The opioid epidemic is not new to ED providers, but many will be shocked to learn about the true extent of the harm caused by opioids in Canada. In the first half of 2022, there were 3556 recorded deaths due to opioid toxicity in Canada.
This amounts to 19 deaths every day.
On an average, we see 7 opioid-related deaths and 16 opioid-related hospitalizations per day in Ontario. This problem got dramatically worse during the COVID-19 pandemic. In the first two years of the COVID-19 pandemic, Canada saw a 91% increase in deaths due to opioid overdose. Moreover, these numbers only capture deaths from opioid overdose. We don’t have data on secondary harms, such as soft tissue infections, endocarditis, and physical injury due to opioid use.
In the face of what seems like such an insurmountable problem, some ED providers may understandably feel helpless when it comes to treating patients with substance use. But in truth, there are life-saving interventions for opioid use disorder that we can safely and easily initiate in the emergency department: Buprenorphine is a form of opioid agonist therapy, and is considered the first line treatment for opioid use disorder. For a variety of reasons, we are currently under prescribing this medication in the ED. In this post, I will provide you with a simple, easy-to-follow algorithm for prescribing this potentially life-saving medication to patients in your ED.
*For a comprehensive approach to initiating buprenorphine in the ED, jump to Take home #5.
Take home #1: ED visits are critical opportunities to initiate lifesaving interventions for people who use drugs
Patients who survive an opioid overdose are at exceedingly high risk of mortality compared to the general population. 1 out of every 19 patients who overdose with opioids will die within the next year. Studies have repeatedly shown the one-year mortality for patients following an opioid overdose to be just above 5%. For comparison, the 1-year mortality of patients following nonfatal myocardial infarction is 5.9%.
Patients who die from an overdose also visit the ED with a much greater frequency than the general public. 54% of patients with an overdose visited an ED within the last year. People who die from overdose visit the ED 9.6x more than age-matched controls.
When you see a 29 year-old male patient in the morning who was treated for an opioid overdose, take a minute to consider that this patient is at an extremely high risk of dying within the next year compared to other adults in our ED. Ask yourself, “is there anything I can do to prevent this person from dying?”
Take home #2: Buprenorphine has several qualities that make it extremely useful for treating opioid use disorder.
For a complete overview of Suboxone, see METAPHI’s amazing reference guide here. For an overview on opioid agonist therapy, and treating patients on it, see the previous EMOttawa blog post by Dr. Isabelle Miles here.
Suboxone includes two medications as a combination pill: Buprenorphine and Naloxone.

Buprenorphine has three qualities that make it useful in the treatment of opioid use disorder:
-
- Partial opioid agonist. Buprenorphine provides enough opioid agonist activity to prevent withdrawal symptoms and cravings, with less euphoria and sedation than full agonist opioids
- High affinity, slow dissociation. Buprenorphine binds strongly to mu-opioid receptors and dissociates slowly, preventing withdrawal symptoms for 24 hours and beyond. The high affinity for opioid receptors means that it is not displaced by other opioids. It blocks the activity of other opioids used concurrently, making the use of other opioids less rewarding and reinforcing.
- Ceiling effect. Doses beyond 24–32mg do not have additional effects with regard to respiratory depression. As a result, the risk of respiratory depression and overdose is substantially reduced relative to other opioids.
Finally, Naloxone is included in Buprenorphine tablets (e.g., Suboxone®️) to reduce the risk of diversion through injection; it is not absorbed when tablets are taken sublingually and does not impact the action of Buprenorphine.
Take home #3: Prescribing Buprenorphine to patients with opioid use disorder increases retention in treatment and may reduce mortality.
A 2018 Canadian Research Initiative in Substance Misuse (CRISM) guideline published in CMAJ recommends Buprenorphine-Naloxone (brand name Suboxone) as the first line treatment for opioid use disorder (strong recommendation, high quality of evidence). Let’s briefly dig into the evidence behind these recommendations.

https://crism.ca/wp-content/uploads/2018/03/CRISM_NationalGuideline_OUD-ENG.pdf
Evidence
A 2014 Cochrane review compared Buprenorphine to either placebo or Methadone maintenance in the management of opioid dependence. They included only RCTs of buprenorphine maintenance versus methadone maintenance or versus placebo medication in the management of opioid dependence. The population they examined was all individuals with opioid dependence, regardless of the specific substance used. Pregnant women were excluded.
