Right here, Right now!
Pumping up our Knowledge on Right Ventricular Failure and Acute Pulmonary Embolism
The treatment of patients with right ventricular failure from pulmonary embolism follows some key principles that differ from our typical resuscitation of hypotension and hypoxemic respiratory failure. This typical management will not only not help these patients, but can actually be fatal.
RV failure and acute PE management matters to us in the emergency department because:
- The mortality rate for unstable patients with a PE is 50% 1.
- For all patients with PE, the in-hospital mortality ~7% 2.
- Up to 10% of PE cases are fatal within the first hour of symptoms 3.
- Death due to PE is largely from progressive RV failure.
Recognition and the appropriate management of acute RV failure is critical in the emergency department. Routine interventions often performed early in a patient’s ED course and a delay in definitive therapy can lead to deterioration in these patients. It is important for us to be comfortable with the ED management of acute RV failure. There is, truthfully, not a robust body of evidence to guide ED management. Recently, more attention has been placed in understanding RV anatomy, physiology and dysfunction, and most of the recommendations now around managing RV failure come from an understanding of this pathophysiology. Before getting straight to the management of these patients, we first need to briefly review the pathophysiology of acute pulmonary hypertension and RV failure. In this post, we will:
- Provide an overview for the pathophysiology of right ventricular (RV) failure in acute pulmonary embolism (PE)
- Outline the principles of managing a patient with RV failure and acute PE
- Provide a brief review of the evidence for thrombolytics and other therapy options in an acute PE
- Discuss the role of Thrombosis and PERT in the management of acute PE
Pathophysiology of RV Failure in Acute PE
RV failure can produce a set of vicious spirals, or a ‘spiral of death’ as you may have read of before. This tends to cause progressively worse RV failure, and if, left untreated or mistreated, this will spiral out of control. This explains why patients with RV failure can suddenly die despite, or because of, our interventions- and this is what makes an acute PE so dangerous. Let’s break this ‘spiral’ down…
There are 3 main causes or precipitants of RV failure, or essentially 3 ways to enter the spiral:
- RV pressure overload
- Reduced RV contractility
- RV volume overload
We’ll focus on RV pressure overload in the context of an acute PE.
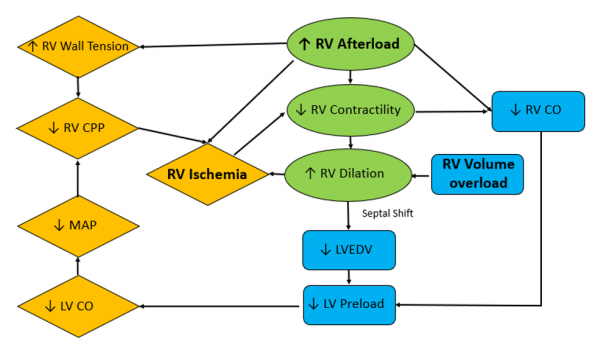
Adapted from Right Heart Failure with Sara Crager, EMCrit 272 https://www.tomwademd.net/links-to-emcrit-272-right-heart-failure-with-dr-sara-crager/
An acute PE can cause a sudden hike in RV afterload by an acute increase in pulmonary vascular resistance. This leads to a rapid decrease in stroke volume and contractility, leading to RV dilation. This RV volume and pressure overload results in a decrease in RV perfusion, leading to ischemia. RV dilation also leads to interventricular septum deviation toward the left ventricle. This limits LV filling and preload, which is followed by a decrease in cardiac output and systemic hypotension. Ultimately, this results in decreased RV coronary perfusion and ischemia, and worsening RV function.
Any of the boxes highlighted above can act as entry points to the cycle of acute RV failure. Processes that can contribute to this cycle include pathologic processes, such as an acute PE or arrythmias, but also unfortunately our own interventions, such as administration of large volumes of fluid.
It is therefore important that we follow an RV-centric resuscitation.
Principles of Management of RV Failure in an acute PE
There are 4 main principles that guide management of RV failure in an acute PE:
- Appropriate fluid resuscitation
- Supporting the systemic blood pressure
- Decreasing RV afterload
- Avoiding intubation
1. Appropriate fluid resuscitation
The initial strategy in managing most hemodynamically unstable patients is often to increase RV preload with a bolus of IV fluids. The failing and dilated RV, however, will not tolerate overfilling. There is limited experimental data on fluid administration in RV failure. Experimental models in PE and pulmonary HTN have shown that aggressive volume expansion can worsen RV function 5.
If we return to the image of the RV spiral diagram above, we can see that excessive volume resuscitation can exacerbate RV failure by causing further distention of the RV. This can lead to RV ischemia, limiting LV filling and decreasing CO. Even smaller boluses can be poorly tolerated in acute RV failure. If fluid is considered for the hypotensive patient in the setting of acute RV failure, most guidelines recommend a technique of a low volume trial of 100 to 500cc with very cautious clinical monitoring. 6-8
Bottom line:
We should be cautious when administering fluids to a hypotensive patient with a high likelihood of PE with signs of RV strain. It is reasonable to trial a small bolus of IV fluids. If HD improvement is not noticed after approximately 500cc of fluid, we should be cautious about giving more.
