were positive for xanthochromia (47%) even though 13/15 LPs were performed after 12 hours!18 Using a receiver operating curve, they identified the optimal RBC cut-point to be 2000×106.18 When combining this RBC threshold with positive xanthochromia, meaning that an LP is positive if either are present, they found a sensitivity of 100% (CI 75-100%).18 It is important to note, however, that this ‘rule’ was established based on a total of 15 positive cases. This results in confidence intervals that extend as low as 75% for sensitivity. Obviously, this is a worst-case scenario, but a validation study to confirm these results would be useful. For now, if we keep in mind how few patients will have a SAH following a negative NCCT, this rule will conservatively lower our post-LP odds of a missed SAH to below 1 in 1000.
Although effective at ruling out SAH, the LP has its share of limitations. Its main complication is the dural puncture headache which can occur in up to 35% of patients depending on needle type and gauge.19,20Although most often self-limited, these headaches can be debilitating and take up to a week to recover from.20,21 Epidural hematoma formation and meningitis are much less frequent complications. The high rate of traumatic taps leads to a significant number of patients requiring further testing beyond just LP, often with a CTA. Another drawback is that an LP adds between 2 and 4 hours to a patient’s overall ED length of stay.8,22Finally, patients dislike this procedure and frequently refuse it despite strong physician recommendation.23
Lumbar Puncture Take-Aways
- There is no good evidence to support the practice of waiting 12 hours before performing a lumbar puncture.
- The threshold of 2000 x 106/L RBCs was established on only 15 positive cases. Prospective validation would be useful.
- Applying a Bayesian approach, an LP that is negative for xanthochromia and has fewer than 2000 x 106/L RBCs should conservatively decrease the post-test probability of SAH to less than 1 in 1000 following a negative NCCT.
- Drawbacks to LP include dural puncture headaches, traumatic taps leading to further diagnostic testing, increased ED length of stay, and patient discomfort.
CT Angiogram
With improved multi-row CT scanners and superior software for image reformatting, the less invasive CT angiogram (CTA) has largely supplanted invasive vascular studies as the modality of choice for investigating cerebral aneurysms as well as making subsequent treatment decisions. All patients who have a positive NCCT scan or positive LP will now go on to getting a CTA to look for aneurysms, and less commonly, other vascular malformations. More recently, some practitioners have moved to using CTA instead of the LP for the initial work-up of subarachnoid hemorrhage. But is this approach sufficient?
To dive into this, we need to better understand the etiology of non-traumatic subarachnoid hemorrhage which breaks down as follows:
- Aneurysmal – 80-85%2,24
- No bleeding source identified – 10-15%2
- Perimesencephalic SAH(50%)25
- Non-perimesencephalic SAH(45%)25
- Radiology negative SAH (5%)25
- Other – 5%2
- Vascular malformations
- Tumours
- Vasculitis
- Dissection
Of all the causes listed above, the only source of bleeding that cannot be identified by a NCCT + CTA approach is the radiology negative subarachnoid hemorrhages. These are the hemorrhages with a negative initial CT scan, a positive LP, and then a negative CTA. They make up less than 1% of all subarachnoid hemorrhages.25 When identified, these patients are typically admitted to hospital but with the sole purpose of identifying a vascular cause that was possibly missed on initial CTA. Rarely, aneurysms can clot off in the setting of an acute rupture and be missed on initial imaging. Radiology negative SAH patients undergo further vascular investigation, often with a formal 6 vessel angiogram. The yield on this additional testing is very low.25 In patients where no vascular cause is ultimately found, functional outcomes are excellent with rates of re-bleeding, hydrocephalus and vasospasm approaching zero.25 As such, if CTA is effective at identifying vascular causes of SAH then the NCCT + CTA approach is a reasonable one.