They looked at the following outcomes:
-
- Retention in treatment
- Non-prescription opioid use
- Use of other substances
- Mortality
- Criminal activity
Overall, 31 studies with 5430 participants (predominantly male heroin users) were included. Quality of evidence was high-moderate. See their major findings below:
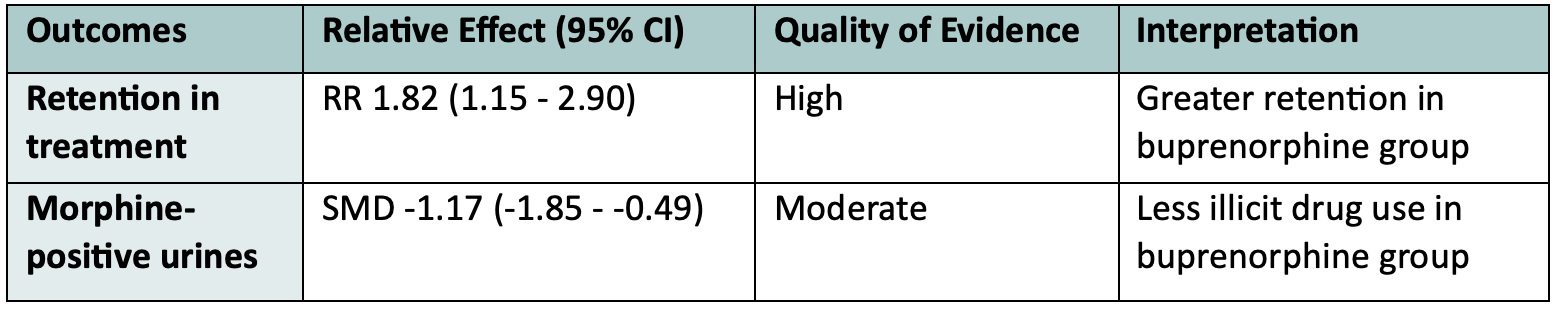
They concluded that Buprenorphine is effective in retaining people in treatment at any dose above 2 mg, and that at doses above 16 mg it suppresses non-prescribed opioid use. Unfortunately, very few studies reported mortality, and in those that did, the overall number of deaths was quite low.
The best evidence we have regarding the impact of Buprenorphine on mortality comes from observational data. In 2018, Larochelle et al. published a retrospective cohort study of 17 568 Massachusetts adults who had survived an opioid overdose between 2012 and 2014, excluding patients who died within 30 days and those with cancer. They followed these patients for 12 months, examining four groups treatment for OUD: Methadone (11%), Buprenorphine (12%), Naltrexone (4%), Multiple treatments (5%), and comparing these to those not on any treatment. They found that the all-cause mortality was 5 per 100 person years in those not on treatment, compared to 3.1 on Buprenorphine. The adjusted hazard ratio for mortality in patients on Buprenorphine was 0.63 compared to those not being on treatment!
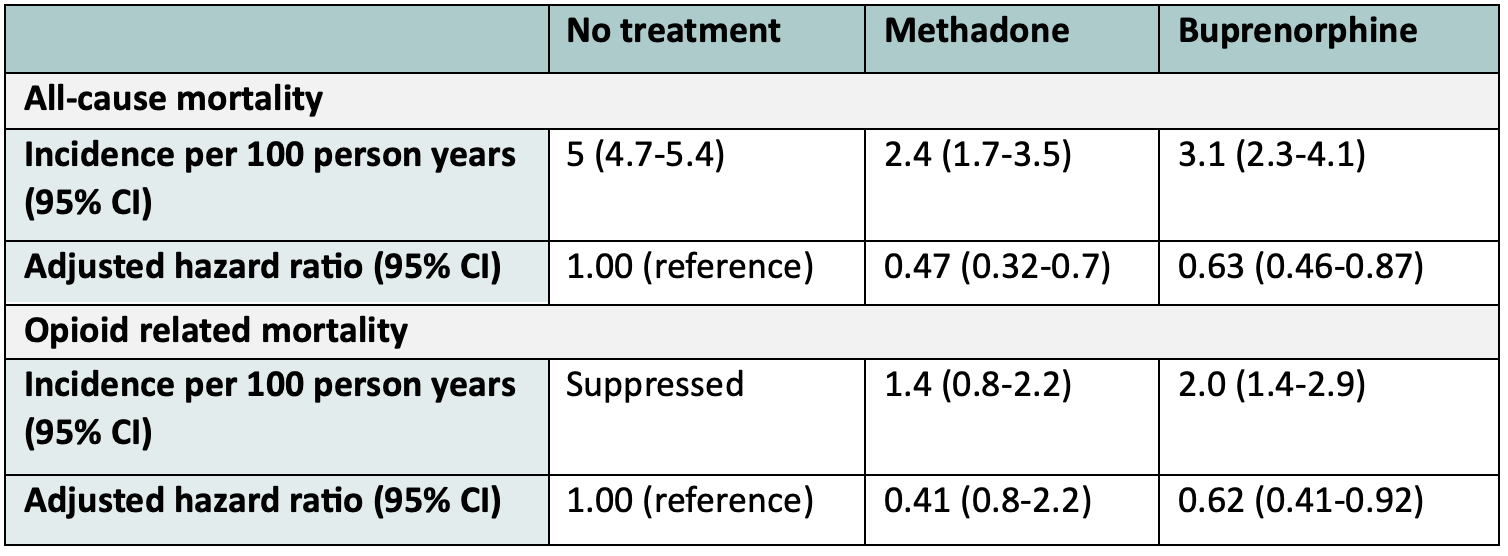
While this paper has several limitations due to being an observational study, it provides real world evidence that those on Buprenorphine have decreased overall mortality than those not on treatment.
Finally, let’s look at the evidence for starting this medication in the ED.
