As emergency physicians, we frequently see patient with metabolic syndromes and obesity. Globally, a significant rise in obesity has been observed over the last 30 years, including a dramatic rise in Canada28,32. From 1985 to 2016, obesity increased by more than 300% in Canada29, and currently 1.2 million Canadians have severe obesity23. It is estimated that 1000 bariatric surgeries were performed in 2017, and these numbers are rising. Although the advancement of modern laparoscopic surgical procedures has decreased the rates of post-operative complications, hospitalizations and death, these risks are still present for patients undergoing bariatric surgery.

Types of Bariatric Surgery
Generally speaking, bariatric surgeries can be divided into 2 main categories:
- Gastric restriction
- Malabsorption
Gastric restriction:
- Concept: creation of a smaller gastric pouch that induces early satiety and decreases food intake
- Example: Sleeve Gastrectomy
- Patient factors: Results dependent on patients’ compliance with dietary restrictions.
Malabsorption:
- Procedure: bypassing segments of the small bowel reduces absorption
- Example: Roux-en-Y gastric bypass.
- Patient factors: Less dependent on patients’ dietary compliance compared to restrictive strategies18.
Deeper Dive
Sleeve Gastrectomy
The sleeve gastrectomy is the most commonly performed procedure in Canada and represents 58% of all bariatric surgeries in North America2.The intervention consists of a partial gastrectomy where most of the lateral stomach is removed. The lesser curvature is usually stapled creating a long tubular stomach, a fraction of its original size. It promotes weight loss by reducing meal volume and appetite. From a surgical perspective, this procedure has gained significant popularity due to its simplicity and lower complication rate5.
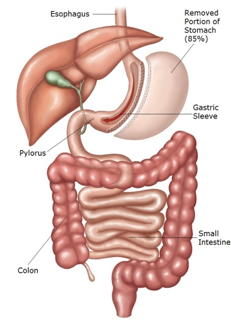 (Picture from: https://weightlosssurgery.ca/about-our-surgeries/comparison-chart-procedures/laparoscopic-vertical-sleeve-gastrectomy/)
(Picture from: https://weightlosssurgery.ca/about-our-surgeries/comparison-chart-procedures/laparoscopic-vertical-sleeve-gastrectomy/)
Roux-en-Y Gastric Bypass
The Roux-en-Y gastric bypass used to be considered the gold standard for bariatric surgery but has since been surpassed by the gastric sleeve. Although it achieves the most effective weight loss, it is associated with more complications7. This procedure promotes weight loss through both malabsorption and gastric restriction. The stomach is divided into 2 segments; the smaller proximal segment is anastomosed to the roux limb which consists of the proximal small bowel. This bypasses approximately the first 75-150cm of small bowel. There is also a biliopancreatic limb, which is anastomosed to the roux limb. There is finally the common channel which then consists of the terminal ileum and continues with normal anatomy5.
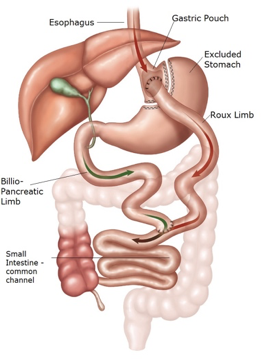 (Picture from: https://weightlosssurgery.ca/about-our-surgeries/comparison-chart-procedures/laparoscopic-roux-en-y-gastric-bypass)
(Picture from: https://weightlosssurgery.ca/about-our-surgeries/comparison-chart-procedures/laparoscopic-roux-en-y-gastric-bypass)
Laparoscopic Adjustable Gastric Banding
Gastric banding has significantly evolved over the last 20 years. It was initially a relatively invasive procedure which involved a laparotomy and a non-adjustable band. The modern gastric band now consists of a laparoscopic procedure with an adjustable gastric band near the GE junction which creates a small stomach pouch. This band is inflatable and is connected to a subcutaneous port which can be accessed to inflate or deflate the band. Although this procedure has the lowest rates of short-term complications, it is associated with the highest rates of long term complications and weight regain. It has largely fallen out of favor over the last few years. Due to these poor outcomes and complications rates, gastric bands are no longer recommended in the Canadian Adult Obesity Clinical Practice Guidelines5.