2. Supporting the Systemic Blood Pressure
Even relatively transient systemic hypotension is harmful to the RV, as it is particularly prone to ischemia when RV coronary perfusion is reduced. Vasoactive agents are the mainstay of treatment, as IVF is unlikely to improve hypotension in patients with acute RV failure. We should have a low threshold to start vasopressors to improve RV perfusion. The goal is to keep systemic pressure above RV systolic pressure to maintain RV perfusion. Practice guidelines recommend maintaining a MAP above 65 to 75mmHg 8-10.
Which vasopressor or inotrope should be used as first line? The ideal vasopressor agent in RV failure in acute PE would:
- Increase CO
- Maintain SVR
- Decrease pulmonary vascular resistance
- Not increase tachycardia or dysrhythmias
Unfortunately, no sole agent exists to achieve these goals. Additionally, there is sparse literature and no consensus on specifically which vasopressor(s)/inotrope(s) to initiate. There are, however, several consistencies across guidelines and reviews.
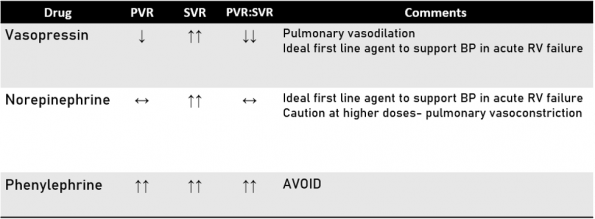
Vasopressin
Vasopressin is an intriguing option here, as it has the unique advantage of pulmonary vasodilation, which clinically means a reduction in PVR. It has been used effectively to support BP in hypotensive patients with acute PH after cardiac surgery. There is limited high quality data, however, on the use of vasopressin in patients with acute PE. The recommendations come mainly from animal models and cardiac surgery patients 11-12.
Norepinephrine
Norepinephrine is also recommended for hypotensive patients with acute RV failure, as it improves systemic vascular resistance and CO, despite potential increases in pulmonary vascular resistance at higher doses.
Studies of NE in RV dysfunction in animal models was shown to improve RV contractility and CO. A small study of patients with sepsis and RHF, NE use was associated with improved RV myocardial oxygen delivery 13-14.
Phenylephrine
Phenylephrine, on the other hand, should be entirely avoided in acute RV failure because of its activity as a potent alpha agonist, known to increase pulmonary vascular resistance 15.
Inotropes
And finally, inotropes such as milrinone and dobutamine are indicated to support RV contractility. Their benefit, however, can be offset by the risk of precipitating tachyarrhythmias and systemic hypotension. These agents should not routinely be started for these patients by us in the ED, and should be saved for use in the ICU 16.
Bottom Line
We should have a low threshold to start vasoactive agents. Most critical care reviews do recommend a combination of vasopressin and norepinephrine in the initial resuscitation of patients with RV failure from acute PE 15.
3. Decrease RV Afterload
Another key to managing RV strain from PE is to decrease pulmonary vascular resistance (PVR) and therefore the RV afterload.
Hypoxia and Hypercapnia
Hypoxia and hypercapnia can both increase pulmonary vascular resistance (PVR) and should be addressed early. High concentrations of oxygen can cause some pulmonary vasodilation and decrease PVR. Supplemental oxygen is best delivered for these patients via high flow. Positive pressure should be avoided as it can increase RV afterload.
Inhaled Pulmonary Vasodilators
Inhaled pulmonary vasodilators also have a role in treating patients with acute PE. They reduce RV afterload and improve oxygenation. Two of several inhaled pulmonary vasodilators used and reviewed in the literature are nitric oxide and epoprostenol. These agents may not be easily available to us in the emergency department, but they are often used in the ICU.
The use of epoprostenol in the acute setting has been described primarily in patients with RV after cardiac surgery 17.
Bottom Line
- Minimize or reverse hypoxia and hypercapnia
- High flow O2 should be considered early
- Consider inhaled pulmonary vasodilators if accessible
- In the case of an acute PE, decreasing clot burden and RV strain is also crucial for decreasing RV afterload. This will be explored in greater detail below.
4. Intubation
When do we make the decision to intubate patients with an acute PE?
Induction and intubation of a patient with RV failure from a PE is high risk and can lead to hemodynamic collapse and arrest. Intubation and subsequent positive pressure ventilation will decrease RV preload, increase RV afterload and lead to worsening RV failure and cardiac output. Intubation should be avoided entirely if possible. In one study by Rosenberger et al. in 2006 in patients undergoing pulmonary embolectomy in the operating room, hemodynamic collapse occurred in 20% of patients after GA induction 18. Intubation should really only be considered if the patient fails the above steps as listed above: increasing systemic BP, decreasing RV afterload, and an appropriate trial of high flow oxygen. In the ED, intubation should only be performed in an emergent, forced-to-act scenario.