In 2015, D’Onofrio et al. published an RCT in JAMA examining ED-initiated Buprenorphine for opioid dependence. They included 329 opioid-dependent adults in seen in the ED at a large urban teaching hospital, who had non-medically used prescription opioids or heroin in the preceding 30 days, and were not enrolled in addiction treatment. The randomized these patients as follows:
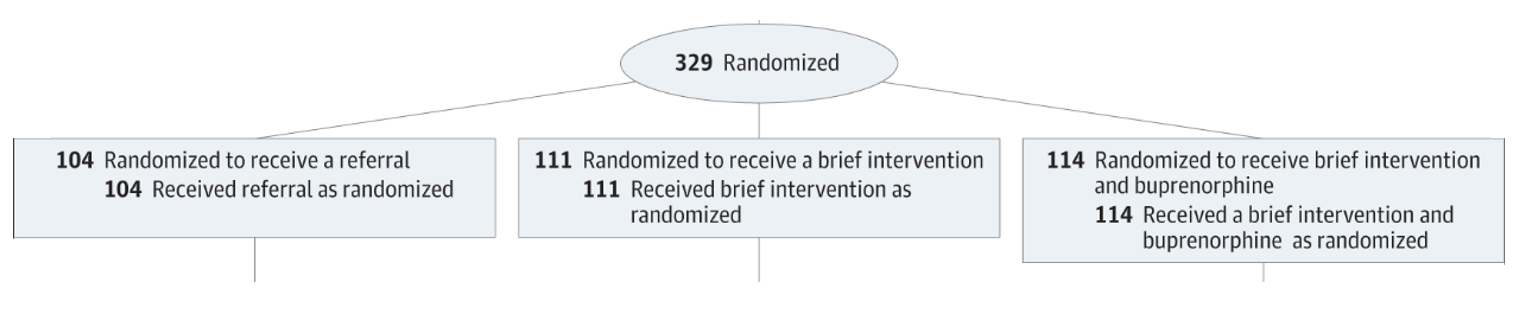 From: D’Onofrio et al. (2015) https://doi.org/10.1001/jama.2015.3474
From: D’Onofrio et al. (2015) https://doi.org/10.1001/jama.2015.3474
The primary outcome was retention in treatment at 30 days. These were their results:
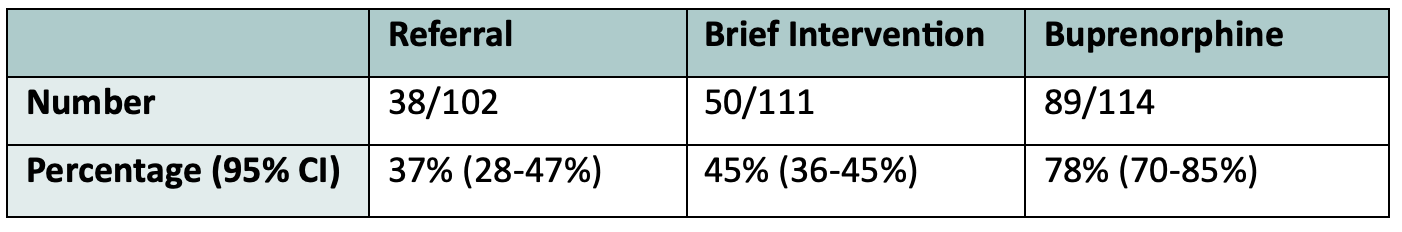
As you can see, the rates of engagement in treatment at 30 days for patients started on Buprenorphine in the ED was double that of patients who just received a referral to an addictions centre. Click here for an EMOttawa journal club deeper dive into this study.
Take home #4: As emergency physicians, we are currently under-prescribing Suboxone in the ED.
There is good evidence to suggest that we can do better when it comes to prescribing this potentially life-saving therapy from the ED. In 2020, Hoyeck et al. conducted a cross-sectional study of 179 emergency physicians across Canada. They asked physicians about their current management practices with respect to opioid use disorder. Only 5% of respondents reported often or always providing outpatient prescriptions for buprenorphine, and 7.4% reported often or always prescribing it in the ED. Another 2020 study performed a survey of physicians at 4 academic EDs in Baltimore, NY, Seattle and Cincinatti. They found only 21% of respondents ready to initiate buprenorphine in the ED.
Barriers to prescribing Suboxone from the ED identified in the literature include:
-
- Lack of experience and comfort with this medication
- Fear of precipitating withdrawal
- Perceived time limitations
- Limited knowledge of local treatment resources
- Perceptions about it being outside the scope of practice of emergency physicians
Take home #5: Initiating Suboxone in the ED is easy and provides a valuable bridge to further care. Here are your easy-to follow steps for prescribing this medication in the ED.
Note: these algorithms are largely based on those published on METAPHI’s website. For a guide on Suboxone for ED providers as well as countless other useful resources, access their published toolkit for ED providers here.
1. Consider Buprenorphine-naloxone (Suboxone) in any patient presenting with:
| 1. Opioid withdrawal |
| 2. Opioid overdose |
| 3. Complication of opioid use |
2. Assess for exclusion criteria to Buprenorphine.
Exclusion criteria to Buprenorphine include:
|
1. Allergy or hypersensitivity to buprenorphine or naloxone |
|
2. Methadone use in the last 72h |
|
3. Inability to provide informed consent |
|
4. Altered mental status or delirium |
|
5. Acute intoxication |
|
6. Unstable medical illness |
3. Assess for the possibility of precipitated withdrawal.
Precipitated withdrawal is a state of acute and severe withdrawal that occurs if the initial dose of Buprenorphine is given when the patient still has other opioids active on the receptors.
Because Buprenorphine is a partial opioid agonist with high affinity, it displaces other opioids from opioid receptors. The partial mu agonist effect of buprenorphine may not fully compensate for the loss of the current opioid. This results in severe withdrawal symptoms due to a net opioid deficit.