In 2011, Westerlaan et al. published a meta-analysis in Radiology that looked at the sensitivity and specificity of CTA at identifying cerebral aneurysms in patients with confirmed subarachnoid hemorrhage. This meta-analysis included a total of 50 studies between 1997 and 2008 with strong methodology (mean QUADAS score of 11). The majority of studies used 4-detector row scanners. As a reference, the stroke scanner used at the Ottawa Civic Hospital has 320-detector rows whereas the remainder of TOH scanners have 64-detector rows. Overall, CTA had a sensitivity of 98% (CI 97-99%) and a specificity of 100% (CI 97-100%) at identifying cerebral aneurysms!24 This results in a negative likelihood ratio of 0.02.24 Combining this with a negative NCCT would give us a post-test probability of a missed aneurysmal subarachnoid hemorrhage that approaches zero.
Performance of CTA at identifying cerebral aneurysms24
| Sensitivity | Specificity | Negative LR |
| 98% | 100% | 0.02 |
In reality, NCCT + CTA does not perform quite as well as the above numbers would suggest. The miss rate is not zero. One reason for this is that up until this point we have had to make one major assumption; that NCCT and CTA are distinctly independent tests. Is this the case? In favour of this assumption is that NCCT and CTA look for different disease entities; blood for NCCT and aneurysms for CTA. The factors that would diminish their respective sensitivities are largely different. For NCCT, these factors include time from headache onset, volume of blood, and hemoglobin concentration.19 Conversely, for CTA these factors include aneurysm size and location as well as contrast injection timing and technique.19 Against the independent test assumption is the fact that both are interpreted by the same physician. Theoretically, if you have a less experienced physician interpreting the images, the likelihood of having a miss on both CT and CTA could increase.
Weighing all of the factors above, it would seem reasonable to assume that CT and CTA are mostly, but not completely, independent. There likely exists a small degree of interdependence between the two. To account for this, McCormack et al. performed a sensitivity analysis using an interdependence value of 50% to explore a worst-case type scenario. Despite this conservatively high degree of overlap, they found that NCCT + CTA ruled out aneurysmal subarachnoid hemorrhage with a probability of 99.43% if both tests were negative!19 In this analysis, they also underestimated the sensitivity of NCCT using a value of only 91%. Accounting for these conservative estimates, our actual miss rate for the NCCT + CTA approach is probably less than 1 in 1000, similar to that of LP when using the higher RBC cut-off discussed previously.
In addition to effectively working up subarachnoid hemorrhage, the incremental benefit of CTA is that it provides additional diagnostic yield. When a patient presents with a thunderclap headache, our differential diagnosis needs to extend beyond just SAH and should include:
1. Reversible cerebral vasoconstriction syndrome (RCVS)
2. Cervical artery dissection
3. Cerebral venous sinus thrombosis (CVST)
First coined in 2007, RCVS is a unifying diagnosis for multiple vasoconstriction syndromes11 and is caused by reversible segmental vasospasm in the circle of Willis. A thunderclap headache is the primary complaint in 94-100% of patients and is often the only presenting symptom.26 Although many diagnoses can present with a thunderclap headache, RCVS along with SAH are among the few that present primarily with a thunderclap. As more data continues to emerge, RCVS may be the most common secondary cause of thunderclap headache.27,28 Patients have a mean of four thunderclap attacks over a one-month period at which time symptoms typically subside.27 By definition, vasospasm is no longer detected on vascular imaging after 3 months. Risk factors include the peripartum period and vasoactive substance use27 including sympathomimetics, marijuana, and serotonergic agents. Common triggers, similar to SAH, include exercise, valsalva, and sex. Although most patients with RCVS have a good prognosis, a small but important subset experience significant negative sequelae. Cortical subarachnoid hemorrhage occurs in 30-34%, seizures in 1-17%, cerebral infarction in 6-39%, and PRES 9-38%.26 Mortality approaches 1%29 with 5-10% of patients experiencing permanent disability.26 Importantly, these sequelae are potentially avoidable. Because we currently have no way of predicting which patients will experience poor outcomes, the Neurology service at TOH is currently admitting all RCVS patients where they are started on calcium channel blockers to help minimize cerebral vasoconstriction. RCVS is a diagnosis that needs to be considered, especially in the setting of recurrent thunderclap episodes or when any of the above risk factors are present, and CTA is the test of choice.
