Sotrovimab
Sotrovimab is a monoclonal antibody that neutralizes sarbecoviruses (respiratory viruses detected by PCR on oral/nasal swabs or from bronchial/lung/respiratory tract specimens). This antibody was first isolated from a patient with SARS-CoV-1 (the virus responsible for the 2002-2004 SARS outbreak). Sotrovimab also works against SARS-CoV-2, which is responsible for the current COVID-19 pandemic.
This antibody binds to a conserved epitope on the spike protein of SARS-CoV-2. An epitope (aka antigenic determinant) is the portion of an antigen that is recognized by the immune system to stimulate an immune response; it is the part of an antigen that binds to antibodies. Sotrovimab inhibits an undefined step that occurs after virus attachment but prior to fusion of the viral and cell membranes.
There are advantages to Sotrovimab. First, it targets a highly conserved part of the SARS-CoV-2 spike protein – in other words, it targets an area of the virus that has not been mutating. This means that sotrovimab should work against variants. Second, there are two amino acid substitions at the Fc portion of the antibody (termed the LS modification) which extends its half-life (median 49 days).
Rationale
The investigators assessed the efficacy and safety of sotrovimab among high-risk outpatients with mild to moderate COVID-19 infection. This was an interim analysis of a phase 3 multicenter double-blind randomized controlled trial: COVID-19 Monoclonal Antibody Efficacy Trial – Intent to Care Early (COMET-ICE).
PICOS
Patients:
- Age ≥18 years with confirmed mild to moderate COVID-19 infection
- Symptom onset ≤5 days
- SpO2 ≥94% on RA
- ≥1 risk factor for progression of COVID-19:
- Age ≥55 years (irrespective of comorbidities)
- Diabetes (on medication)
- Obesity (BMI > 30 kg/m2)
- CKD (eGFR < 60 mL/min/1.73 m2)
- CHF (NYHA class II–IV)
- COPD or moderate-to-severe asthma
- Exclusion criteria:
- Severe COVID-19
- Dyspnea at rest or respiratory distress requiring supplemental O2
- Severely immunocompromised
- Cancer patients receiving active therapy
- Solid organ transplant or allogeneic stem cell transplant in past 3 months
- Systemic corticosteroids equivalent to ≥0.5 mg/kg of prednisone within past 6 weeks
- Investigator felt patient was likely to die in the next 7 days
- Hypersensitivity or anaphylaxis to a monoclonal antibody
- Severe COVID-19
Intervention:
- Sotrovimab 500 mg IV over 1 hour
Comparator:
- Saline placebo
Outcome:
- Primary: percentage of patients with hospitalization for >24 hours or death from any cause up to 29 days after randomization
- Secondary:
- ED visit, hospitalization or death
- Disease progression warranting use of supplemental O2
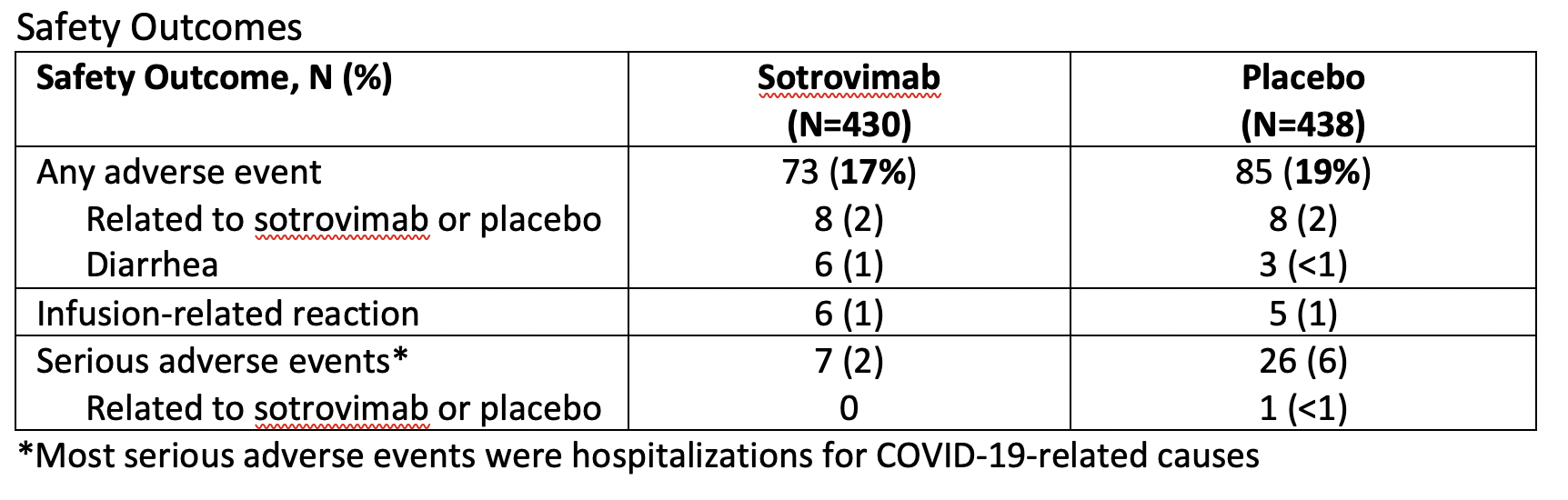
- Safety outcomes:
- Adverse events
- Serious adverse events (included hospitalization for any cause)
- Adverse events of special interest (infusion-related reactions)
Setting:
- 37 sites in 4 countries (USA, Canada, Brazil and Spain)
Main Results
The main results for efficacy and safety are summarized in the tables below.
