On first glance, bite management may seem like a seemingly simple presentation in the ED. However, in today’s literature, there remains a lot of controversy regarding bite management. Who do you give antibiotics to? Can you suture the wound? When should you worry about rabies? Without a good understanding of proper management and follow up care, for some of these cases it can lead to significant morbidity and even mortality if not dealt with properly.
The goal of this post is to help you navigate some of the controversies and provide you with an evidence-based approach that you can use the next time you see a bite wound in the ED.
In part 1 of this 2 part series we will tackle animal bites, part 2 will focus on human bites and considerations around rabies.
Animal Bites
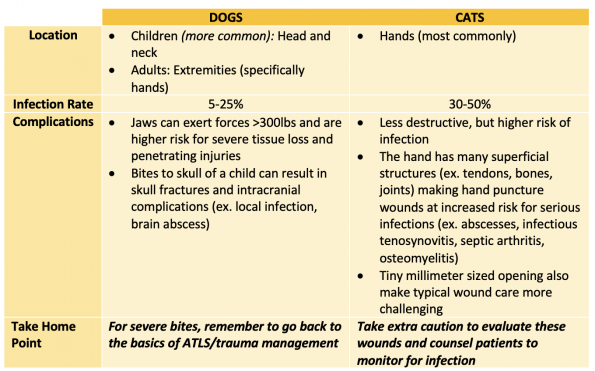
Bite related injuries represent 1% of ED visits each year, with the most common bites being from dogs (90%) and cats (10%). While dog bites can cause more damage from shear force, cat bites pose a unique risk of infection because of their needle like teeth that can inoculate deeper structures.
Microbiology
When looking at the microbiology of any bite wound, we need to consider both the normal skin flora of the bitten and the oral flora of the biter. Bite wounds are typically of mixed aerobic and anerobic flora.
Common isolates from bite wounds:
- Pasteurella species, isolated from 50% of dog bites and 75% of cat bites
- Associated with more rapid development of infection (even within 12hrs)
- Important note: Some of our common antibiotics for skin infections (ex. cephalexin) have poor activity against Pasteurella. So make sure to adjust your antibiotic treatments to cover this bug!
- Capnocytphagia canimorsus, particularly with dog bites
- Relatively low incidence in immunocompetent healthy patients, but it is especially important to consider in patients with asplenia, underlying hepatic disease, alcoholism or are immunocompromised.
- Case reports have shown that it can progress rapidly to severe sepsis and death in only 24hs after the initial signs of infection, with a quoted mortality rate of up to 25-30%.
- Unfortunately, with a relatively short incubation time but a long growth time in the lab (mean culture time of 6 days), it can be very challenging to diagnose.
Clinical Pearl: For bites that present early without clinical infection, we should NOT be routinely swabbing them for culture. Only consider culturing if there is clear infection present on your assessment.
Wound Management
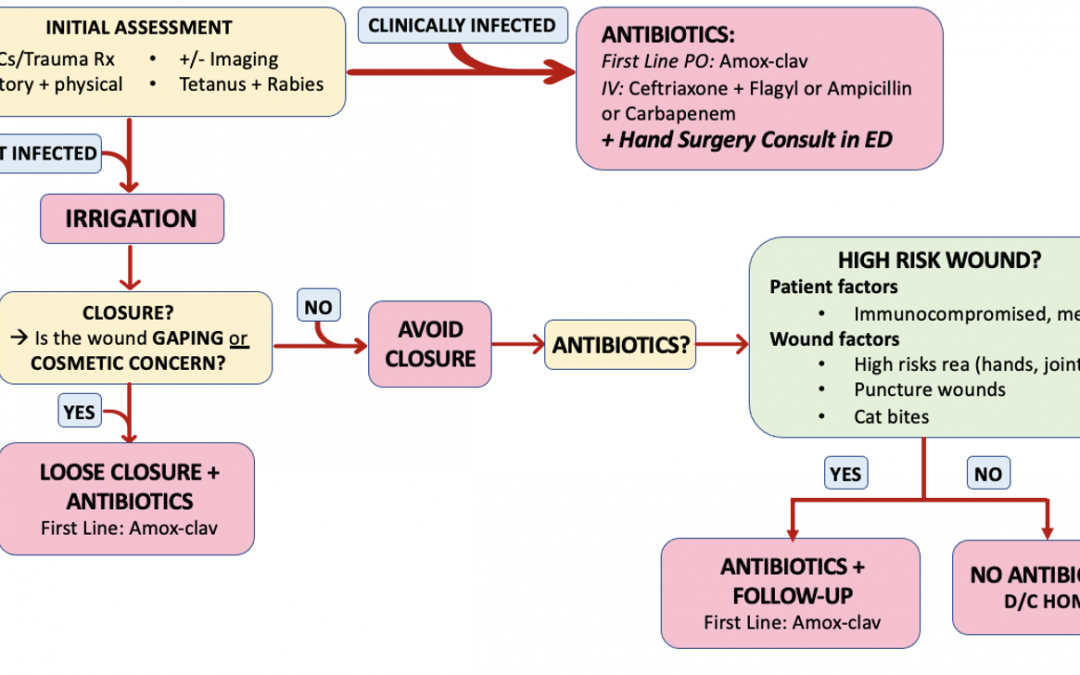
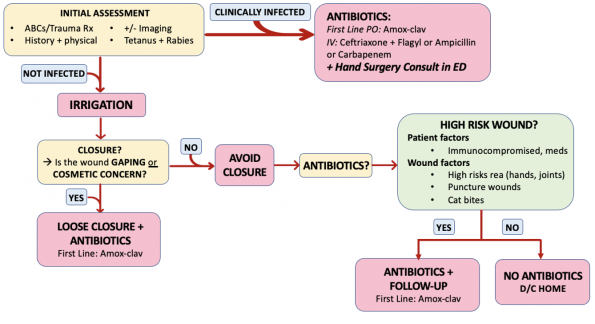
TAKE HOME POINT #1: Copious irrigation is key and one of the most important aspects of ED care for bites.