Taking a deep dive into cervical artery dissection and CVST is beyond the scope of this discussion. In brief, both can present with thunderclap headache anywhere from 10-22% of the time11 and should be on our ED differential. Cervical artery dissection is often associated with neck pain as well as additional neurological signs and symptoms depending on whether we are referring to the vertebral or carotid artery. Importantly, however, the headache may precede these neurological symptoms by hours to days.11 To diagnose, CTA is the test of choice. Similar to dissection, CVST presents with a headache as the most common clinical feature which can be antecedent to signs of increased ICP or focal deficits by the order of several days.30 With oral contraceptives and the peripartum period being primary risk factors, CVST frequently affects young women.30 Clinical outcomes can be devastating. Surprisingly, multi-detector CTA demonstrates exceptionally high sensitivity and specificity at imaging the venous sinuses, similar to that of MR venography in one recent study.31
The main drawbacks to CTA include increased radiation exposure, contrast allergy, and the identification of incidental aneurysms. Radiation exposure and incidental aneurysm detection are probably the most important. The addition of CTA to NCCT increases a patient’s total radiation exposure to 5 mSV (2.5 mSV each).32 For reference, this is half the dose of a CT abdomen and pelvis. In a 20-year-old female, this translates to 1 additional malignancy for every 2000 CTAs performed,33 a risk that decreases progressively with age. Concerning incidental aneurysm detection, it is estimated that 2.5% of the general population lives their lives with a cerebral aneurysm.19,34 With an annual rupture risk of 0.5-1.4%34, the majority of these will never lead to any morbidity. This said, the psychological burden of being aware of an incidental intracranial aneurysm can be significant. Although there exist evidence-based scoring systems to help better quantify individual patient risk, patient preference plays a significant role in treatment decisions. If intervention is sought, the outcomes are not necessarily benign with morbidity associated with coiling, which includes peri-procedural stroke and iatrogenic aneurysm rupture, being quoted at 2%.34
CTA Take-Aways
- When combined with NCCT, CTA rules out clinically important subarachnoid hemorrhage with similar efficacy to lumbar puncture.
- CTA has the added benefit of diagnosing other important causes of thunderclap headache that should be on our radar including cervical artery dissection, CVST and RCVS.
- Drawbacks to CTA include increased radiation exposure, contrast allergy, and the psychological burden of identifying asymptomatic aneurysms.
My Approach to Subarachnoid Hemorrhage in the ED
Under 6 hours
- NCCT will be sufficient to rule out subarachnoid hemorrhage in a majority of patients. I will often stop here if subarachnoid hemorrhage is my only concern.
- If I believe there is a reasonable possibility of an alternative diagnosis, I will add a CTA even in this below 6-hour group. It is important to think beyond just SAH.
- Neck pain, characteristic clinical features, and disease-specific risk factors are the main elements that raise my suspicion of an alternative etiology.
- In select high-risk patients with a perfect story for SAH or in patients with severe anemia, I will proceed with further testing to rule out SAH recognizing that the sensitivity of NCCT is not 100%.
- LP will be my test of choice if SAH is my only concern.
- CTA will be my test of choice if I am considering alternative diagnoses or I believe the LP will be technically challenging.
- Patient preference should factor into decision-making.
Beyond 6 hours
- Although the sensitivity of NCCT decreases with time, it likely remains in the mid 90% range until up to 24 hours. When my clinical suspicion is low, and subarachnoid hemorrhage is my only concern, stopping here is likely reasonable.
- In the remainder of patients, further diagnostic testing is indicated.
- LP will be my test of choice if SAH is my only concern.
- CTA will be my test of choice if I am considering alternative diagnoses or I believe the LP will be technically challenging.
- Patient preference should factor into decision-making.
References
1. Hop J, Rinkel G, Algra A, van Gijn J. Case-Fatality Rates and Functional Outcome After Subarachnoid Hemorrhage: A Systematic Review. Stroke. 1997;28(3):660-664.