To prevent precipitated withdrawal, it is important to ensure that the patient is already in moderate withdrawal before starting Suboxone.
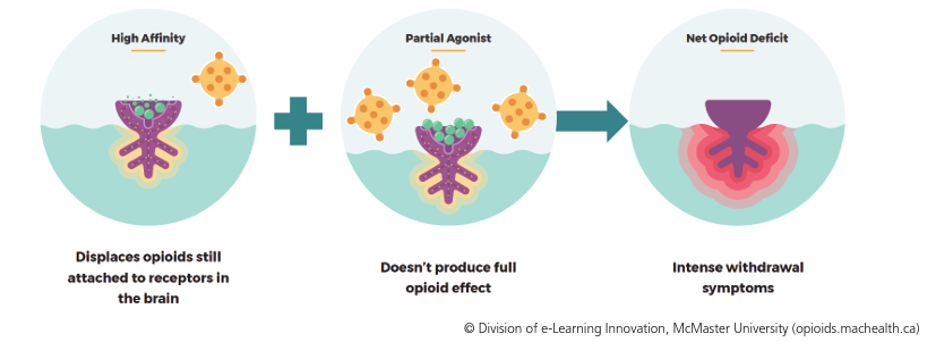
With that said, it is important to know three things before starting Suboxone:
| 1. The timing of the last used opioid |
| 2. The specific opioid used |
| 3. Degree of withdrawal |
To assess the degree of withdrawal, we use the Clinical Opiate Withdrawal Scale (COWS). A COWS score of 13 or greater indicates moderate withdrawal. For a more comprehensive version of the COWS score, see here.
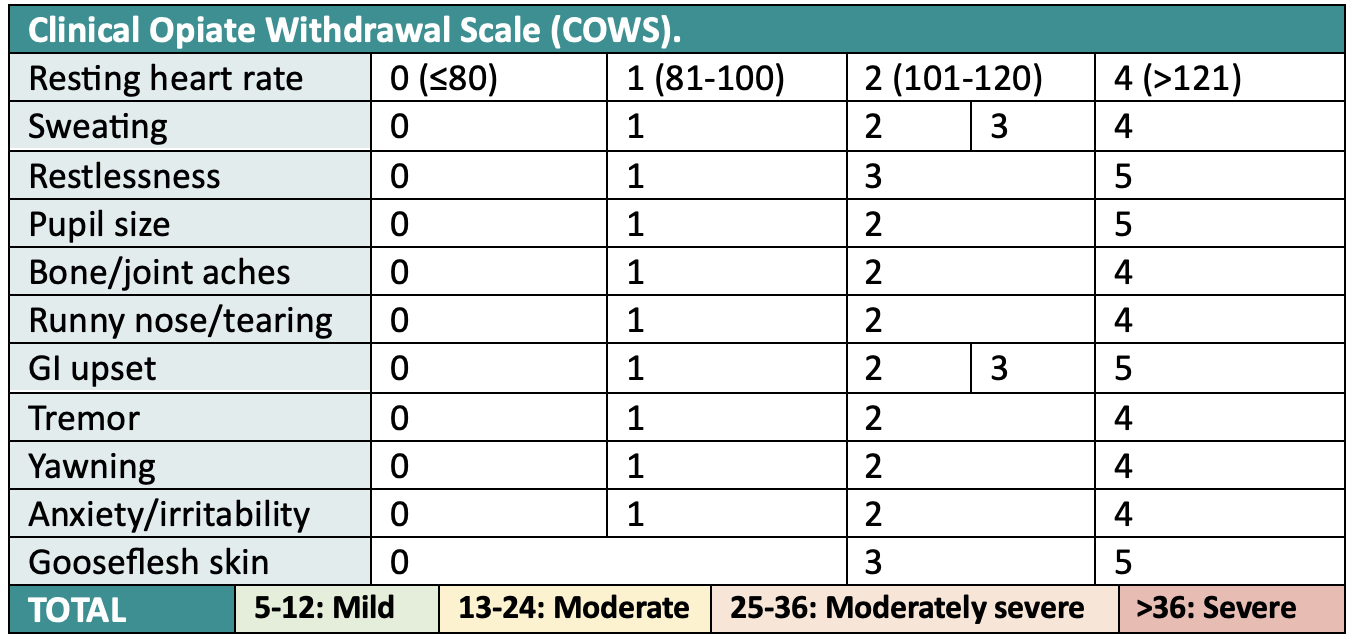
Important additional points:
-
- Withdrawal in and of itself is not dangerous, except potentially in people who are elderly, frail, have cardiac or neurovascular disease
- Outside of these groups, the main danger about opioid withdrawal is that it makes people feel terrible. Beyond being painful and unpleasant for patients, this may harm the therapeutic relationship and lead to patients avoiding care in the future.
- You will NOT cause precipitated withdrawal by giving opioids to patients already on Suboxone. You may just needed higher doses of opioids than you’re used to in order to treat pain in this population.
Once you have confirmed that your patient is in moderate withdrawal (COWS of 13 or more), as well as the timing of last use and the specific opioid used, move on to step 4.