A few key points to highlight if you are in a situation where you are forced to intubate a patient with acute RV failure from a PE:
- There is no universal approach to intubating a patient with the acute RV failure.
- These cases should be discussed with consultant such as anesthesia and ICU, for their support.
- Vasopressors should be running prior to induction targeting a high-normal blood pressure.
- Some reviews suggest placing the patient on BiPAP prior to intubation. This allows you to ease the patient on to PPV slowly and in a reversible fashion 19.
- RSI is not optimal for these patients, because any apneic period is dangerous. Some texts recommend delayed-sequence induction with gentle ventilation during the apneic period to avoid hypercapnia and hypoxia 19.
To make matters more challenging, intubating and securing the airway has not fixed the underlying problem of RV failure in these unstable patients. Ventilating these patients will be extremely difficult. The increase in intrathoracic pressure with ventilation after intubation will worsen RV hemodynamics. This is one case where, after intubation, we should stick in the room to monitor the patient closely. This is a critical time when they can fall into the RV spiral. It is recommended to avoid aggressive bagging, and to minimize pressures and tidal volumes to minimize hemodynamic changes.
PE Risk Stratification
Once a PE is diagnosed in the ED, risk stratification is necessary to identify patients at increased risk of adverse outcomes and to guide treatment decisions. PE is classified into three main categories: high-risk or massive, intermediate-risk or submassive, and low-risk. This classification involves:
- Clinical assessment (vital signs, signs, or symptoms of shock)
- Imaging (CT/ echocardiography).
- Laboratory indicators of PE severity (troponin).
This summary will focus only on high- and intermediate-risk PE.
A high-risk (massive) and intermediate-risk (submassive) PE are defined as below, according to AHA guidelines in 2011 10.
Notably, high-risk PE accounts for approximately 5% of hospitalized patients with PE and have an average mortality of approximately 30% within 1 month 20.
This classification exists because patients with high and intermediate risk PE are associated with adverse prognosis and increased mortality compared with low risk PE 8,10,21.
Treatment of Acute PE in the ED
Anticoagulation
The cornerstone for treatment of PE is anticoagulation. Consistent across recent guidelines, specifically Thrombosis Canada, the American Heart Association, the American College of Chest Physicians and the European Society of Cardiology, anticoagulation should be initiated early in the management of acute PE. With respect to empiric anti-coagulation, this depends on patient and clinical factors. In a hemodynamically stable patient, if the pre-test probability for a PE is high, and/or there is an expected delay to CT imaging, consider giving anticoagulation prior to CT-confirmation 22.
For patients with an acute PE, our options for the first dose of anticoagulation in the ED is LMWH or UFH. In view of our local thrombosis experts, it is advised to give anticoagulation early once an acute PE is confirmed. LMWH is generally preferred given its reliable pharmacokinetics. If, however, you are treating a patient and thrombolysis is being considered, UFH is preferred due to its short half-life and option for rapid reversal. It would not be wrong to give these patients LMWH, however, as long as ultimately there is no delay in this decision of anticoagulation 21.
Decreasing RV Afterload in Acute PE
Decreasing RV afterload is critical in the management of patients with high and intermediate-risk PE. Physical obstruction from a pulmonary embolus contributes to RV strain and increased PVR, which makes patients with high and intermediate-risk PE at an increased risk for deterioration. These patients therefore need a reduction of clot burden, and to be considered for more advanced therapies, beyond anti-coagulation alone.
Advanced therapies to reduce clot burden and PVR include 23:
- Systemic thrombolytics
- Full dose
- Reduced dose
- Catheter-directed thrombolytics
- Catheter embolectomy
- Surgical embolectomy
Treatment of High-Risk PE in the ED
Systemic Thrombolysis
Early studies of alteplase in PE showed that thrombolytic agents result in a greater and faster improvement dissolution of pulmonary thrombus with an associated rapid decrease in PA pressures compared with heparin 1.
In unstable patients with a high-risk PE, current guidelines (AHA, ACCP, ESC) 8,10,22 consistently recommend systemic thrombolytics as first-line therapy, in the absence of contraindications due to patient bleeding risk. The recommended alteplase (rt-PA) dose is 100mg over 2 hours.
There is practice variation in exactly how to give this dose of rt-PA, and there is nothing specifically in the guidelines about administration. In view of our local thrombosis physicians, the dosing strategy of 10mg push followed by an infusion of 90mg over 2 hours is recommended.
Systemic thrombolytics, however, is a double-edge sword. It likely has a reduction in mortality for massive PE (Zuo et al. 2015 24), but it confers a risk of major hemorrhage to all patients. The challenge is to weigh the risk of mortality from PE with the risk from major hemorrhage.