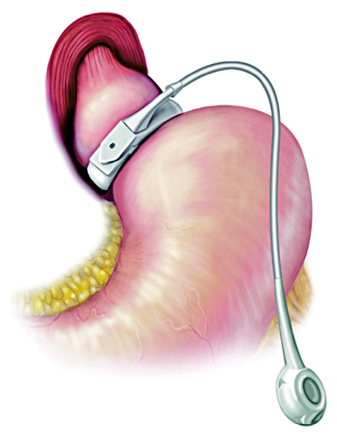 (Picture from UpToDate)
(Picture from UpToDate)
Duodenal Switch
A duodenal switch is a procedure that combines a gastric sleeve and Roux-en-Y gastric bypass. It uses both a restrictive and malabsorptive mechanism to induce weight loss. A wide sleeve gastrectomy is performed. The duodenum is then transected and anastomosed to an alimentary limb. This leaves a very short segment of bowel to allow nutrient absorption. It is quickly gaining popularity due to its sustained weight loss, but is limited by its surgical complexity5. It currently represents approximately 5% of the bariatric surgeries performed in Canada2.
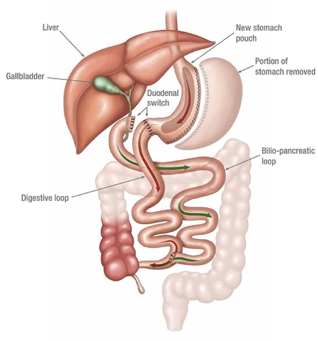
(Picture from: https://weightlosssurgeon.com/surgical-weight-loss-tips/duodenal-switch)
Post-Operative Complications
Sleeve Gastrectomy Complications
1) Bleeding
- The long staple line in sleeve gastrectomy makes it prone to bleeding compared to other bariatric surgeries. Bleeding tends to be extra-luminal as there are no mucosal staples. Most bleeding occurs at the staple line from short gastric vessels. Often, these develop into perigastric hematomas near the staple line, which then have the potential to become infected.
- Signs and symptoms of bleeding include hemodynamic instability such as hypotension, tachycardia, anemia and abdominal pain17. Given that bleeding is mostly extra-luminal, endoscopy has little diagnostic value. A CT abdomen with contrast is the most appropriate test to detect this complication9.
2) Stenoses/Strictures
- After a gastric sleeve, a long tubular stomach is formed which is prone to stenotic lesions.A common risk factor for developing this is oversewing of the staple line intra-operatively, which can lead to swelling and ischemia with subsequent fibrosis10.
- Patients tend to present with obstructive symptoms such as abdominal pain and vomiting.
- In suspected severe cases patients should get imaging in the Emergency Department. The best imaging modality for this is a CT abdomen with PO and IV contrast9. Chronic cases with mild GERD symptoms can be referred for outpatient follow-up with their bariatric surgeon. They can be treated endoscopically or surgically depending on the severity and location of the stenotic lesion.
3) Staple Line Leaks
- Gastric leaks are the most feared complication of gastric sleeves due to its complexity in management and high morbidity. They tend to present within the first month after surgery1.
- Common symptoms include abdominal pain, fevers and tachycardia.
- There are many theories behind the etiology of leaks; high gastric pressures and ischemia likely both play a significant role in leaks.
- Staple line leaks should be investigated with a CT abdomen using PO and IV contrast9.