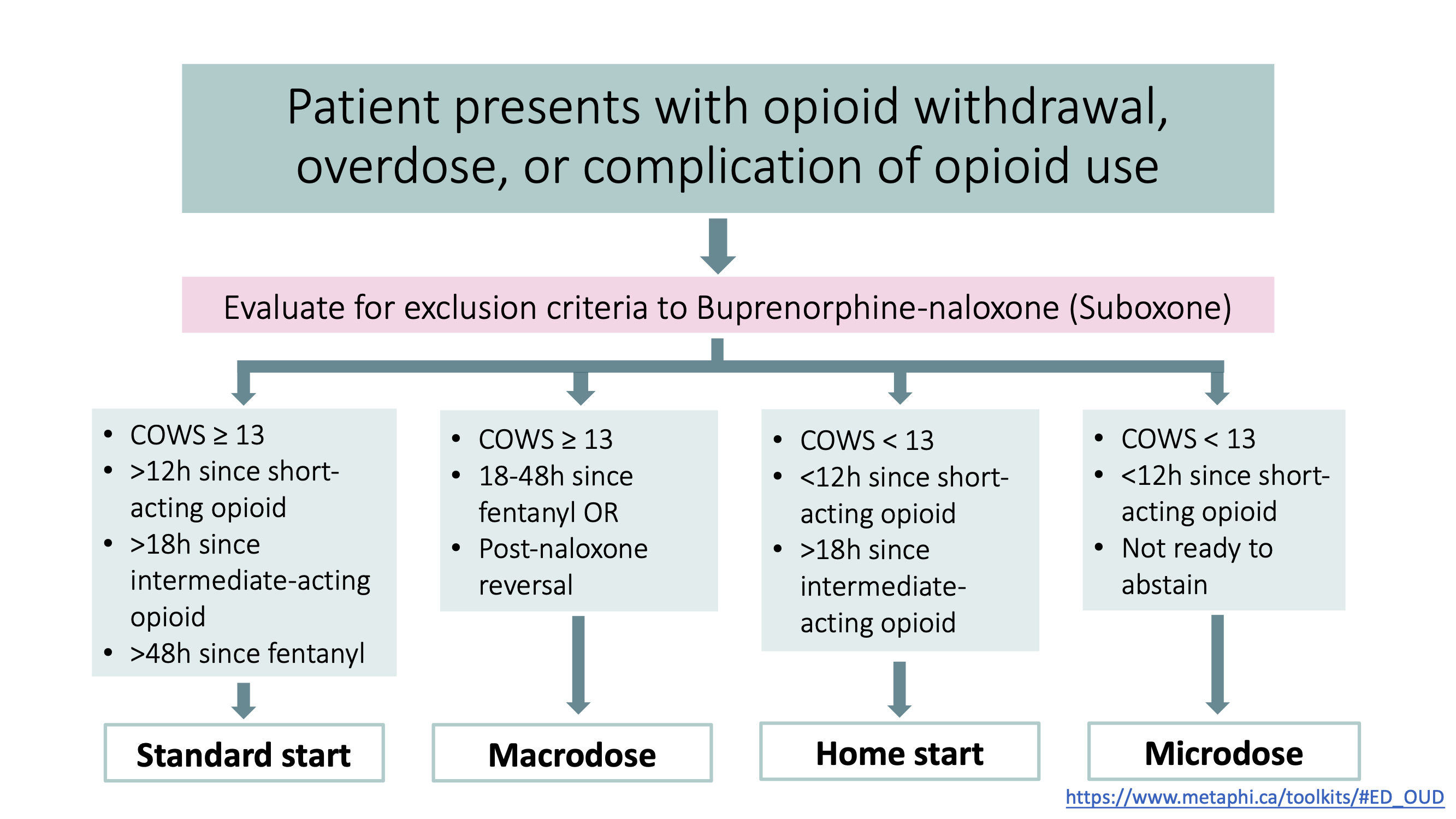
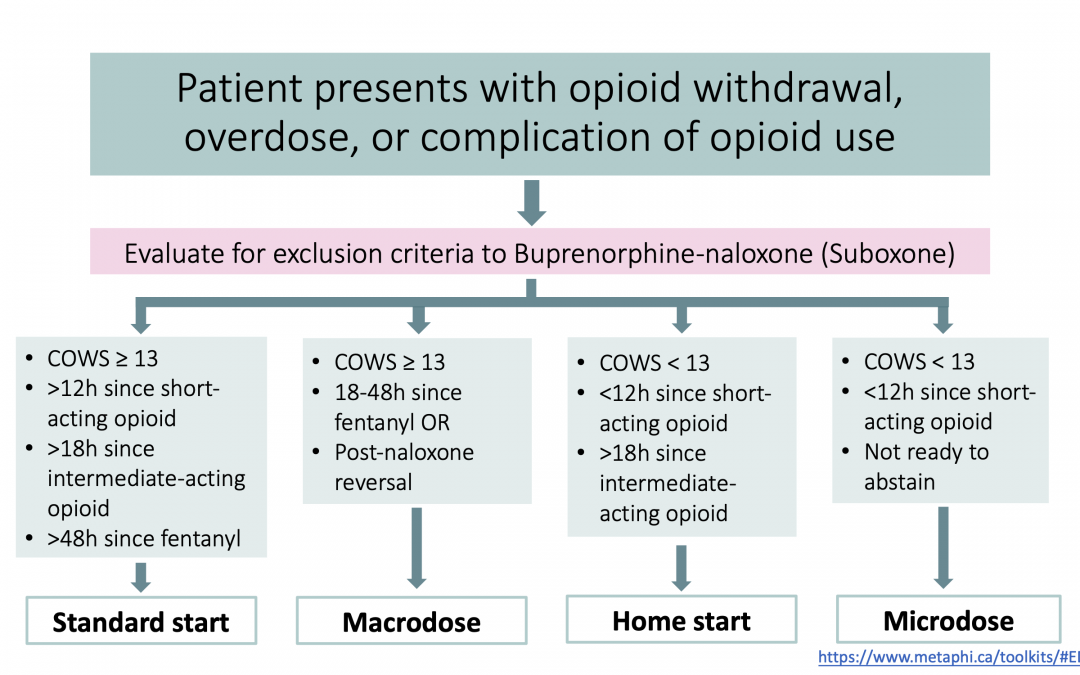
4. Choose an induction method.
There are 4 general options for starting Suboxone in the ED. They are:
| 1. Standard start |
| 2. Home start |
| 3. Macrodosing |
| 4. Microdosing |
Each one of these methods is discussed in detail below. For an algorithm to help decide which induction method is best for your patient, scroll to the bottom of this page.
| Standard start |
This is the default approach for starting Buprenorphine-naloxone (Suboxone) in the ED. Below is an easy-to-follow algorithm published by METAPHI for standard starts in the ED. A link to this pdf can also be found here.
After starting the patient on Buprenorphine in the ED, use this template for writing a discharge prescription.
Remember, any patient receiving a prescription for Buprenorphine should also receive a referral to your local Rapid Access Addiction Medicine (RAAM) clinic. A list of RAAMs in Canada can be found here.
| Home start |
If you have someone who is motivated to abstain from opioid use, and meets all standard start criteria, but is not yet in sufficient withdrawal for a standard start, they can actually perform a standard start at home.
A home start will not be suitable for everyone. Indications are as follows:
-
- Standard start criteria with COWS < 13
