Atrial fibrillation (AF) is encountered with significant frequency in the Emergency Department (ED). In fact, atrial fibrillation is the single most common arrhythmia seen in the ED. As per the 2020 CCS review, AF resulted in 8815 same-day procedures, 76 964 ED visits, 25 892 acute care admissions in Canada in a given year.1 The CAEP 20212 guidelines were created in consensus with academic, community ED physicians and cardiologists from multiple sites across Canada. This post will review the literature leading to the new best practice guidelines and highlight major updates since the last management guidelines were posted in 2018 while identifying sources of practice variation.
This blog post will review the following and apply guidelines to common cases seen below:
- New safe rhythm control timelines
- Rate control and pitfalls
- Stroke prevention
Case 1
Case 1: Ms. Rapide is a 63 yo female with hx of HTN, CAD with previous PCI (2014) and peripheral vascular disease. Her medications include amlodipine and ASA. She presents to your ED with a clear onset of palpitations 15 hours ago.
Case 2
Case 2: Mr. Beaucoeur is a 50-year-old male with hx of EToH use disorder and hypothyroidism presenting to your ED with a first-time presentation of paroxysmal atrial fibrillation within 48 hours.
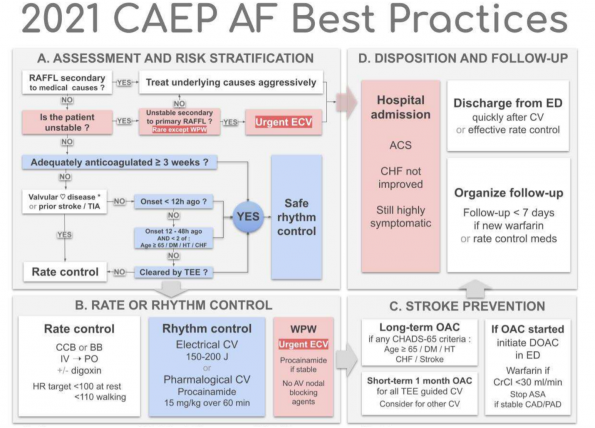
CAEP 2021 Atrial Fibrillation/Flutter Guidelines
Management of patients with atrial fibrillation / flutter can be broken down into four steps:
- Is this a primary or secondary arrhythmia?
- Is the patient stable or unstable?
- Has the patient been adequately anticoagulated?
- Select appropriate management
STEP 1 – PRIMARY VS SECONDARY ARRHYTHMIA
Initial Assessment
Acute, symptomatic atrial fibrillation / flutter is a complex condition that may be caused by an isolated electrophysiological disorder (primary arrhythmia) or is a symptom of an underlying disease process (secondary arrhythmia, note that this is less common). Some etiologies leading to secondary atrial fibrillation include: sepsis, hemorrhage, pulmonary embolism, heart failure, ACS, thyrotoxicosis, cardiac and non-cardiac surgery, acute infections, catecholamine excess and mitral valve disease.1 The treatment pathway for atrial fibrillation will vary depending on the presence of an underlying cause which can be identified on history and physical examination.
Complete a thorough history and physical exam to assess for the following underlying secondary condition1:
- Decompensated Heart Failure: increasing weight, orthopnea, dyspnea, pitting edema, adventitious lung sounds suggestive of pulmonary edema, jugular venous distension. Global decreased LV function and B lines on POCUS
- Hypovolemia: recent illness or decreased PO intake, dry mucous membranes, decrease urine output
- Pulmonary Embolism: VTE history or risk factors, dyspnea, clear lung sounds, enlarged RV on POCUS, septal flattening, McConnell’s sign
- ACS: Cardiac risk factors, Chest pain, diaphoresis, N/V
- Sepsis: Clinical Assessment
The following signs and symptoms may suggest that AF / AFL may more likely be secondary to underlying medical cause and therefore may require additional workup12:
- No palpitations reported
- Lack of sudden onset of symptoms
- Dyspnea present
- Persistent retrosternal chest pain
- No prior history of ED cardioversions
- Permanent atrial fibrillation on OACs
- HR < 150
- Fever
Work-up / Investigations
Upon presentation to the ED, the workup required for patients with AF / AFL can be variable. CCS (2020) suggests that all initial evaluation of patient’s newly diagnosed with AF AFL include: ECG, TTE and basic laboratory investigations – but what about initial presentation to ED? Investigations in these patients is guided based on the potential for identifying a reversible cause and baseline to guide anticoagulation as required where it is suggested to complete CBC, electrolytes, extended electrolytes, creatinine / BUN, coagulation studies and TSH. In the setting where there is suspicion for cardiogenic shock, LFTs, VBG and lactate is recommended.

It is worth noting variation in guideline recommendations, as the CAEP 2021 best practice checklist does not suggest ordering coagulation studies, TSH or TEE.