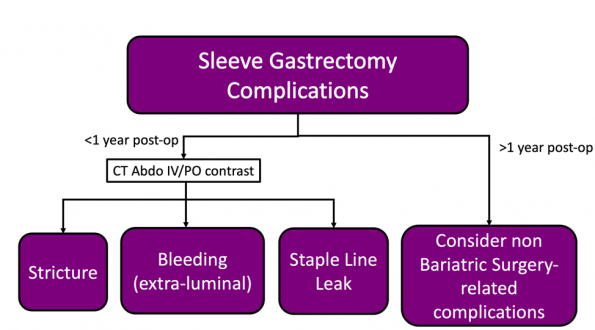
The vast majority of post-operative complications from a sleeve gastrectomy occur in the first year following surgery. These complications can be investigated with a CT abdomen with IV/PO contrast.
Sleeve gastrectomy complications in the late post-operative period (over 1 year post-op) are uncommon. In these cases, consider investigating for non bariatric surgery-related complications.
Gastric Bypass (Roux-en-Y and Duodenal Switch) Complications
1) Anastomotic Leaks
- Anastomotic leaks post gastric bypass present early in the post-operative period, typically within the first 10 days post-op1,3.
- They can present with a variety of nonspecific symptoms such as fever, tachycardia, hypotension, abdominal pain, nausea/vomiting, and shortness of breath.
- We know from some small retrospective studies that tachycardia and respiratory distress are both predictors for anastomotic leaks. A retrospective study published in 2013 found that a heart rate >120bpm and respiratory distress were predictive of leaks with odds ratios of 23.2 and 6.0, respectively13. Patients presenting with tachycardia and respiratory distress in the early post-operative period are also very high risk for pulmonary embolism and appropriate investigations to rule-out venous thromboembolism (VTE) should be strongly considered (see section below on pulmonary embolisms).
- The best imaging test to assess anastomotic leaks is a CT with PO and IV contrast.
2) Anastomotic Stenoses
- Most anastomotic stenoses present in the first few weeks after surgery, but can also present later.The stenotic lesions can occur at either anastomosis (gastro-jejunostomy or jejuno-jejunostomy) and each present with slightly different symptoms.
- Early stenosis of the proximal gastro-jejunostomy anastomosis typically presents in the first few weeks after surgery. Patients have typical obstructive symptoms such as abdominal pain and vomiting. These can often be treated endoscopically21.
- Obstructions at the distal jejuno-jejunostomy anastomosis can have serious consequences. These patients tend to not have any vomiting. The biliopancreatic limb can begin to dilate up to the blind limb of the stomach, potentially leading to perforation and peritonitis. These obstructions are typically surgically managed9.
- Anastomotic stenoses can be investigated using an abdominal CT with PO and IV contrast.
3) Marginal Ulceration
- Marginal ulcers typically form on the proximal anastomosis and occur in 1-7% of patients9. Approximately half of patients present within the first 2 months after surgery but some can occur as late as two years later in up to 15% of patients24.
- Symptoms include abdominal pain, nausea, vomiting and melena. Marginal ulcers have the potential to perforate.
- Typically, these ulcers can be managed with a PPI and patient education on avoiding NSAIDs and smoking. In severe cases some patients may require a revision.
- Abdominal CTs and upper GI series are not sensitive in detecting marginal ulcerations.Endoscopy is the most appropriate test to diagnose marginal ulcerations.
4) Internal Hernias
- Internal hernias occur in approximately 5% of patients. They are typically a late complication following a gastric bypass as it requires significant weight loss. Weight loss can lead to large potential spaces within surgically created mesenteric defects allowing bowel to herniate through12. They tend to present multiple months (typically over a year) post-op.
- Signs and symptoms of internal hernias are obstructive in nature such as abdominal pain, nausea and vomiting.
- The diagnosis of internal hernias can be challenging as they are often intermittent in nature. The bowel can intermittently herniate through mesenteric defects, leading to episodes of pain with potentially negative imaging studies15.
- The overall sensitivity of cbdominal CTs with PO/IV contrast to detect internal hernias varies in the literature. Some studies report sensitivities as low as 56-78%, while others report sensitivities as high as 96-99% 11.