- Can abstain from opioid use
- Can follow instruction sheet
- Support at home
- No concurrent benzodiazepine or alcohol use
First, write the patient their home start prescription using this template. The prescription will look like this:
Next, provide the patient with a copy of this handout with a guide for starting the medication at home. As shown below, this is an easy-to-follow sheet that guides the patient through symptoms
| Macrodosing |
This is an alternative approach to initiating Buprenorphine for patients who don’t meet standard criteria. Many patients seen in the ED post-overdose would benefit from Buprenorphine. However, use of fentanyl within 48 hours of presentation is an exclusion for standard Buprenorphine initiation.
Moreover, standard initiation suggests a maximum Day 1 dose of 16mg of Buprenorphine; this dose is generally inadequate to provide relief of withdrawal symptoms for people who use fentanyl.
So, instead of providing a moderate dose of Buprenorphine that could precipitate withdrawal, we give a large enough dose in the ED such that Buprenorphine will provide enough of an agonist effect to overcome withdrawal symptoms. A primer on macrodosing can be found here.
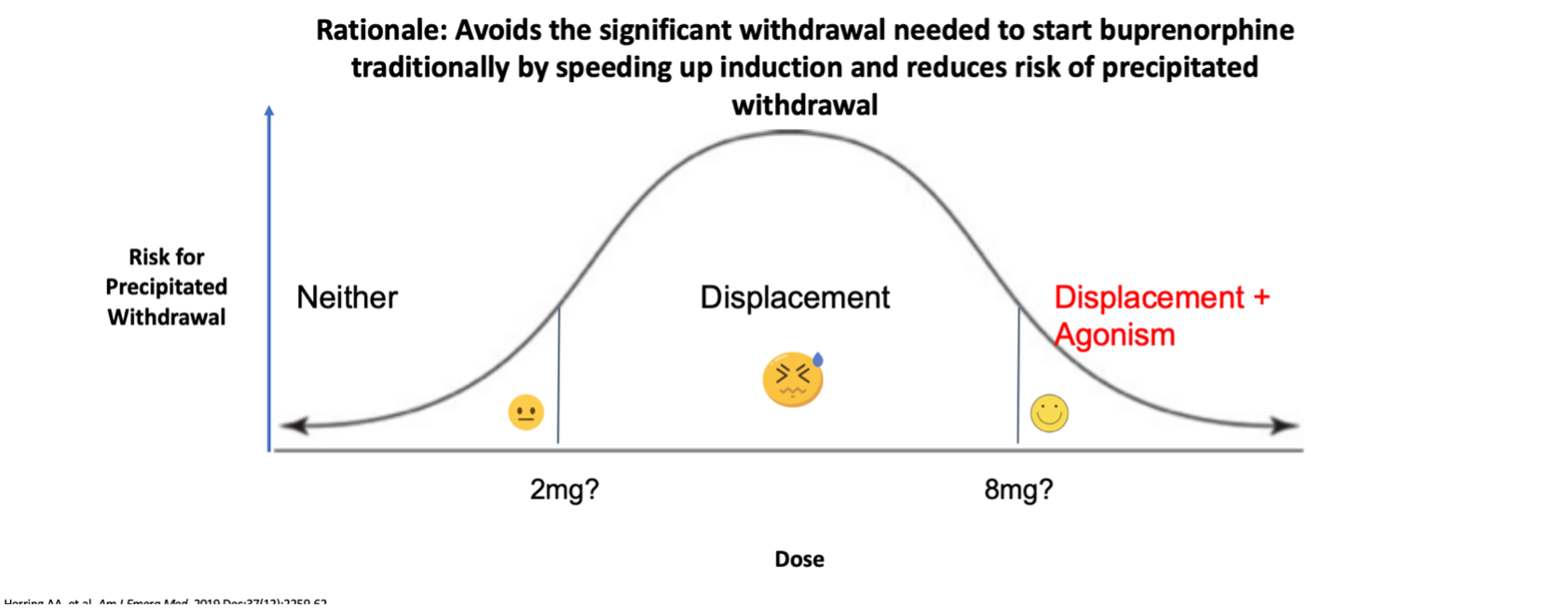
See below an algorithm for macrodosing in the ED.