It is important to note that, when reviewing the literature on thrombolysis in PE, the evidence can be challenging to interpret for several reasons, including: the heterogenous groups of patients with a range of PE severity and age, the small number of included patients and the variation of thrombolytic drugs used and the doses.
A meta-analysis by Zuo et al. was done in 2015 assessing the effect of systemic thrombolysis for high and intermediate risk PE. It included 21 RCTs and 2401 participants. The study demonstrated a reduction in mortality (OR: 0.58, 95% confidence interval [CI]: 0.38-0.88, 19 studies, 2319 participants, low-certainty evidence) when systemic thrombolysis was used 24. It should be noted, however, that these effects on mortality are notably weakened when six studies at high risk of bias were excluded from analysis. Also, major hemorrhagic events were more common in the thrombolytics group than in the control group (OR 2.84, 95% CI 1.92 to 4.20; 15 studies, 2101 participants; moderate-certainty evidence) 24.
For patients with high-risk PE with a low bleeding risk: the ACCP recommends administering systemic thrombolytics (100mg/2h), as this is likely to reduce mortality from PE more than it would increase fatal bleeding (Grade 2C recommendation). At this time, research and guidelines suggest the benefit of thrombolytics outweighs the risk of hemorrhage 22.
Patients with a High-Bleeding Risk
What about patients with a high risk of bleeding? The risk to benefit ratio of thrombolytics is less clear in these cases. Reduced-dose thrombolysis has been suggested here to mitigate the risks of major hemorrhage.
Small studies, such as that done by Wang et al. in 2010, have compared half (50mg over 2h) and full-dose (100mg over 2h) systemic thrombolytics. This study included 118 patients with hemodynamically or anatomically massive PE. It showed similar improvements in surrogate end points including RV dysfunction between the two groups, with a decrease in major and fatal bleeding in the reduced dose group 25. Major bleeding occurred in 10% of patients in the full dose group and 3% in the half dose group, with one fatal bleeding episode of intracranial hemorrhage (ICH) in the full-dose group. Evidence overall for reduced-dose thrombolytics is lacking, however 25.
For patients with high-risk PE with a high bleeding risk: reduced-dose systemic thrombolytics may be considered in select patients with massive PE who have relative contraindications to thrombolytics. This decision needs to be made in consultation with the Thrombosis physician on service 22.
Bottom Line: Management of the High-Risk PE
In the ED we need to assess for contraindications for thrombolytic therapy. In patients with a low risk of bleeding, give a full dose of tPA 100mg over 2h. In patients with a high risk of bleeding, consider reduced dose systemic thrombolytics in consultation with the Thrombosis physician/ service at your center.
Treatment of Intermediate-Risk PE in the ED
Patients with an intermediate-risk PE are not imminently unstable or hypotensive. So what is the concern with the intermediate-risk PE? This group is a heterogenous patient population; they are clinically not all the same. Some of the patients are quite stable, while others are closer to borderline hypotensive and are at higher risk for deterioration. The latter group, therefore, requires specialized treatment and monitoring.
Systemic Thrombolysis
This diverse group has generated the most debate regarding optimal management. Thrombolytic therapy for patients with an intermediate-risk PE is controversial from a risk-benefit perspective, and the current evidence is fairly murky.
Many studies, more recently, have looked at the role of thrombolytics in the intermediate-risk PE patients. The largest RCT to date using full-dose thrombolysis in submassive PE was the PEITHO trial by Meyer et al. in 2014. In this study, systemic thrombolytics was shown to reduce HD deterioration and rescue thrombolysis 26. There was, however, an increased rate of major hemorrhage in the systemic thrombolytics group compared to the control group (heparin alone) (11.5% versus 2.4%, RR: 5.55, 95% CI: 2.30-13.39), and a 2.0% rate of ICH compared to 0.2% in the placebo group 26. Overall, there was no difference in mortality.
Given that the clinical presentation and progression of submassive PE patients can be quite varied, there may be some patients who benefit from systemic thrombolysis, and many who won’t. Overall, there is no clear consensus in guidelines for thrombolytic therapy 10,22,27.
This may change, however, as there are studies emerging for alternative therapies that may offer treatment benefit with an improved safety profile.
Alternative Therapies
These alternative therapies include:
- Reduced-dose systemic thrombolytics (again)
- Catheter-directed thrombolysis
- And new therapies such as catheter embolectomy
The advantages of these approaches is that they 1) may be used in patients with high bleeding risk, 2) they offer rapid improvement, and 3) they are less invasive 23.
Reduced-dose thrombolytics:
As discussed above, the evidence for reduced-dose thrombolytics is quite limited and further studies in this area are needed. Notably, a randomized trial by Sanchez et al. comparing the safety and efficacy of reduced dose alteplase with heparin in patients with intermediate risk PE is currently underway (PEITHO-3) 28.