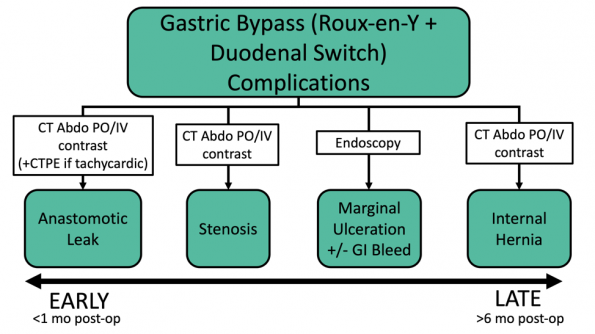
Timing in the post-operative period can greatly help in narrowing the differential diagnosis in patients who have undergone a gastric bypass. The appropriate imaging studies/investigations differ depending on the suspected complication. In the early post-operative period (less than a month after surgery), anastomotic leaks and stenoses are most common. A CT abdomen with PO/IV contrast is the best test to investigate these complications. A chest CTA should be considered in any patient that is tachycardic or has any other signs of a pulmonary embolism. Internal hernias occur multiple months to years following a gastric bypass, after a patient has lost a significant amount of weight. These are best detected using an abdominal CT with PO/IV contrast.
Gastric Band Complications
1) Band Slippage
- Slippage of the band leads to gastric prolapse through the band and dilatation of the gastric pouch.
- Band slippage is a complication that can present both in the early and late post-operative period (days to years post-operatively).
- Acute presentations of band slippage are relatively uncommon but can lead to serious morbidity. These should be considered a severe complication and surgical emergency that could be potentially fatal. The herniated stomach may become strangulated with the potential for perforation and gastric necrosis.
- Symptoms include nausea, vomiting, delayed satiety, gastric reflux symptoms, slowed weight loss, as well as abdominal and chest pain.
- In mild and chronic cases, these patients can be referred to their outpatient bariatric surgeon for a lap band adjustment. In acute and severe cases with potential strangulation, these patients often require urgent deflation and band removal9.
- Imaging studies:
- Abdominal X-rays are a good screening test to detect band slippage
- Phi Angle
- The angle created by the band and the vertebral column on a plain AP abdominal X-ray is referred to as the phi angle. A normal phi angle is typically 5-60 degrees, with the band pointing towards the left shoulder.
- With band slippage, the band is often seen in a horizontal position, often pointing towards the right shoulder.
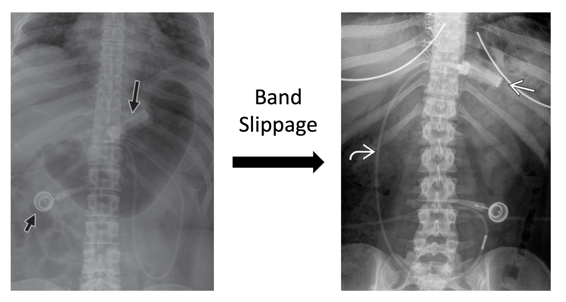
(Left picture: https://www.ajronline.org/doi/pdf/10.2214/AJR.09.3933. Right picture: https://radiologykey.com/imaging-of-bariatric-surgery)
-
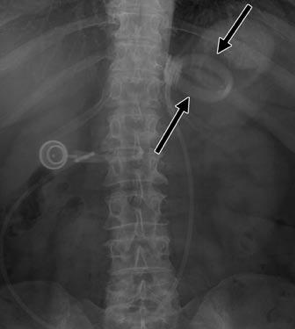
- O Sign
- In a normal AP abdominal X-ray the band should look like a radio-opaque bar a few centimeters below the diaphragm.
- As the gastric pouch herniates through the band during slippage, this can cause the band to rotate along its horizontal axis, taking the characteristic appearance of the letter “O” (Pieroni).
- O Sign
- Band Deflation
- Acute band slippage can be life-threatening and emergent surgical consultation and band deflation is indicated.