Irrigation is a mainstay of ED management, and is likely overlooked. For dirtier and more contaminated bite wounds, large volume (500-1000mL) and pressure irrigation are key.
Irrigation Options:
Option 1: Use the Tap
- For wounds that can be easily placed in the sink, starting at the tap and running the wound under water for 15 minutes is a great option.
- There was a Cochrane Study in 2012 that looked at the use of tap water to clean acute wounds. It included 11 trials and found that the use of tap water to clean acute wounds was not associated with statistically significant difference in infection compared to saline, and that in the presence of clean potable tap water it is a cost effective alternative.
Option 2: Use normal saline
- Multiple different options exist for irrigating with saline:
- Use the 500cc saline bottles and pour it over the wound or try making holes at the top of the bottle using a blunt needle to create a more controlled irrigation.
- Use saline bags to irrigate. To irrigate large wounds, try using the 3L bag of saline with cystoscopy tubing to provide a higher flow of irrigation.
- Clinical Take Away: With any of these irrigation methods, the key is to wash with large volumes and ensure you are irrigating the entirety of the wound. After a large volume wash out, ensure you use higher pressure irrigation to ensure the deeper aspects of the wound are flushed out (ex. >20mL syringe with 18 gauge angio cath or irrigation guard).
But what about antiseptic solutions for irrigation?
- You can consider using diluted solutions of antiseptic (ex. 1% iodine mixture) for grossly contaminated wounds, although there is a lack of evidence. Interestingly, both the CDC and public health recommend using a diluted iodine solution for irrigation wounds that are high risk of rabies as the iodine is considered to be virucidal.
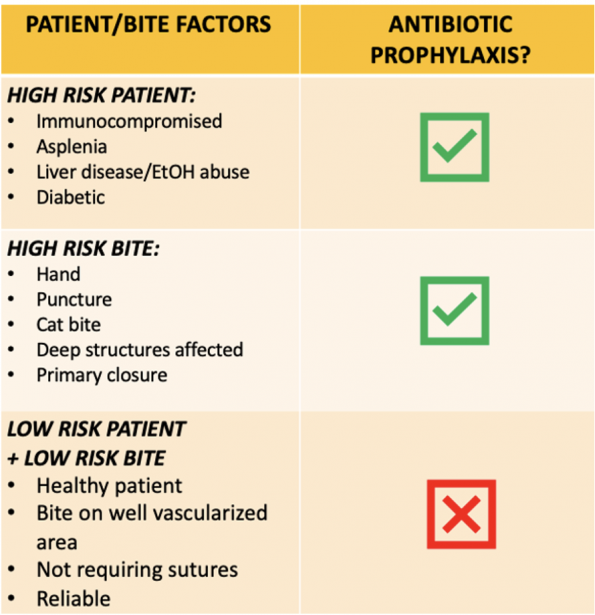
TAKE HOME POINT #2: Prophylactic antibiotics are recommended for high risk patients or high risk bites as summarized below.
Evidence Review
“Antibiotic prophylaxis for mammalian bites.” Cochrane Database of Systematic Reviews 2 (2001).
- This is likely one of the cornerstones in the available prophylactic antibiotic literature
- Included 8 RCTs that assessed uninfected mammalian bites presenting to hospital within 24hrs of injury, with the majority of studies looking at dog bites.
- Conclusion: Prophylactic antibiotics did not appear to reduce the rate of infection after bites from dogs or cats. However, there was limited evidence that antibiotics reduced the rate of infection specifically with bites to the hand.
- Caveats:
- Studies included had small sample sizes, significant heterogeneity and were generally of poor methodological quality with high rates of loss to follow up.