Catheter-directed thrombolysis:
This represents another approach, that has been suggested to lower bleeding risk of select patients. This involves positioning a catheter directly into a clot in the pulmonary arteries and infusing low doses of a thrombolytic drug. A large randomized RCT by Klok et al. looking at clinical outcomes in catheter-directed therapies is currently underway (HI-PEITHO) 29.
Catheter embolectomy:
This is an emerging therapy where a large bore cannula is placed into the pulmonary artery to aspirate embolic material with manual suction. The advantage of catheter embolectomy is that it may be used in patients with high bleeding risk or in those with contraindications to systemic anticoagulation, and it may offer rapid improvement. This is available at certain centres that have the appropriate equipment.
Take Home: Management of the Intermediate-Risk PE
The intermediate-risk PE group is truly a diverse population in which optimal management is uncertain and variable. Emerging therapies will likely change our understanding of the ideal management in this patient population.
Pulmonary Embolism Response Team (PERT)
Treatment decisions for the intermediate-risk PE is clearly complex. Thrombosis is accessible at many centres for consultation and expert advice. This service is integral in response to the now increasing therapeutic options and complex decisions for high and intermediate risk patients. In addition, the PERT concept has evolved. PERTs are multidisciplinary teams that specialize in rapid PE assessment and treatment. The PERT concept was initially established in 2012 and has been increasingly adopted worldwide. The PERT Consortium created a practice document by Rivera-Lebron et al. in 2019 outlining PE management 30. Although it may vary by the centre, PERT often includes thrombosis, ICU and interventional radiology.
In our local centre, PERT is activated when specific criteria are met (confirmed PE with significant proximal clot burden, RV strain, abnormal troponin value, and any additional finding such as syncope, pregnancy, altered level of consciousness, and others).
When activated, the patient may be taken to the ICU. Here, the patient can be closely monitored, and the full range of medical and interventional therapies can be discussed by the multidisciplinary team.
Despite a need for more robust prospective studies on PERT outcomes, these teams have been endorsed by the ESC. One retrospective analysis by Myc et al. was performed in 2020 on patients with acute PE at the University of Virginia. 317 patients treated for a PE following institution of the PERT program were compared to 237 patients in a pre-PERT era. Patients in the control group demonstrated worse survival when compared with patients treated during the PERT era (survival 89.6% vs. 84.8% at 30d, 81.4 vs 75.9% at 180d) 31. 30-day readmission rates were significantly lower in the PERT era (9.1% versus 2%) 31. There was no significant difference in bleeding complications.
In view of our local intensivists, in a patient who meets PERT criteria, there should be no delay in giving anticoagulation (LMWH or UFH), regardless of the potential for additional advanced therapies.
Take Home: Role in the ED
Our role in the ED for managing patients with massive and submassive PE, in addition to an RV-centric resuscitation, is to:
- Recognize patients with a PE and risk stratify them
- Assess for contraindications for thrombolysis
- Administer thrombolytics in a high-risk unstable patient
- Discuss the case with Thrombosis or activate PERT if the patient meets the criteria
- Do not delay giving anticoagulation
References
- Goldhaber SZ, Visani L, De Rosa M. Acute pulmonary embolism: clinical outcomes in the International Cooperative Pulmonary Embolism Registry (ICOPER). Lancet. 1999;353(9162): 1386-1389. doi:10.1016/S0140-6736(98)07534-5.
- Casazza F, Becattini C, Bongarzoni A, et al. Clinical features and short term outcomes of patients with acute pulmonary embolism. The Italian Pulmonary Embolism Registry (IPER). Thromb Res 2012;130:847–52
- Stein PD, Henry JW. Prevalence of acute pulmonary embolism among patients in a general hospital and at autopsy. Chest. 1995; 108(4):978-981. doi:10.1378/chest.108.4.978.
- Crager, S. Right Heart Failure with Sara Crager, EMCrit 272. https://www.tomwademd.net/links-to-emcrit-272-right-heart-failure-with-dr-sara-crager/
- Ghignone et al. Volume Expansion versus Norepinephrine in Treatment of a Low Cardiac Output Complicating an Acute Increase in Right Ventricular Afterload in Dogs. 1984. Anesthesiology. Vol 60: 132-135.
- Hrymak C, Strumpher J, Jacobsohn E. Acute Right Ventricle Failure in the Intensive Care Unit: Assessment and Management. Can J Cardiol. 2017 Jan;33(1):61-71. doi: 10.1016/j.cjca.2016.10.030. Epub 2016 Nov 11. PMID: 28024557.
- Olsson KM, Halank M, Egenlauf B, Fistera D, Gall H, Kaehler C, Kortmann K, Kramm T, Lichtblau M, Marra AM, Nagel C, Sablotzki A, Seyfarth HJ, Schranz D, Ulrich S, Hoeper MM, Lange TJ. Decompensated right heart failure, intensive care and perioperative management in patients with pulmonary hypertension: Updated recommendations from the Cologne Consensus Conference 2018. Int J Cardiol. 2018 Dec 1;272S:46-52. doi: 10.1016/j.ijcard.2018.08.081. Epub 2018 Aug 26. PMID: 30190155.