- If timely surgical consultation is not available, band deflation can be performed in the Emergency Department. Generally, most ports can be easily palpated and successfully accessed. However, some ports may be difficult to palpate and require fluoroscopy or point-of-care ultrasound to identify their position. Huber point needles should be used to access these ports to avoid a port leak.
- A video reviewing the steps in gastric band deflation can be found here.
2) Band Erosions
- Band erosions occur in approximately 1% of patients8. Similarly to band slippage, this complication can occur both in the early and late post-operative period. Early erosions are rare. Late erosions are however more frequent and occur at a mean of 22 months after surgery27.
- This complication typically occurs due to an overtightened band or irritation from the band. This leads to the band eroding into the stomach wall with possible perforation.
- Signs and symptoms include loss of satiety (most common symptom), abdominal pain, fever, and GI bleeding. They can also be accompanied by a port site infection.
- The gold standard to diagnose band erosion is endoscopy. If access to endoscopy is limited, an abdominal CT with PO/IV contrast is a reasonable alternative study25.
- A surgical consultation for band removal is indicated with band erosions6.

Timing in the post-operative period does not narrow the differential diagnosis for serious gastric band complications. A plain abdominal X-ray is a useful screening tool for band slippage. If the abdominal X-ray does not reveal any signs of band slippage, a CT abdomen with PO/IV contrast or endoscopy (if available) is required to assess for band erosion.
Pulmonary Embolisms
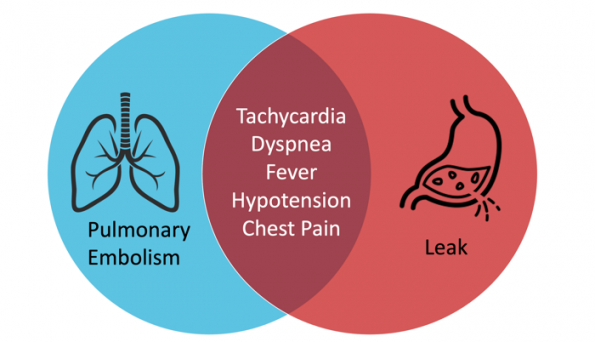
- Bariatric surgery patients are at very high risk of developing a pulmonary embolism (PE) post-operatively. The risk of post-operative VTE is 2-3 times higher in obese patients9. VTE are the second most common cause of death in post-operative bariatric surgery patients with an incidence of 2%. The mortality rate in these patients is 30%20,31. Rates are highest in the immediate post-operative period but this risk can persist for several months after surgery. This diagnosis can be challenging given the significant overlap in symptoms between PE and anastomotic leaks.
- TACHYCARDIA = WARNING SIGN
Tachycardia (especially with a HR>120bpm), in patients within the first few weeks after gastric bypass or sleeve gastrectomy, is a PE or a leak until proven otherwise. These patients should undergo a chest CTA to rule-out PE as well as a CT abdomen with IV/PO contrast to rule out staple line/anastomotic leaks.
Pharmacologic Considerations
NSAIDs
- NSAIDs are a broad class of drugs that are commonly used for analgesia in the emergency department.However, their use is strongly discouraged in postsurgical bariatric patients4. They have the potential to cause gastric damage through 2 main mechanisms. Firstly, they increase gastric acid production, which increases the risk for erosions. Secondly NSAIDs may cause direct irritation to the stomach mucosa33.
- A small retrospective study looking at patients who had undergone Roux-en-Y surgeries, showed that NSAID use was associated with endoscopically detected complications like marginal ulcers. PPIs on the other hand were found to be protective in patients using NSAIDs, with an OR of 0.330.
- Another small study showed that bariatric surgery patients do use NSAIDs at home, despite being instructed to avoid them. Yet these patients seem to have overall low rates of complications related to their NSAID use4.
- Based on current literature, the effect of NSAIDs remains unclear, but there likely is some associated risk.
