Pneumothorax is a relatively rare emergency department presentation with a number of controversies existing regarding its management. Over the last decade, the standard of care has shifted from invasive strategies to more conservation management approaches. What factors should you consider in managing the next pneumothorax you see on shift, and what do we really know from the existing evidence?
In part one of our two-part grand round series on pneumothorax, Dr. Simon Wells covers the varying definitions, epidemiology, diagnostic criteria, and imaging findings of pneumothorax including on point of care ultrasound (POCUS).
Part two of the series will focus on pneumothorax treatment and management options, as well as a summary of the 2020 NEJM publication ‘Conservative versus Interventional Treatment for Spontaneous Pneumothorax’.
Definitions
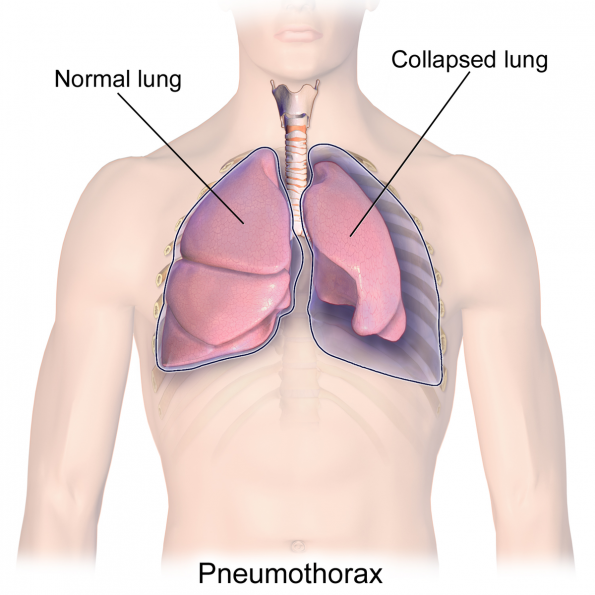
Pneumothorax refers to air in the pleural cavity. It was first described in medical literature in 1803 as a complication of tuberculosis infection.1 It was not until 1932 that the first case of “pneumothorax simple”, or pneumothorax in someone with no apparent underlying lung disease, was reported.2 This type of pneumothorax would come to be known as a primary spontaneous pneumothorax (PSP).
Secondary spontaneous pneumothorax (SSP) occurs in patients with clinically apparent underlying lung disease. They are both important to differentiate from traumatic pneumothoraces, which can be either iatrogenic or caused by blunt or penetrating traumatic injuries.
PSP vs. SSP
The concept of PSP occurring in patients with “no underlying lung disease” is traditionally taught, but this concept is in-fact outdated.3
- Advances in chest imaging demonstrate underlying emphysema-like changes, most notably blebs and bullae, in most cases of PSP.3,4
- Pathologic assessments of underlying lung also show less visible changes, such as disrupted areas of mesothelial cells, underlying inflammation, and pores 10-20 microns in diameter. 5
- These findings suggest that the distinction between PSP and SSP is an artificial construct.6
- Future study is needed to determine if patients are well served by this traditional differentiation and whether it should influence management.
Epidemiology
- PSP incidence:
- 7-28/100 000 /year (males), 1.2-9.8/100 000 /year (females) 7–9
- PSP is much more common in males.
- Male/Female ratio ~ 5:1
- PSP typically occurs in younger adults, as opposed to SSP typically occur later in life.
- The incidence of SSP is similar to that of PSP.
- SSP are still more frequent in men, but at a lower ratio of 3:1.
Smoking and PSP
- The majority of patients with PSP are smokers.
- A Swedish population study found that there was a direct dose dependent relationship between the number of cigarettes smoked and the relative risk of pneumothorax, as indicated in the following table.7
| Relative Risk of SP vs Non-Smokers | Men | Women |
| Light smokers (1-12/day) | 7 | 4 |
| Medium smokers (13-22/day) | 21 | 11 |
| Heavy smokers (>22/day) | 80 | 42 |
| Overall | 22 | 9 |
- Smokers have higher recurrence rates.10
- Smoking cessation is the only evidence-based modifiable risk factor for PSP recurrence.11
- One systematic review, including 29 pooled studies looking at recurrence risk of pneumothorax found smoking cessation was associated with a four-fold decrease in risk (OR 0.26, 95% CI 0.10–0.63).12
- All patients with pneumothorax should be counselled on the importance of smoking cessation and referred to appropriate support services.