There is much discussion to be had regarding troponin levels. In general, patients with AF / AFL can be expected to have an elevated troponin due to increased cardiac demand from the arrhythmia. An unpublished review of charts at the University Health Network in Toronto showed that 86% had their troponin drawn, 14% had a positive test and 5% were treated with ACS.4 Of note, most of those treated for ACS had hypotension, signs of heart failure or ECG changes after rhythm or rate control.5 Briefly, having a slightly positive troponin in an asymptomatic patient with AF/AFL can be problematic as it may lead to further delays to discharge and, potentially, unwarranted treatment of ACS. The bottom line is that routine testing for troponin is not recommended in these patients UNLESS there is a clinical suspicion for underlying ACS.
Management of Secondary AF / AFL
In 2014, Scheuermeyer et al. published a retrospective study in ACEP comparing 416 patients with “complex” AF / AFL. What they found was that active management (cardioversion or rate control) produced major adverse events (13.1%, 95% CI 7.4%-20.3%) including intubation, CPR, death, stroke, and new hypotension requiring vasopressors in patients with secondary AF.9 This study has led to the primordiality of focusing on resuscitation and treatment of the acute illness rather than control of rate or rhythm. Ultimately, treatment for secondary AF / AFL is guided by the underlying cause that was identified given that their arrhythmia may be transient as a compensatory mechanism for their underlying condition. Cardioversion is therefore avoided in these patients due to the potential for harm. Furthermore, aggressive rate control is not recommended as this may compromise the patient’s compensatory abilities for increasing cardiac output.
STEP 2 – PATIENT STABILITY
The next step is to identify your unstable patients. Of note, instability due to primary atrial fibrillation and atrial flutter is uncommon except for AF with RV pre-excitation (WPW). Any patients identified to have any of the following signs of cardiovascular instability require urgent electrical cardioversion12:
-
- Hypotension (sBP <90mmHg) and Signs of shock (altered mental status); or
- Pulmonary edema (significant dyspnea, crackles and hypoxia; or
- Cardiac ischemia (severe chest pain, ST depression (<2mm) despite therapy.
It is important to note that ST depression in atrial fibrillation is common and often due to rate-related ischemia in the setting of CAD; however, this should not be considered unstable unless the patient has anginal chest pain or if the ST segments do not resolve once the patient is no longer in atrial fibrillation.
STEP 3 – SAFETY TO CARDIOVERT PRIMARY ARRHYTHMIA
Once a patient has been identified as more stable after assessing criteria from Step 2, an assessment should be performed to determine the safety of cardioverting a primary arrhythmia. The AFFIRM trial (2002) fathered the dogma of rate vs rhythm control having no significant difference in terms of morbidity and quality of life outcomes however, those who were unlikely to tolerate being in AF were excluded from the study.18 Furthermore, the RAFF 3 showed us that this method reduces length of stay in ED and utilizes less hospital resources.14 For this reason, unless there is a compelling reason to do so otherwise, rhythm control is the preferred method of treatment when safety is determined. Ultimately, when deciding to proceed with rate vs rhythm control, an assessment of your patient’s eligibility is necessary in preventing any risk of a serious embolic event (SEE). Generally, thromboembolic risk peri-cardioversion can be conceptualized by 2 mechanisms:
-
- Generation of thrombi during persistent AF with embolization after restoration of organized atrial contraction
- Atrial stunning which is responsible for thrombi post-cardioversion regardless of modality (pharmacological, electrical, spontaneous).2
Given the high risk of stroke in certain populations, the new CAEP 2021 Guidelines provide recommendations to guide safe cardioversion recommendations as outlined below2:
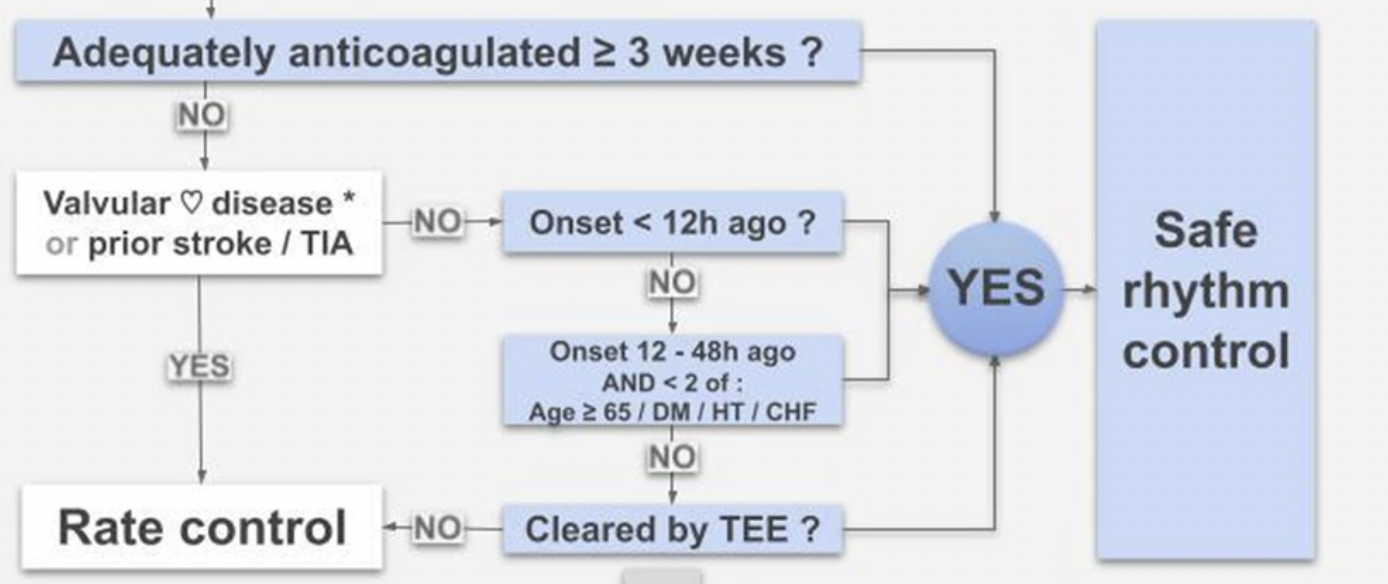
Finally, determination of adequate anticoagulation for 3 weeks is based on MD judgement but also confirmation of compliance on history for DOAC and INR > 2 on current or most recent test for Warfarin.12
STEP 4A – MANAGEMENT – RHYTHM CONTROL