- Bottom Line: Administering a single dose of NSAIDs to a post-bariatric surgery patient in the Emergency Department is likely safe, when other modes of analgesia have been exhausted. The theoretical risks associated with NSAIDs should be discussed with these patients.
Anticoagulation
- Anticoagulants are frequently prescribed to patients in the Emergency Department. Direct oral anticoagulants (DOACs) are typically considered first line for various conditions such as atrial fibrillation.
- The Canadian Adult Obesity Clinical Practice Guidelines suggest avoiding the use of DOACs due to the potential for decreased drug absorption in gastric bypass patients26.
- Research has shown that warfarin can be safely taken in bariatric surgery patients although its absorption may be altered. However, the dose can be easily adjusted by monitoring INR levels. The evidence surrounding DOACs is limited and their levels are not typically monitored. For these reasons, vitamin K antagonists, such as warfarin, are the recommended agent for long term anticoagulation in post bariatric surgery patients19.
- A study published in 2020 in the American Journal of Cardiology concluded that DOACs have similar efficacy and safety compared to warfarin in bariatric surgery patients with atrial fibrillation. This is the largest study that currently exists comparing DOACs and warfarin in patients with prior bariatric surgery14.
- Bottom line: Although vitamin K antagonists are the recommended anticoagulant in bariatric surgery patients requiring anticoagulation, DOACs are likely a safe and effective alternative.
References
- Altieri MS, Wright B, Peredo A, Pryor AD. Common weight loss procedures and their complications. Am J Emerg Med. 2018;36(3):475-479
- Angrisani L, Santonicola A, Iovino P, et al. IFSO worldwide survey 2016: Primary, endoluminal, and revisional procedures. Obes Surg. 2018;28(12):3783-3794.
- Ballesta C, Berindoague R, Cabrera M, Palau M, Gonzales M. Management of Anastomotic Leaks After Laparoscopic Roux-en-Y Gastric Bypass. Obes Surg. 2008;18(6):623-630.
- Begian A, Samaan J, Hawley L, Alicuben E, Hernandez A, Samakar K. The use of nonsteroidal anti-inflammatory drugs after sleeve gastrectomy. Surgery for obesity and related diseases, 2021.
- Biertho L, Hong D, Gagner M. Canadian Adult Obesity Clinical Practice Guidelines: Bariatric Surgery: Surgical Options and Outcomes. Available from: https://obesitycanada.ca/guidelines/surgeryoptions. Accessed November 2021.
- Brown WA, Egberts KJ, Franke-Richard D, Thodiyil P, Anderson ML, OʼBrien PE. Erosions After Laparoscopic Adjustable Gastric Banding: Diagnosis and Management. Ann Surg. 2013;257(6):1047-1052.
- Chang S-H, Stoll CRT, Song J, Varela JE, Eagon CJ, Colditz GA. Bariatric surgery: An updated systematic review and meta-analysis, 2003-2012. JAMA Surg. 2014;149(3):275–287.
- Chevallier JM, et al. Predictive factors of outcome after gastric banding: a nationwide survey on the role of center activity and patients’ behavior. Ann Surg 2007;246:1034—9.
- Contival N, Menahem B, Gautier T, Le Roux Y, Alves A. Guiding the non-bariatric surgeon through complications of bariatric surgery. J Visc Surg. 2018;155(1):27-40.
- Cottam D, Qureshi FG, Mattar SG, et al. Laparoscopic sleeve gastrectomy as an initial weight-loss procedure for high-risk patients with morbid obesity. Surg Endosc Interv Tech. 2006;20(6):859-863.
- Dilauro M, McInnes MDF, Schieda N et al (2017) Internal hernia after laparoscopic Roux-en-Y gastric bypass: optimal CT signs for diagnosis and clinical decision making. Radiology 282:752–760.
- Ellsmere J. Late complications of bariatric surgical operations. In: UpToDate, UpToDate, Waltham, MA.
- Hamilton EC, Sims TL, Hamilton TT, Mullican MA, Jones DB, Provost DA. Clinical predictors of leak after laparoscopic Roux-en-Y gastric bypass for obesity. Surgical Endoscopy, 2003.