- Konstantinides SV, Meyer G, Becattini C, Bueno H, Geersing GJ, Harjola VP, Huisman MV, Humbert M, Jennings CS, Jiménez D, Kucher N, Lang IM, Lankeit M, Lorusso R, Mazzolai L, Meneveau N, Ní Áinle F, Prandoni P, Pruszczyk P, Righini M, Torbicki A, Van Belle E, Zamorano JL; ESC Scientific Document Group. 2019 ESC Guidelines for the diagnosis and management of acute pulmonary embolism developed in collaboration with the European Respiratory Society (ERS). Eur Heart J. 2020 Jan 21;41(4):543-603. doi: 10.1093/eurheartj/ehz405. PMID: 31504429.
- Stevens SM, Woller SC, Kreuziger LB, Bounameaux H, Doerschug K, Geersing GJ, Huisman MV, Kearon C, King CS, Knighton AJ, Lake E, Murin S, Vintch JRE, Wells PS, Moores LK. Antithrombotic Therapy for VTE Disease: Second Update of the CHEST Guideline and Expert Panel Report. Chest. 2021 Dec;160(6):e545-e608. doi: 10.1016/j.chest.2021.07.055. Epub 2021 Aug 2. Erratum in: Chest. 2022 Jul;162(1):269. PMID: 34352278.
- Jaff MR, McMurtry MS, Archer SL, Cushman M, Goldenberg N, Goldhaber SZ, Jenkins JS, Kline JA, Michaels AD, Thistlethwaite P, Vedantham S, White RJ, Zierler BK; American Heart Association Council on Cardiopulmonary, Critical Care, Perioperative and Resuscitation; American Heart Association Council on Peripheral Vascular Disease; American Heart Association Council on Arteriosclerosis, Thrombosis and Vascular Biology. Management of massive and submassive pulmonary embolism, iliofemoral deep vein thrombosis, and chronic thromboembolic pulmonary hypertension: a scientific statement from the American Heart Association. Circulation. 2011 Apr 26;123(16):1788-830. doi: 10.1161/CIR.0b013e318214914f. Epub 2011 Mar 21. Erratum in: Circulation. 2012 Aug 14;126(7):e104. Erratum in: Circulation. 2012 Mar 20;125(11):e495. PMID: 21422387.
- Eichinger MR, Walker BR: Enhanced pulmonary arterial dilation to arginine vasopressin in chronically hypoxic rats. Am J Physiol 1994, 267: H2413-H2419.
- Tayama E, Ueda T, Shojima T, Akasu K, Oda T, Fukunaga S, Akashi H, Aoyagi S: Arginine vasopressin is an ideal drug after cardiac surgery for the management of low systemic vascular resistant hypotension concomitant with pulmonary hypertension. Interact Cardiovasc Thorac Surg 2007, 6:715-719.
- Schreuder WO, Schneider AJ, Groeneveld AB, Thijs LG. Effect of dopamine vs norepinephrine on hemodynamics in septic shock: emphasis on right ventricular performance. Chest 1989;95: 1282–1288.
- Martin C, Perrin G, Saux P, Papazian L, Gouin F: function Effects of norepinephrine on right ventricular in septic shock patients. Intensive Care Med 1994, 20:444-447.
- Price LC, Wort SJ, Finney SJ, et al. Pulmonary vascular and right ventricular dysfunction in adult critical care: current and emerging options for management: a systematic literature review. Crit Care 2010; 14:R169.
- Kerbaul F, Rondelet B, Motte S, Fesler P, Hubloue I, Ewalenko P, Naeije R, Brimioulle S. Effects of norepinephrine and dobutamine on pressure load-induced right ventricular failure. Crit Care Med 2004;32:1035–1040.
- Muzaffar S, Shukla N, Angelini GD, Jeremy JY. Inhaled prostacyclin is safe, effective, and affordable in patients with pulmonary hypertension, right-heart dysfunction, and refractory hypoxemia after cardiothoracic surgery. J Thorac Cardiovasc Surg 2004;128: 949–950
- Rosenberger P, Shernan SK, Shekar PS, Tuli JK, Weissmüller T, Aranki SF, Eltzschig HK. Acute hemodynamic collapse after induction of general anesthesia for emergent pulmonary embolectomy. Anesth Analg. 2006 May;102(5):1311-5.
- Wanner PM, Filipovic M. The Right Ventricle-You May Forget it, but It Will Not Forget You. J Clin Med. 2020 Feb 5;9(2):432. doi: 10.3390/jcm9020432. PMID: 32033368; PMCID: PMC7074056.