| Microdosing |
This approach is useful for patients with the following characteristics:
-
- Using methadone or street fentanyl
- Continued opioid use, not ready to abstain
- Should not undergo withdrawal for medical reasons
In these patients, we can start Buprenorphine at a low enough dose that there is no risk of precipitated withdrawal, but we still get some of the benefits of being on buprenorphine.
This is the safest and easiest option for patients who continue to use opioids.
It is assumed that these patients will continue using opioids, and that is ok. But while on Buprenorphine, those drugs are going to be less rewarding. And when they’re not using, they’re still going to have some relief from the Buprenorphine.
And finally, by the time they do get to a RAAM clinic, their dose can then safely be rapidly escalated to one that is therapeutic.
Use this template to write a microdosing prescription, and provide the patient with this handout.

http://www.metaphi.ca/wp-content/uploads/ED_OUD_RxMicrodosing.pdf
When considering which approach to use for Buprenorphine initiation, consult the following flow chart:
Final pearls and pitfalls
- Suboxone only works when taken sublingually. Communicate with nurses to make sure the doses are not swallowed.
- Buprenorphine will not be suitable for patients already receiving a prescription for methadone. If a patient is on methadone, consider dispensing them their usual dose in the ED, and having them follow up with their addictions provider.
- Some patients may be reluctant to take buprenorphine for various reasons. For example, they may worry it will make them feel sick, or have had a bad experience with it in the past if it wasn’t started properly. Listen to the patient’s perspective, and try to address these concerns. Ask them, “what do you know about Suboxone?”.
- Consider the following phrasing:
“Suboxone can help you feel a lot better. We can get it started quickly and make arrangements for you to follow up. It will help more than anything else I can give you in the ED. And if you decide you want to switch to another medication it’s really straight forward.”
- Many patients with substance use disorder come to the ED expecting to be treated with judgement and implicit criticism, The language and tone we use with these patients is important.
- Suboxone doesn’t have to be a commitment to long-term opiod agonist therapy or to abstinence! It can treat withdrawal, prevent overdose while someone is taking it and provide a bridge to longer-term care.
References
- Bruneau, J., Ahamad, K., Goyer, M.-È., Poulin, G., Selby, P., Fischer, B., Wild, T. C., & Wood, E. (2018). Management of opioid use disorders: a national clinical practice guideline. Canadian Medical Association Journal, 190(9), E247 LP-E257. https://doi.org/10.1503/cmaj.170958
- Canadian Institute for Health Information. (n.d.). Opioid-Related Harms in Canada.
- D’Onofrio, G., O’Connor, P. G., Pantalon, M. v, Chawarski, M. C., Busch, S. H., Owens, P. H., Bernstein, S. L., & Fiellin, D. A. (2015). Emergency department-initiated buprenorphine/naloxone treatment for opioid dependence: a randomized clinical trial. JAMA, 313(16), 1636–1644. https://doi.org/10.1001/jama.2015.3474
- D’Onofrio, G., Chawarski, M. C., O’Connor, P. G., Pantalon, M. v, Busch, S. H., Owens, P. H., Hawk, K., Bernstein, S. L., & Fiellin, D. A. (2017). Emergency Department-Initiated Buprenorphine for Opioid Dependence with Continuation in Primary Care: Outcomes During and After Intervention. Journal of General Internal Medicine, 32(6), 660–666. https://doi.org/10.1007/s11606-017-3993-2
- D’Onofrio, G., McCormack, R. P., & Hawk, K. (2018). Emergency Departments — A 24/7/365 Option for Combating the Opioid Crisis. New England Journal of Medicine, 379(26), 2487–2490. https://doi.org/10.1056/NEJMp1811988
- Duber, H. C., Barata, I. A., Cioè-Peña, E., Liang, S. Y., Ketcham, E., Macias-Konstantopoulos, W., Ryan, S. A., Stavros, M., & Whiteside, L. K. (2018). Identification, management, and transition of care for patients with opioid use disorder in the emergency department. Annals of Emergency Medicine, 72(4), 420–431.