2. Suarez JI. Diagnosis and management of subarachnoid hemorrhage. Contin Lifelong Learn Neurol. 2015;21(5):1263-1287. doi:10.1212/CON.0000000000000217
3. Carpenter CR, Hussain AM, Ward MJ, et al. Spontaneous Subarachnoid Hemorrhage: A Systematic Review and Meta-analysis Describing the Diagnostic Accuracy of History, Physical Examination, Imaging, and Lumbar Puncture With an Exploration of Test Thresholds. Acad Emerg Med. 2016;23(9):963-1003. doi:10.1111/acem.12984
4. Edlow JA. Diagnosis of subarachnoid hemorrhage in the emergency department. Emerg Med Clin North Am. 2003;21(1):73-87. doi:10.1016/S0733-8627(02)00081-0
5. Perry JJ, Spacek A, Forbes M, et al. Is the Combination of Negative Computed Tomography Result and Negative Lumbar Puncture Result Sufficient to Rule Out Subarachnoid Hemorrhage? Ann Emerg Med. 2008;51(6):707-713. doi:10.1016/j.annemergmed.2007.10.025
6. Perry JJ, Stiell IG, Sivilotti MLA, et al. Clinical decision rules to rule out subarachnoid hemorrhage for acute headache. JAMA – J Am Med Assoc. 2013;310(12):1248-1255. doi:10.1001/jama.2013.278018
7. Perry JJ, Sivilotti MLA, Sutherland J, et al. Validation of the Ottawa Subarachnoid Hemorrhage Rule in patients with acute headache. CMAJ. 2017;189(45):E1379-E1385. doi:10.1503/cmaj.170072
8. Perry JJ, Stiell IG, Sivilotti MLA, et al. High risk clinical characteristics for subarachnoid haemorrhage in patients with acute headache: Prospective cohort study. BMJ. 2010;341(7781):1035. doi:10.1136/bmj.c5204
9. Perry JJ, Stiell IG, Sivilotti MLA, et al. Sensitivity of computed tomography performed within six hours of onset of headache for diagnosis of subarachnoid haemorrhage: Prospective cohort study. BMJ. 2011;343(7817):1-10. doi:10.1136/bmj.d4277
10. Muehlschlegel S. Subarachnoid hemorrhage. Continuum (N Y). 2018;24(6):1623-1657. doi:10.1017/CBO9781139030854.100
11. Matharu MS, Schwedt TJ, Dodick DW. Thunderclap headache: An approach to a neurologic emergency. Curr Neurol Neurosci Rep. 2007;7(2):101-109. doi:10.1007/s11910-007-0004-8
12. Perry JJ, Sivilotti MLA, Émond M, et al. Prospective Implementation of the Ottawa Subarachnoid Hemorrhage Rule and 6-Hour Computed Tomography Rule. Stroke. 2020;51(2):424-430. doi:10.1161/STROKEAHA.119.026969
13. Smith W, Batnitzky S, Rengachary S. Acute isodense subdural hematomas: a problem in anemic patients. Am J Roentgenol. 1981;136(3):543-546.
14. Backes D, Rinkel GJE, Kemperman H, Linn FHH, Vergouwen MDI. Time-dependent test characteristics of head computed tomography in patients suspected of nontraumatic subarachnoid hemorrhage. Stroke. 2012;43(8):2115-2119. doi:10.1161/STROKEAHA.112.658880
15. Chu K, Hann A, Greenslade J, Williams J, Brown A. Spectrophotometry or visual inspection to most reliably detect xanthochromia in subarachnoid hemorrhage: Systematic review. Ann Emerg Med. 2014;64(3):256-264.e5. doi:10.1016/j.annemergmed.2014.01.023
16. Vermeulen M, Hasan D, Blijenberg BG, Hijdra A, Van Gijn J. Xanthochromia after subarachnoid haemorrhage needs no revisitation. J Neurol Neurosurg Psychiatry. 1989;52(7):826-828. doi:10.1136/jnnp.52.7.826
17. Perry JJ, Sivilotti MLA, Stiell IG, et al. Should spectrophotometry be used to identify xanthochromia in the cerebrospinal fluid of alert patients suspected of having subarachnoid hemorrhage? Stroke. 2006;37(10):2467-2472. doi:10.1161/01.STR.0000240689.15109.47
18. Perry JJ, Alyahya B, Sivilotti MLA, et al. Differentiation between traumatic tap and aneurysmal subarachnoid hemorrhage: Prospective cohort study. BMJ. 2015;350. doi:10.1136/bmj.h568
19. McCormack RF, Hutson A. Can computed tomography angiography of the brain replace lumbar puncture in the evaluation of acute-onset headache after a negative noncontrast cranial computed tomography scan? Acad Emerg Med. 2010;17(4):444-451. doi:10.1111/j.1553-2712.2010.00694.x
20. Nath S, Koziarz A, Badhiwala JH, et al. Atraumatic versus conventional lumbar puncture needles: a systematic review and meta-analysis. Lancet. 2018;391(10126):1197-1204. doi:10.1016/S0140-6736(17)32451-0