- Vanni S, Nazerian P, Pepe G, Baioni M, Risso M, Grifoni G, Viviani G, Grifoni S. Comparison of two prognostic models for acute pulmonary embolism: clinical vs. right ventricular dysfunction-guided approach. J ThrombHaemost. 2011;9:1916–1923. doi: 10.1111/j.1538-7836.2011.04459.x
- Thrombosis Canada. Pulmonary Embolism (PE) Treatment. https://thrombosiscanada.ca/clinicalguides/#
- Kearon C, Akl EA, Ornelas J, Blaivas A, Jimenez D, Bounameaux H, Huisman M, King CS, Morris TA, Sood N, Stevens SM, Vintch JRE, Wells P, Woller SC, Moores L. Antithrombotic Therapy for VTE Disease: CHEST Guideline and Expert Panel Report. Chest. 2016 Feb;149(2):315-352. doi: 10.1016/j.chest.2015.11.026. Epub 2016 Jan 7. Erratum in: Chest. 2016 Oct;150(4):988. PMID: 26867832.
- Giri J, Sista AK, Weinberg I, Kearon C, Kumbhani DJ, Desai ND, Piazza G, Gladwin MT, Chatterjee S, Kobayashi T, Kabrhel C, Barnes GD. Interventional Therapies for Acute Pulmonary Embolism: Current Status and Principles for the Development of Novel Evidence: A Scientific Statement From the American Heart Association. Circulation. 2019 Nov 12;140(20):e774-e801. doi: 10.1161/CIR.0000000000000707. Epub 2019 Oct 4. PMID: 31585051.
- Zuo Z, Yue J, Dong BR, Wu T, Liu GJ, Hao Q. Thrombolytic therapy for pulmonary embolism. Cochrane Database Syst Rev. 2021 Apr 15;4(4):CD004437.
- Wang C, Zhai Z, Yang Y, et al. Efficacy and safety of low dose recombinant tissue-type plasminogen activator for the treatment of acute pulmonary thromboembolism: a randomized, multicenter, controlled trial. Chest. 2010;137(2):254-262.
- Meyer G, Vicaut E, Danays T, Agnelli G, Becattini C, Beyer-Westendorf J, Bluhmki E, Bouvaist H, Brenner B, Couturaud F, Dellas C, Empen K, Franca A, Galiè N, Geibel A, Goldhaber SZ, Jimenez D, Kozak M, Kupatt C, Kucher N, Lang IM, Lankeit M, Meneveau N, Pacouret G, Palazzini M, Petris A, Pruszczyk P, Rugolotto M, Salvi A, Schellong S, Sebbane M, Sobkowicz B, Stefanovic BS, Thiele H, Torbicki A, Verschuren F, Konstantinides SV; PEITHO Investigators. Fibrinolysis for patients with intermediate-risk pulmonary embolism. N Engl J Med. 2014 Apr 10;370(15):1402-11
- Konstantinides SV, Torbicki A, Agnelli G, Danchin N, Fitzmaurice D, Galiè N, Gibbs JS, Huisman MV, Humbert M, Kucher N, Lang I, Lankeit M, Lekakis J, Maack C, Mayer E, Meneveau N, Perrier A, Pruszczyk P, Rasmussen LH, Schindler TH, Svitil P, Vonk Noordegraaf A, Zamorano JL, Zompatori M; Task Force for the Diagnosis and Management of Acute Pulmonary Embolism of the European Society of Cardiology (ESC). 2014 ESC guidelines on the diagnosis and management of acute pulmonary embolism. Eur Heart J. 2014 Nov 14;35(43):3033-69, 3069a-3069k. doi: 10.1093/eurheartj/ehu283. Epub 2014 Aug 29. Erratum in: Eur Heart J. 2015 Oct 14;36(39):2666. Erratum in: Eur Heart J. 2015 Oct 14;36(39):2642. PMID: 25173341.
- Sanchez O, Charles-Nelson A, Ageno W, Barco S, Binder H, Chatellier G, Duerschmied D, Empen K, Ferreira M, Girard P, Huisman MV, Jiménez D, Katsahian S, Kozak M, Lankeit M, Meneveau N, Pruszczyk P, Petris A, Righini M, Rosenkranz S, Schellong S, Stefanovic B, Verhamme P, de Wit K, Vicaut E, Zirlik A, Konstantinides SV, Meyer G; PEITHO-3 Investigators. Reduced-Dose Intravenous Thrombolysis for Acute Intermediate-High-risk Pulmonary Embolism: Rationale and Design of the Pulmonary Embolism International THrOmbolysis (PEITHO)-3 trial. Thromb Haemost. 2022 May;122(5):857-866.