Cannabis smoking and pneumothorax
- Cannabis smoking has been shown to be associated with emphysema-like changes in the lungs.13
- Regular cannabis smoking increases the risk of PSP.14
- Underlying cause assumed to be related to both parenchymal damage from smoke, and the longer breath-holds manoeuvres often used in cannabis smoking.15
- All patients with PSP should be questioned about cannabis use and counselled to stop.
Other risk factors for pneumothorax
- Increased height (in men)
- Thought to be due to an increased pleural pressure gradient from the lung base to the apex in taller individuals, causing increased apical distending pressures that potentially lead to the formation of subpleural blebs.16
- Low BMI & caloric restriction
- There is conflicting data on these potential risk factors.11
- Up to 5-10% of individuals have a family history.17
- Some are eventually found to have hereditary syndromes such as Birt-Hogg-Dube syndrome, Marfan syndrome, Ehler-Danlos syndrome, Cutis laxa.
- These should then be considered cases of SSP, not PSP.
- Sudden atmospheric pressure changes, diving.18–20
Causes of SSP
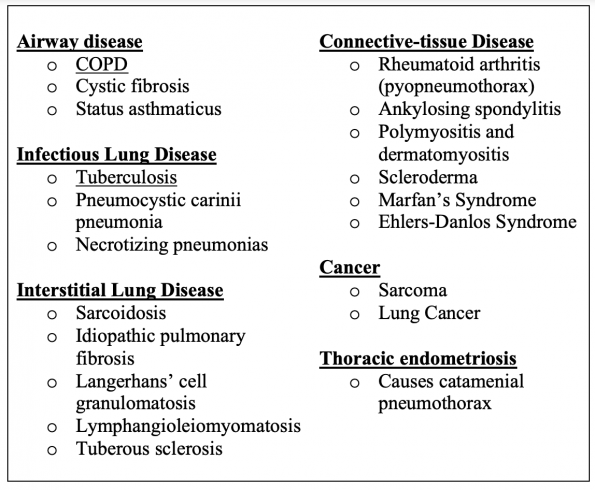
- Nearly every lung disease can be complicated by SSP.
- The most commonly associated disease is COPD.21
- In endemic areas, tuberculosis (TB) is a common cause.
Catamenial pneumothorax
- Occurs in women around the time of menstruation.
- Due to pleural endometriosis implant or defect in the diaphragm.
- Treatment: ovulatory suppression or surgery.22
- ~5% of all pneumothoraces in women.
- Tend to be recurrent; multiple typically occur before a diagnosis is made.23
Traumatic pneumothorax
- The current rate of traumatic pneumothorax seems to be greater than the rate of spontaneous pneumothorax. 24
- Can occur iatrogenically from procedures, including transbronchial biopsy, central venous catheterization, thoracentesis and mechanical ventilation.
- Penetrating wounds can lead to open pneumothorax.
- With blunt traumatic mechanisms, rib fractures or sudden increases in alveolar pressure from chest compression with a closed glottis can cause pneumothorax.
Tension pneumothorax
- A one-way valve mechanism allows air to enter the pleural space during inspiration but not exit fully during expiration
- Air in the pleural space builds up enough pressure to interfere with venous return, leading to hypotension, tachycardia, low cardiac output, and cardiac or respiratory failure
- Tension pneumothorax is very rare in both PSP and SSP.
- More common in traumatic pneumothorax and in patients on mechanical ventilation.
- Needs immediate treatment with finger thoracostomy or needle decompression, followed by large bore chest tube placement.
Clinical Presentation
- Common symptoms: sudden chest tightness, pain and SOB.
- Onset does not usually occur during exertion.
- SSP has been traditionally associated with more morbidity and mortality, as well as more severe symptoms. However, the evidence for this is largely anecdotal.
- In mechanically ventilated patients, pneumothorax should manifest as a sudden decrease in pulmonary compliance.
- Volume control ventilation = elevation in both peak and plateau pressures.
- Pressure control ventilation = decreased tidal volume.