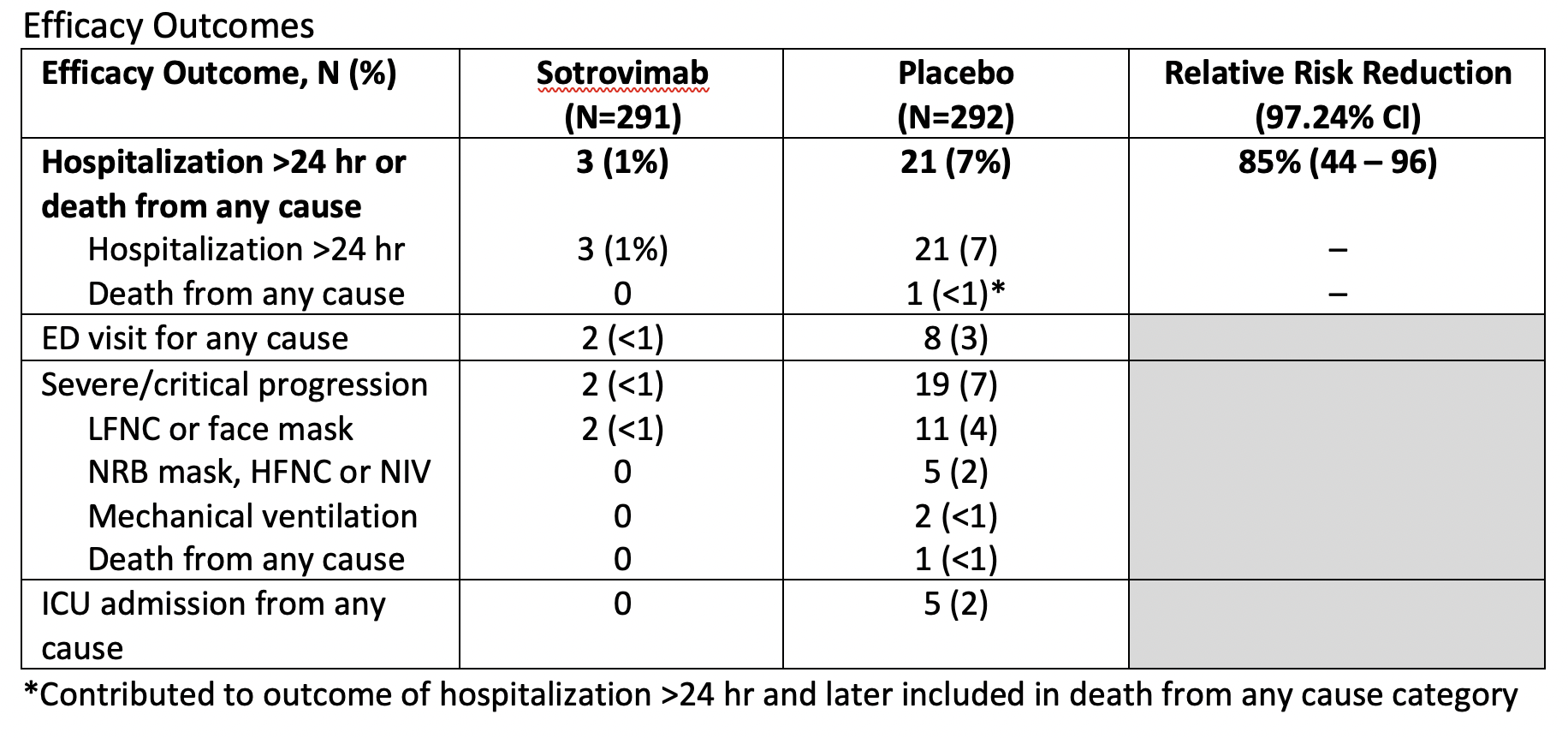
The relative risk reduction is an estimate of the proportion of baseline risk (i.e., hospitalization >24 hr or death from any cause) that is removed by the therapy (i.e., sotrovimab). If we focus on the primary outcome, we can say there is approximately an 85% lower risk for hospitalization >24 hr or death from any cause when treating mild to moderate COVID-19 infection with sotrovimab. More accurately, we can be approximately 97% confident that the risk reduction lies somewhere between 44 and 96%.
Final thoughts (what does this mean for my practice?)
Sotrovimab is a monoclonal antibody therapy that is effective in reducing the risk of progression of COVID-19 in outpatients with mild to moderate disease that have certain risk factors (see inclusion criteria). It is not for patients with severe disease requiring hospitalization. Importantly, it appears the main benefit is through a reduction in the need for hospitalization.
I would follow local criteria for appropriate patient selection (this may slightly vary at different sites). Additionally, I would counsel patients regarding the safety of this medication and would quote a 1% chance of diarrhea. Furthermore, I would explain that no serious adverse events were found to be directly linked to sotrovimab itself.
It’s important to recognize that this is a fairly small trial that is an interim analysis. I look forward to reading further published data about this treatment.
Disclaimer: my wording has been intentionally careful – the above are recommendations for my own practice. Social media, podcasts, and so on are fantastic resources. However, if you come across something that may genuinely change your practice, then I strongly encourage you to read the article for yourself.



Trackbacks/Pingbacks