- Klok FA, Piazza G, Sharp ASP, Ní Ainle F, Jaff MR, Chauhan N, Patel B, Barco S, Goldhaber SZ, Kucher N, Lang IM, Schmidtmann I, Sterling KM, Becker D, Martin N, Rosenfield K, Konstantinides SV. Ultrasound-facilitated, catheter-directed thrombolysis vs anticoagulation alone for acute intermediate-high-risk pulmonary embolism: Rationale and design of the HI-PEITHO study. Am Heart J. 2022 Sep;251:43-53
- Rivera-Lebron B, McDaniel M, Ahrar K, Alrifai A, Dudzinski DM, Fanola C, Blais D, Janicke D, Melamed R, Mohrien K, Rozycki E, Ross CB, Klein AJ, Rali P, Teman NR, Yarboro L, Ichinose E, Sharma AM, Bartos JA, Elder M, Keeling B, Palevsky H, Naydenov S, Sen P, Amoroso N, Rodriguez-Lopez JM, Davis GA, Rosovsky R, Rosenfield K, Kabrhel C, Horowitz J, Giri JS, Tapson V, Channick R; PERT Consortium. Diagnosis, Treatment and Follow Up of Acute Pulmonary Embolism: Consensus Practice from the PERT Consortium. Clin Appl Thromb Hemost. 2019 Jan-Dec;25:1076029619853037.
- Myc LA, Solanki JN, Barros AJ, Nuradin N, Nevulis MG, Earasi K, Richardson ED, Tsutsui SC, Enfield KB, Teman NR, Haskal ZJ, Mazimba S, Kennedy JLW, Mihalek AD, Sharma AM, Kadl A. Adoption of a dedicated multidisciplinary team is associated with improved survival in acute pulmonary embolism. Respir Res. 2020 Jun 22;21(1):159.
- Smith SB, Geske JB, Maguire JM, et al. Early anticoagulation is associated with reduced mortality for acute pulmonary embolism. Chest 2010;137:1382–90.
- Dalla-Volta S, Palla A, Santolicandro A, Giuntini C, Pengo V, Visioli O, Zonzin P, Zanuttini D, Barbaresi F, Agnelli G, et al. PAIMS 2: alteplase combined with heparin versus heparin in the treatment of acute pulmonary embolism. Plasminogen activator Italian multicenter study 2. J Am Coll Cardiol. 1992 Sep;20(3):520-6.
- Jerjes-Sanchez C, Ramirez-Rivera A, de Lourdes Garcia M, et al. Streptokinase and heparin versus heparin alone in massive pulmonary embolism: a randomized controlled trial. J Thromb Thrombolysis. 1995; 2: 227–229.
- Piazza G. Advanced Management of Intermediate- and High-Risk Pulmonary Embolism: JACC Focus Seminar. J Am Coll Cardiol. 2020 Nov 3;76(18):2117-2127. doi: 10.1016/j.jacc.2020.05.028. PMID: 33121720.
- Hountras P, Bull TM. Advanced therapies for pulmonary embolism. Curr Opin Pulm Med. 2020 Sep;26(5):397-405. doi: 10.1097/MCP.0000000000000714. PMID: 32740381.
- Reardon PM, Yadav K, Hendin A, Karovitch A, Hickey M. Contemporary Management of the High-Risk Pulmonary Embolism: The Clot Thickens. J Intensive Care Med. 2019 Aug;34(8):603-608. doi: 10.1177/0885066618789879. Epub 2018 Aug 1. PMID: 30068253.
- Trott T, Bowman J. Diagnosis and Management of Pulmonary Embolism. Emerg Med Clin North Am. 2022 Aug;40(3):565-581. doi: 10.1016/j.emc.2022.05.008. Epub 2022 Jul 8. PMID: 35953217.
- Vieillard-Baron A, Naeije R, Haddad F, Bogaard HJ, Bull TM, Fletcher N, Lahm T, Magder S, Orde S, Schmidt G, Pinsky MR. Diagnostic workup, etiologies and management of acute right ventricle failure : A state-of-the-art paper. Intensive Care Med. 2018 Jun;44(6):774-790. doi: 10.1007/s00134-018-5172-2. Epub 2018 May 9. PMID: 29744563.
- Tapson VF, Weinberg AS. Overview of Management of Intermediate- and High-Risk Pulmonary Embolism. Crit Care Clin. 2020 Jul;36(3):449-463. doi: 10.1016/j.ccc.2020.02.003. Epub 2020 May 7. PMID: 32473691.
- Kostura M, Smalley C, Koyfman A, Long B. Right heart failure: A narrative review for emergency clinicians. Am J Emerg Med. 2022 Aug;58:106-113. doi: 10.1016/j.ajem.2022.05.030. Epub 2022 May 25. PMID: 35660367.
- Kaplovitch E, Shaw JR, Douketis J. Thrombolysis in Pulmonary Embolism: An Evidence-Based Approach to Treating Life-Threatening Pulmonary Emboli. Crit Care Clin. 2020 Jul;36(3):465-480. doi: 10.1016/j.ccc.2020.02.004. PMID: 32473692.








Trackbacks/Pingbacks