- Hendricks A, et al.Safety and efficacy of oral anticoagulants for atrial fibrillation in patients after bariatric surgery. The American Journal of Cardiology, 2020.
- Higa KD, Ho T, Boone KB. Internal hernias after laparoscopic Roux-en-Y gastric bypass: incidence, treatment and preven- tion. Obes Surg 2003;13:350—4.
- Ianelli A, Treacy P, Sebastianelli L, Schiavo L, Martini F. Perioperative complications of sleeve gastrectomy: review of the literature. J Minim Access Surg, 2019.
- Jossart G. Complications of sleeve gastrectomy: bleeding and prevention. Surgical laparoscopy, endoscopy and percutaneous techniques, 2010.
- Luber SD, Fischer DR, Venkat A. Care of the Bariatric Surgery Patient in the Emergency Department. J Emerg Med. 2008;34(1):13-20.
- Martin K, Lee C, Farrell T, Moll S. Oral anticoagulant use after bariatric surgery: a literature review and clinical guidance. AJM, 2017.
- Melinek J, Livingston E, Cortina G, Fishbein MC. Autopsy findings following gastric bypass surgery for morbid obesity. Arch Pathol Lab Med, 2002.
- Msika S. [Surgical treatment of morbid obesity by gastrojejunal bypass using laparoscopic roux-en-Y (gastric short circuit)]. J Chir (Paris) 2002;139:214—7.
- Pieroni, Sommer E, Hito R, Burch M, Tkacz J. The “O” Sign, a simple and helpful tool in the diagnosis of laparoscopic adjustable gastric band slippage. AJR, 2010.
- Public Health Agency of Canada. Obesity in Canada: A Joint Report from the Public Health Agency of Canada and the Canadian Institute for Health Information.; 2011. http://www.phac-aspc.gc.ca/hp-ps/hl-mvs/oic-oac/assets/pdf/oicoac-eng.pdf.
- Rasmussen JJ, Fuller W, Ali MR. Marginal ulceration after laparoscopic gastric bypass: an analysis of predisposing factors in 260 patients. Surg Endosc 2007;21:1090—4.
- Regusci L, Groebli Y, Meyer JL, et al. Gastroscopic removal of an adjustable gastric band after partial intragastric migration. Obes Surg 2003; 13:281.
- Shiau J, Biertho L. Canadian Adult Obesity Clinical Practice Guidelines: Bariatric Surgery: Postoperative Management. Downloaded from: https://obesitycanada.ca/guidelines/postop. Accessed November 2021.
- Suter M, Giusti V, Heraief E, Calmes JM. Band erosion after laparoscopic gastric banding: occurrence and results after conversion to roux-en-y gastric bypass. Obes Surg 2004; 14:382.
- Twells LK, Gregory DM, Reddigan J, Midodzi WK. Current and predicted prevalence of obesity in Canada: A trend analysis. CMAJ Open. 2014;2(1):E18-E26.
- Twells LK, Janssen I, Kuk JL. Canadian Adult Obesity Clinical Practice Guidelines: Epidemiology of Adult Obesity. Available from: https://obesitycanada.ca/guidelines/epidemiology.
- Wilson J, Romagnuolo J, Byrne T, Morgan K, Wilson F. Predictors of endoscopic findings after Roux-en_y gastric bypass. AM J Gastroenterol, 2006.
- Wittgrove AC, Clark GW. Laparoscopic gastric bypass, roux en y 500 patients: technique and results, with 3-60 month followup. Obes Surg, 2000.
- World Health Organization. Obesity and Overweight. https://www.who.int/ news-room/fact-sheets/detail/obesity-and-overweight. Published 2018.
- Yska J, Gertsen S, Flapper G, Emous M, Wilffert B, van Roon E. NSAID USE after bariatric surgery: a randomized controlled intervention study. Obseity surgery, 2016.