Diagnosis and Imaging
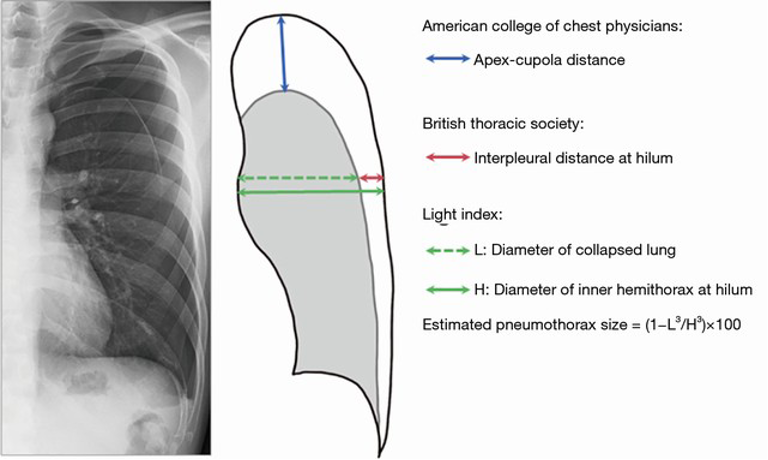
- The first issue in the imaging and management of pneumothorax is that there is no international consensus as to what quantifies a large pnemothorax.
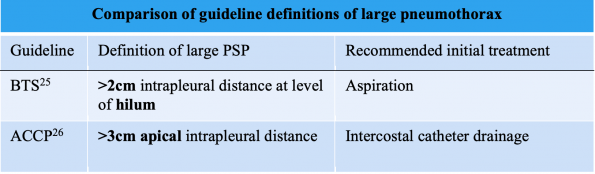
- Available guidelines from the American College of Chest Physicians (ACCP) and British Thoracic Society (BTS) are below.25,26 However, they agree less than 50% of the time in clinical settings based available data.27
- This is an important issue, as guidelines currently recommend different management options for large vs small pneumothoraces.
The Light Index28
-
- % Pneumothorax = 100 – [(lung diameter3/hemithorax diameter3) x 100]
- One study found strong correlation with volume of air removed29
- Poor correlation with CT volumetrics found in another study30
- Overall seems to be inaccurate, especially seems to underestimate size for moderate/large pneumothoraces.31
The Collins Method32
-
- % Collins = 4.2 + 4.7(A+B+C)
- It is obtained by getting measurements in centimetres at three locations and inputting them into the formula above.
- Seems to be better at estimating the size of larger pneumothoraces.27
Overall however, neither of these methods is routinely used in clinical practice. Additionally, at present, it is reasonable to choose either the BTS or ACCP definition of large pneumothorax.
X-ray criteria for diagnosing pneumothorax
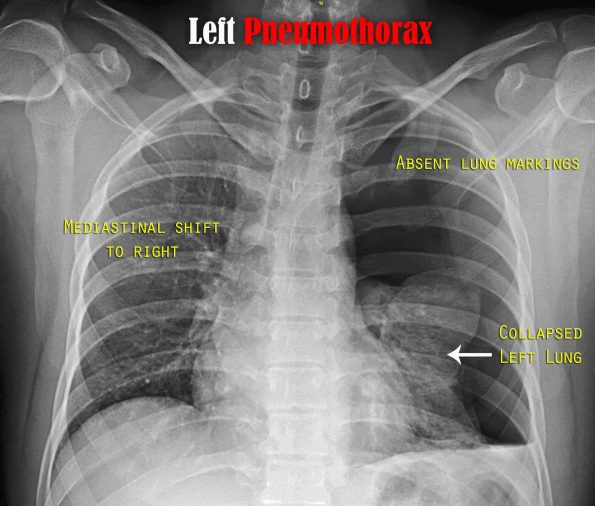
- Displacement of the pleural line
- Absence of lung markings peripheral to the pleural line
Inspiratory vs expiratory views
- Thomsen et al. found that pneumothoraces during inspiration demonstrate a greater width than during expiration, contrary to widely held but unstudied assumptions.33
- Inspiratory and expiratory views have been shown to perform similarly.34,35
- Guidelines recommend inspiratory views alone; no need for expiratory views!
- Measurement standards are based on views obtained during inspiration.
Signs of pneumothorax on supine x-rays
- Deep sulcus sign – a deep and sometimes lateral costophrenic sulcus
- Double diaphragm sign – visualization of the anterior costophrenic sulcus
- Increase in sharpness of the cardiac borders, adjacent mediastinal margin, or diaphragm
- Visualization of inferior edge of collapsed lung
- Increased lucency on affected side
- Depression of the ipsilateral hemidiaphragm
- X-ray mimics: skin fold, bullae, gastric herniation
CXR vs PoCUS
- Most studies comparing the two modalities are in trauma and use supine x-rays.36
- Meta-analysis, 8 studies, traumatic and iatrogenic pneumothorax:37
- PoCUS: 9% sensitive (95% CI, 86.5-93.9), 98.2% specific (95% CI, 97.0-99.0)
- CXR (almost all supine): 50.2% sensitive (95% CI, 43.5-57.0), 99.4% specific (95% CI, 98.3-99.8)
- PoCUS significantly outperforms supine CXR for diagnosing pneumothorax in trauma.