- Fudala, P. J., Bridge, T. P., Herbert, S., Williford, W. O., Chiang, C. N., Jones, K., Collins, J., Raisch, D., Casadonte, P., Goldsmith, R. J., Ling, W., Malkerneker, U., McNicholas, L., Renner, J., Stine, S., & Tusel, D. (2003). Office-Based Treatment of Opiate Addiction with a Sublingual-Tablet Formulation of Buprenorphine and Naloxone. New England Journal of Medicine, 349(10), 949–958. https://doi.org/10.1056/NEJMoa022164
- Hawk, K. F., D’Onofrio, G., Chawarski, M. C., O’Connor, P. G., Cowan, E., Lyons, M. S., Richardson, L., Rothman, R. E., Whiteside, L. K., Owens, P. H., Martel, S. H., Coupet Jr, E., Pantalon, M., Curry, L., Fiellin, D. A., & Edelman, E. J. (2020). Barriers and Facilitators to Clinician Readiness to Provide Emergency Department–Initiated Buprenorphine. JAMA Network Open, 3(5), e204561–e204561. https://doi.org/10.1001/jamanetworkopen.2020.4561
- Herring, A. A., Vosooghi, A. A., Luftig, J., Anderson, E. S., Zhao, X., Dziura, J., Hawk, K. F., McCormack, R. P., Saxon, A., & D’Onofrio, G. (2021). High-Dose Buprenorphine Induction in the Emergency Department for Treatment of Opioid Use Disorder. JAMA Network Open, 4(7), e2117128. https://doi.org/10.1001/jamanetworkopen.2021.17128
- Hoyeck, P., Wiercigroch, D., Clarke, C., Moineddin, R., Sheikh, H., & Hulme, J. (2020). Understanding current practice of opioid use disorder management in emergency departments across Canada: A cross-sectional study. CJEM, 22(4), 494–498. https://doi.org/10.1017/cem.2020.362
- Hu, T., Snider-Adler, M., Nijmeh, L., & Pyle, A. (2019). Buprenorphine/naloxone induction in a Canadian emergency department with rapid access to community-based addictions providers. CJEM, 21(4), 492–498. https://doi.org/10.1017/cem.2019.24
- Koh, J. J., Klaiman, M., Miles, I., Cook, J., Kumar, T., Sheikh, H., Dong, K., Orkin, A. M., Ali, S., & Shouldice, E. (2020). CAEP position statement: emergency department management of people with opioid use disorder. Canadian Journal of Emergency Medicine, 22(6), 768–771.
- Krawczyk, N., Eisenberg, M., Schneider, K. E., Richards, T. M., Lyons, B. C., Jackson, K., Ferris, L., Weiner, J. P., & Saloner, B. (2020). Predictors of Overdose Death Among High-Risk Emergency Department Patients With Substance-Related Encounters: A Data Linkage Cohort Study. Annals of Emergency Medicine, 75(1), 1–12. https://doi.org/10.1016/j.annemergmed.2019.07.014
- Larochelle, M. R., Bernson, D., Land, T., Stopka, T. J., Wang, N., Xuan, Z., Bagley, S. M., Liebschutz, J. M., & Walley, A. Y. (2018). Medication for Opioid Use Disorder After Nonfatal Opioid Overdose and Association With Mortality: A Cohort Study. Annals of Internal Medicine, 169(3), 137–145. https://doi.org/10.7326/M17-3107
- Ling, W., Charuvastra, C., Collins, J. F., Batki, S., Brown Jr, L. S., Kintaudi, P., Wesson, D. R., Mcnicholas, L., Tusel, D. J., Malkerneker, U., Renner, J. A., Santos, E., Casadonte, P., Fye, C., Stine, S., Wang, R. I. H., & Segal, D. (1998). Buprenorphine maintenance treatment of opiate dependence: a multicenter, randomized clinical trial. Addiction, 93(4), 475–486. https://doi.org/https://doi.org/10.1046/j.1360-0443.1998.9344753.x
- Margaret, L., Rachel, M., Dina, A.-R., Jeanmarie, P., F., M. Z., Nicole, O., Christian, W., Gabrielle, S., Rinad, B., & Kit, D. M. (2022). Redesign of Opioid Use Disorder Screening and Treatment in the ED. NEJM Catalyst, 3(1). https://doi.org/10.1056/CAT.21.0297
- Mattick, R. P., Breen, C., Kimber, J., & Davoli, M. (2014). Buprenorphine maintenance versus placebo or methadone maintenance for opioid dependence. Cochrane Database of Systematic Reviews, 2. https://doi.org/10.1002/14651858.CD002207.pub4
- (2021). Bupenorphine/Naloxone (Suboxone). Reference Guide for ED Providers. https://www.metaphi.ca/toolkits/#ED-OUD
- Moe, J., Chong, M., Zhao, B., Scheuermeyer, F. X., Purssell, R., & Slaunwhite, A. (2021). Death after emergency department visits for opioid overdose in British Columbia: a retrospective cohort analysis. CMAJ Open, 9(1), E242 LP-E251. https://doi.org/10.9778/cmajo.20200169
- O’Connor, S., Grywacheski, V., & Louie, K. (2018). At-a-glance – Hospitalizations and emergency department visits due to opioid poisoning in Canada. Health Promotion and Chronic Disease Prevention in Canada : Research, Policy and Practice, 38(6), 244–247. https://doi.org/10.24095/hpcdp.38.6.04
- Otterstatter, M. C., Crabtree, A., Dobrer, S., Kinniburgh, B., Klar, S., Leamon, A., May-Hadford, J., Mill, C., Park, M., & Tu, A. W. (2018). Patterns of health care utilization among people who overdosed from illegal drugs: a descriptive analysis using the BC Provincial Overdose Cohort. Health Promotion and Chronic Disease Prevention in Canada, 38(9).









Trackbacks/Pingbacks