Pharmacologic vs Electrical Cardioversion
For either method of cardioversion, chemical or electrical. The best practice checklist suggest that either is acceptable, if one does not work, simply try the other. They also suggest no rate control pre-treatment to rhythm control as this is ineffective and delays treatment.12 When considering chemical or electrical cardioversion, it is recommended to discuss the options with the patients and review previous visits outlining the most successful method if the patient has a history of paroxysmal AF. The RAFF2 trial suggests that a trial of chemical cardioversion (with procainamide) prior to electrical cardioversion worked in 52% of patients and can avoid a resource intensive procedural sedation. However, generally the drug-shock group vs the shock-only group had similar effective rates (96% vs 92%) (absolute difference 4%, 95% CI 0-9, p=0.07).10
To perform chemical cardioversion, Class I or III antiarrhythmics are commonly employed:Pharmacologic Cardioversion
To perform electrical cardioversion:Electrical Cardioversion
STEP 4B – MANAGEMENT – RATE CONTROL
Rate control can be performed in the ED if cardioversion is deemed unsafe as per the above guidelines. Rate control may also be considered based on patient or physician preference in the stable and minimally symptomatic patient. Given the potential harm associated with rate control agents in patients with new or existing heart failure, one may enhance the sensitivity of their therapeutic approach by looking at a recent echocardiogram report, or performing bedside PoCUS to assess LV function. In patients with grossly reduced LV function, rate control should be approached cautiously.
When considering rate control, your target resting heart rate is <100 bpm and <110 bpm with walking.12 A study by Wong et al. sought to describe ED rate control management and outcomes in patients with AF who were not successfully managed by rhythm control or not attempted rhythm control. Of note, they found that only 59.0% of patients achieved a rate control with resting HR < 100 at ED discharge and 47% of patients were managed with unsuitable choice of agents (2.6%), route of administration (27.4%), underdoses (6.0%), overdosed (3.4%) or administered beyond recommended time frame (7.7%).17
Generally, calcium channel blockers (CCB) and beta blockers (BB) are first line therapies for rate control in the ED. Should the patient be on a home dose of CCB or BB prior to their ED visit, it is best to start with the medication from the same drug class initially. Ultimately, there is no evidence of superiority of either class and should there be challenges with achieving rate control with one class, the other class is a good substitute. An exception to this management algorithm is if the patient has HF where CCB and BB are contraindicated due to risk of myocardial suppression and subsequent cardiogenic shock. In such circumstances, the rate control medication of choice is digoxin.
Select the below tabs for dosing and indications of each medication.
Calcium channel blocker
- Indications:
- First line therapy for rate control in the ED
- Contraindications:
- Acute Heart Failure
- LV dysfunction on recent ECHO12
- Acute Management Dose:
- Diltiazem 0.5mg/kg IV over 10 mins q 15-30 mins (or q15-20 mins at 0.35mg/kg) up to 3 doses
- Start Diltiazem 25-50mg PO within 30 mins of effective IV rate control
- Discharge dose:
- Diltiazem 30-60mg QID or 120-240mg PO daily of extended release
Beta blocker
- Indications:
- First line therapy for rate control in the ED
- Contraindication:
- Acute Heart Failure
- Acute Management Dose:
- Metoprolol 2.5-5mg IV over 2 mins q15-30 min up to 3 doses
- Then start Metoprolol 25-50mg PO within 30 mins of effective IV rate control
- Discharge dose:
- Metoprolol 25-50mg PO BID 12
Digoxin
- Indications:
- First line therapy for patients who are hypotensive or with Acute Heart Failure
- Second line if unsuccessful in achieving rate control with first line therapies
- Acute Management Dose:
- Digoxin 0.25-0.5mg loading dose then 0.25mg IV q 4-6 hr to max of 1.5mg over 24 hrs
- Renal impairment adjusted – start with 0.25mg IV loading dose
- Disposition:
- Due to slow titrate of this medication, a consultation to cardiology referral is warranted for admission.




Excellent article. Edoxaban is spelled incorrectly and There has been an update to the Edoxaban PM. 30 mg crcl is 15-50 ml/min.