- PoCUS is faster and less disruptive to care/resuscitation.38
- E-FAST with bilateral lung assessments should be the standard of care in trauma assessments to add assessment for pneumothorax.
- Cannot make accurate size estimates based on PoCUS as of yet.
PoCUS findings of pneumothorax

- Absence of Pleural sliding
- Absence of Comet tails or B-Lines
- Presence of a lung point
- Absence of lung pulse
- Should be assessed using an 8-zone technique
Check out this excellent video on POCUS assessment for pneumothorax from EM cases for more information:
Lung sliding
- Lack of sliding at the pleural line on POCUS is indicative of pneumothorax, however multiple other conditions can cause loss of sliding.
- This means we need to combine the loss of lung sliding with other signs and not limit our assessment purely to the pleural line.
Comet tails and B-lines
- Comet tails and B lines are normal reverberation artefacts that occur in the presence of lung parenchyma in contact with the pleural line.
- In patients with no lung sliding, the presence of B lines or comet tails can essentially rule out pneumothorax.
- If they are absent and there is no lung sliding, this combination improves the specificity and PPV of lung ultrasound for pneumothorax detection.
Lung point
- The lung point is where lung starts to be in contact with pleural line – this is defined as a transition between an area of active lung sliding and an area without any sliding.
- The presence of a lung point, with the absence of lung sliding and comet tails, improves the specificity of POCUS to 100%.
- Location of lung point can help to roughly estimate size – if found more laterally or posterior then pneumothorax is larger.
Lung Pulse
- This is the slight movements of the pleural line corresponding with heart beats (In the absence of normal lung sliding) – looks like a slight shimmering at the pleural line.
- Indicates normal visceral-parietal pleura interface – not seen in pneumothorax.
- Can be seen in conditions like atelectasis and mainstem intubation.
Potential pneumothorax mimics on POCUS
- Pleural scarring: Previous pleurodesis, radiation changes, infectious/inflammatory pleural disease
- Lung volume loss: atelectasis, mucus plug, right mainstem intubation
- COPD (bullous emphysema)
- Pulmonary fibrosis
- Phrenic palsy
Conclusions
In this post, we explored the varying definitions of pneumothorax, including whether a PSP truly occurs in the absence of any underlying lung disease. We discussed risk factors, including cannabis smoking, and the different sub-typing of pneumothorax described in the literature. Finally, we reviewed imaging findings of pneumothorax and POCUS clinical pearls to help sharpen your bedside skill-set.
Our next post, part two of the series, will focus on treatment and management options, as well as a summary of the 2020 NEJM publication entitled ‘Conservative versus Interventional Treatment for Spontaneous Pneumothorax’. Stay tuned!
References
- Laennec R-T-H (1781-1826) A du texte. De l’auscultation Médiate Ou Traité Du Diagnostic Des Maladies Des Poumons et Du Coeur, Fondé Principalement Sur Ce Nouveau Moyen d’exploration. Tome 2 / Par R. T. H. Laënnec,…; 1819. Accessed November 2, 2020. https://gallica.bnf.fr/ark:/12148/bpt6k98759b
- Kjaergaad H. Spontaneous pneumothorax in the apparently healthy. Acta Med Scand. 1932;43:1-159.