21. Roos KL. Lumbar puncture. Semin Neurol. 2003;23(1):105-114.
22. Perry JJ, Stiell I, Wells G, Spacek A. Diagnostic test utilization in the emergency department for alert headache patients with possible subarachnoid hemorrhage. Can J Emerg Med. 2002;4(5):333-337. doi:10.1017/S1481803500007739
23. Dupont SA, Wijdicks EFM, Manno EM, Rabinstein AA. Thunderclap headache and normal computed tomographic results: Value of cerebrospinal fluid analysis. Mayo Clin Proc. 2008;83(12):1326-1331. doi:10.4065/83.12.1326
24. Westerlaan H.E., Dijk van J.M.C., Jansen-van der Weide M.C., et al. Intracranial Aneurysms in Patients with Subarachnoid Hemorrhage : CT Angiography as a Primary Examination Tool for Diagnosis — Systematic Review and Purpose : Methods : Results : Radiology. 2011;258(1):134-145.
25. Mohan M, Islim AI, Rasul FT, et al. Subarachnoid haemorrhage with negative initial neurovascular imaging: a systematic review and meta-analysis. Acta Neurochir (Wien). 2019;161(10):2013-2026. doi:10.1007/s00701-019-04025-w
26. Miller TR, Shivashankar R, Mossa-Basha M, Gandhi D. Reversible cerebral vasoconstriction syndrome, part 1: Epidemiology, pathogenesis, and clinical course. Am J Neuroradiol. 2015;36(8):1392-1399. doi:10.3174/ajnr.A4214
27. Ducros A. Reversible cerebral vasoconstriction syndrome. Lancet Neurol. 2012;11(10):906-917. doi:10.1016/S1474-4422(12)70135-7
28. Chen SP, Fuh JL, Wang SJ. Reversible cerebral vasoconstriction syndrome: An under-recognized clinical emergency. Ther Adv Neurol Disord. 2010;3(3):161-171. doi:10.1177/1756285610361795
29. Bernard KRL, Rivera M. Reversible cerebral vasoconstriction syndrome. J Emerg Med. 2015;49(1):26-31. doi:10.1016/j.jemermed.2015.01.012
30. Agrawal K, Burger K, Rothrock JF. Cerebral Sinus Thrombosis. Headache. 2016;56(8):1380-1389. doi:10.1111/head.12873
31. Linn J, Ertl-Wagner B, Seelos KC, et al. Diagnostic Value of Multidetector-Row CT Angiography in the Evaluation of Thrombosis of the Cerebral Venous Sinuses. Am J Neuroradiol. 2007;28(5).
32. Alons IME, Goudsmit BFJ, Jellema K, van Walderveen MAA, Wermer MJH, Algra A. Yield of Computed Tomography (CT) Angiography in Patients with Acute Headache, Normal Neurological Examination, and Normal Non Contrast CT: A Meta-Analysis. J Stroke Cerebrovasc Dis. 2018;27(4):1077-1084. doi:10.1016/j.jstrokecerebrovasdis.2017.11.016
33. American Society of Radiologic Technologists. X-Ray Risk. https://www.xrayrisk.com/calculator/select_study.php?id=58. Published 2018. Accessed September 9, 2020.
34. Nasr DM, Brown RD. Management of Unruptured Intracranial Aneurysms. Curr Cardiol Rep. 2016;18(9):1-6. doi:10.1007/s11886-016-0763-4