- Mitlehner W, Friedrich M, Dissmann W. Value of Computer Tomography in the Detection of Bullae and Blebs in Patients with Primary Spontaneous Pneumothorax. RES. 1992;59(4):221-227. doi:10.1159/000196062
- Bense L, Lewander R, Eklund G, Hedenstierna G, Wiman LG. Nonsmoking, non-alpha 1-antitrypsin deficiency-induced emphysema in nonsmokers with healed spontaneous pneumothorax, identified by computed tomography of the lungs. Chest. 1993;103(2):433-438. doi:10.1378/chest.103.2.433
- Noppen M, Dekeukeleire T, Hanon S, et al. Fluorescein-enhanced autofluorescence thoracoscopy in patients with primary spontaneous pneumothorax and normal subjects. Am J Respir Crit Care Med. 2006;174(1):26-30. doi:10.1164/rccm.200602-259OC
- Bintcliffe OJ, Hallifax RJ, Edey A, et al. Spontaneous pneumothorax: time to rethink management? Lancet Respir Med. 2015;3(7):578-588. doi:10.1016/S2213-2600(15)00220-9
- Bense L, Eklund G, Wiman LG. Smoking and the increased risk of contracting spontaneous pneumothorax. Chest. 1987;92(6):1009-1012. doi:10.1378/chest.92.6.1009
- Melton LJ, Hepper NG, Offord KP. Incidence of spontaneous pneumothorax in Olmsted County, Minnesota: 1950 to 1974. Am Rev Respir Dis. 1979;120(6):1379-1382. doi:10.1164/arrd.1979.120.6.1379
- Gupta D, Hansell A, Nichols T, Duong T, Ayres JG, Strachan D. Epidemiology of pneumothorax in England. Thorax. 2000;55(8):666-671. doi:10.1136/thorax.55.8.666
- Cheng Y-L, Huang T-W, Lin C-K, et al. The impact of smoking in primary spontaneous pneumothorax. J Thorac Cardiovasc Surg. 2009;138(1):192-195. doi:10.1016/j.jtcvs.2008.12.019
- Sadikot RT, Greene T, Meadows K, Arnold AG. Recurrence of primary spontaneous pneumothorax. Thorax. 1997;52(9):805-809. doi:10.1136/thx.52.9.805
- Walker SP, Bibby AC, Halford P, Stadon L, White P, Maskell NA. Recurrence rates in primary spontaneous pneumothorax: a systematic review and meta-analysis. European Respiratory Journal. 2018;52(3). doi:10.1183/13993003.00864-2018
- Johnson MK, Smith RP, Morrison D, Laszlo G, White RJ. Large lung bullae in marijuana smokers. Thorax. 2000;55(4):340-342. doi:10.1136/thorax.55.4.340
- Hedevang Olesen W, Katballe N, Sindby JE, et al. Cannabis increased the risk of primary spontaneous pneumothorax in tobacco smokers: a case-control study. Eur J Cardiothorac Surg. 2017;52(4):679-685. doi:10.1093/ejcts/ezx160
- Gill A. Bong lung: regular smokers of cannabis show relatively distinctive histologic changes that predispose to pneumothorax. Am J Surg Pathol. 2005;29(7):980-982. doi:10.1097/01.pas.0000157998.68800.cb
- West JB. Distribution of mechanical stress in the lung, a possible factor in localisation of pulmonary disease. Lancet. 1971;1(7704):839-841. doi:10.1016/s0140-6736(71)91501-7
- Johannesma PC, Reinhard R, Kon Y, et al. Prevalence of Birt–Hogg–Dubé syndrome in patients with apparently primary spontaneous pneumothorax. European Respiratory Journal. 2015;45(4):1191-1194. doi:10.1183/09031936.00196914
- Alifano M, Forti Parri SN, Bonfanti B, et al. Atmospheric pressure influences the risk of pneumothorax: beware of the storm! Chest. 2007;131(6):1877-1882. doi:10.1378/chest.06-2206
- Bertolaccini L, Alemanno L, Rocco G, Cassardo C. Air pollution, weather variations and primary spontaneous pneumothorax. J Thorac Dis. 2010;2(1):9-15.
- Park JH, Lee SH, Yun SJ, et al. Air pollutants and atmospheric pressure increased risk of ED visit for spontaneous pneumothorax. Am J Emerg Med. 2018;36(12):2249-2253. doi:10.1016/j.ajem.2018.04.020
- Hallifax RJ, Goldacre R, Landray MJ, Rahman NM, Goldacre MJ. Trends in the Incidence and Recurrence of Inpatient-Treated Spontaneous Pneumothorax, 1968-2016. JAMA. 2018;320(14):1471-1480. doi:10.1001/jama.2018.14299
- Korom S, Canyurt H, Missbach A, et al. Catamenial pneumothorax revisited: clinical approach and systematic review of the literature. J Thorac Cardiovasc Surg. 2004;128(4):502-508. doi:10.1016/j.jtcvs.2004.04.039
- Alifano M, Jablonski C, Kadiri H, et al. Catamenial and noncatamenial, endometriosis-related or nonendometriosis-related pneumothorax referred for surgery. Am J Respir Crit Care Med. 2007;176(10):1048-1053. doi:10.1164/rccm.200704-587OC
- Weissberg D, Refaely Y. Pneumothorax: experience with 1,199 patients. Chest. 2000;117(5):1279-1285. doi:10.1378/chest.117.5.1279
- MacDuff A, Arnold A, Harvey J. Management of spontaneous pneumothorax: British Thoracic Society pleural disease guideline 2010. Thorax. 2010;65(Suppl 2):ii18-ii31. doi:10.1136/thx.2010.136986
- Baumann MH, Strange C, Heffner JE, et al. Management of spontaneous pneumothorax: an American College of Chest Physicians Delphi consensus statement. Chest. 2001;119(2):590-602. doi:10.1378/chest.119.2.590
- Kelly A-M, Druda D. Comparison of size classification of primary spontaneous pneumothorax by three international guidelines: A case for international consensus? Respiratory Medicine. 2008;102(12):1830-1832. doi:10.1016/j.rmed.2008.07.026
- Pleural Diseases, 6th Edition – Richard W. Light – Gale – 978-1469831398. Accessed November 6, 2020. https://www.cengage.com/search/productOverview.do;jsessionid=78B7AF482E37A1BF84928050DCED3023?N=197+4294888968&Ntk=P_EPI&Ntt=1224060617117256879319453285491368018289&Ntx=mode%2Bmatchallpartial
- Noppen M, Alexander P, Driesen P, Slabbynck H, Verstraete A, Vlaamse Werkgroep voor Medische Thoracoscopie en Interventionele Bronchoscopie. Quantification of the size of primary spontaneous pneumothorax: accuracy of the Light index. Respiration; International Review of Thoracic Diseases. 2001;68(4):396-399. doi:10.1159/000050533
- Hoi K, Turchin B, Kelly A-M. How accurate is the Light index for estimating pneumothorax size? Australas Radiol. 2007;51(2):196-198. doi:10.1111/j.1440-1673.2007.01705.x
- Akamine T, Kometani T, Hashinokuchi A, Akamine S, Shikada Y, Wataya H. Interpleural distance predicts persistent air leak after initial primary spontaneous pneumothorax. Journal of Thoracic Disease. 2020;12(5):2228-2235-2235. doi:10.21037/jtd.2020.04.48
- Collins CD, Lopez A, Mathie A, Wood V, Jackson JE, Roddie ME. Quantification of pneumothorax size on chest radiographs using interpleural distances: regression analysis based on volume measurements from helical CT. AJR American journal of roentgenology. 1995;165(5):1127-1130. doi:10.2214/ajr.165.5.7572489
- Thomsen L, Natho O, Feigen U, Schulz U, Kivelitz D. Value of digital radiography in expiration in detection of pneumothorax. RoFo: Fortschritte Auf Dem Gebiete Der Rontgenstrahlen Und Der Nuklearmedizin. 2014;186(3):267-273. doi:10.1055/s-0033-1350566
- Seow A, Kazerooni EA, Pernicano PG, Neary M. Comparison of upright inspiratory and expiratory chest radiographs for detecting pneumothoraces. AJR Am J Roentgenol. 1996;166(2):313-316. doi:10.2214/ajr.166.2.8553937
- Schramel FM, Golding RP, Haakman CD, Sutedja TG, de Jong KA, Postmus PE. Expiratory chest radiographs do not improve visibility of small apical pneumothoraces by enhanced contrast. The European Respiratory Journal. 1996;9(3):406-409. doi:10.1183/09031936.96.09030406
- Chan KK, Joo DA, McRae AD, et al. Chest ultrasonography versus supine chest radiography for diagnosis of pneumothorax in trauma patients in the emergency department. Cochrane Database of Systematic Reviews. 2020;(7). doi:10.1002/14651858.CD013031.pub2
- Alrajhi K, Woo MY, Vaillancourt C. Test Characteristics of Ultrasonography for the Detection of Pneumothorax: A Systematic Review and Meta-analysis. Chest. 2012;141(3):703-708. doi:10.1378/chest.11-0131
- Wilkerson RG, Stone MB. Sensitivity of bedside ultrasound and supine anteroposterior chest radiographs for the identification of pneumothorax after blunt trauma. Academic Emergency Medicine: Official Journal of the Society for Academic Emergency Medicine. 2010;17(1):11-17. doi:10.1111/j.1553-2712.2009.00628.x