- Another challenge in the interpretation of the results is around the choice of antibiotics prescribed. The antibiotics used in many studies included cephalexin and erythromycin amongst others, which we know doesn’t cover Pasteurella well. Given the fact that inappropriate antibiotics were used these trials, it may shed light onto why they did not see more of a treatment effect in this systematic review.
“Evaluation of prophylactic oxacillin in cat bite wounds.” Annals of emergency medicine 13.3 (1984): 155-157.
- A prospective, double blind, placebo-controlled study and the only study included in the Cochrane review that looked at cat bites specifically.
- Assessed the use of prophylactic oxacillin vs. placebo for cat bites that were uninfected full thickness wounds presenting to the ED within 24hrs of injury.
- The study group only consisted of 11 patients. None of the patients receiving prophylactic antibiotics developed infection while Infections occurred in 4/6 pts receiving placebo.
- Of those infected, 3/4 were due to Pasteurella multocida infections and were puncture wounds.
- Conclusion: Antibiotic prophylaxis showed benefit in cat bite wounds.
- However, similar to the other studies, the sample size was small with only 11 patients
Clinical Pearl: Remember to screen for high risk patient and bite factors when assessing patients with bites and consider giving antibiotics for these patients.
- Most of the studies looking at the use of prophylactic antibiotics look at low risk patients, meaning patients with no comorbidities that would pre-dispose them to infection and bites that have a lower risk of infection. In these patients prophylactic antibiotics doesn’t show to have much benefit.
- There are no high quality RCTs that specifically assess the use of antibiotics in high risk patients or high risk bite wounds. So, this is largely where clinically judgement should come into play.
- High risk patients and high risk bites, given their increased likelihood to develop infections should receive prophylactic antibiotics.
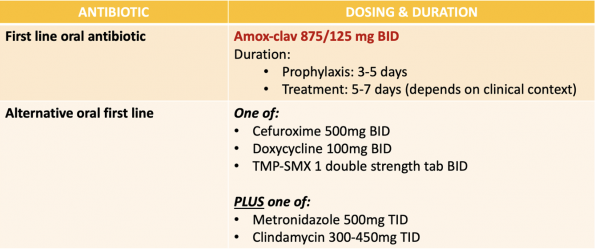
Antibiotic Choices
When Should I consider IV Antibiotics?
- There isn’t much great evidence regarding the role of IV vs. oral antibiotics specifically for bite wounds. Again, this largely comes down to clinical judgement.
- I discussed this with Dr. Yadav, one of our local ED experts in soft tissue infections, and he thought that almost all patients who require antibiotics should receive oral therapy and we should generally abandon the practice of a single IV dose of antibiotics followed by oral prescription. For patients who have clear evidence of sepsis, vomiting or if there is an element of malabsorption where oral therapy would be suboptimal, consider IV antibiotics in these cases.
Take Home Point #3: Suture the wound if it is key to cosmetic result or if the wound is large and gaping (but leave loose if you are closing). If you are closing the wound, give prophylactic antibiotics.
Evidence Review
”Primary closure versus delayed or no closure for traumatic wounds due to mammalian bite.” Cochrane Database of Systematic Reviews 12 (2019).
- Assessed primary vs. delayed/no closure in traumatic wounds from mammalian bites
- Included 4RCTs looking at dog bites only that were uninfected on presentation and did not damage deeper structures.
- Conclusion: Based on these RCTs there were “no high certainty evidence to support or refute existing recommendations”.
- Unfortunately, many of the studies included had a high risk of bias and were considered to be of very low quality.
“Primary closure versus non-closure of dog bite wounds. A randomised controlled trial.” Injury 45.1 (2014): 237-240.
- Assessed primary vs. non-closure or healing by secondary intent of dog bite wounds.
- Included 168 consecutive patient with dog bite injuries. All wounds received standard wound care and antibiotic treatment with amox-clav for 5 days.
- Conclusion: Primary closure of dog bite wounds resulted in no significant increase in infection rate and improved cosmetic appearance.
- Caveats: This study had a small sample size and also important to recognize that all patients did receive antibiotics and there no high risk wounds or patients included.
“Emergency treatment on facial laceration of dog bite wounds with immediate primary closure: a prospective randomized trial study.” BMC emergency medicine. Vol. 13. BioMed Central, 2013.
- Assessed 600 cases of dog facial lacerations that were randomly assigned to be left open vs. immediate primary closure.
- Prophylactic antibiotics were not given, and only given after wound infection occurred.
- Infection rates for open vs. primary closure was 8.3% and 6.3% respectively with a significant difference in healing time, with the primary closure group having faster healing times (6.57day vs. 9.12 days).
- Conclusion: Facial lacerations from dog bites should be primary closed after irrigation and debridement without increasing the rate of infections and with shortened healing times.
Given the conflicting evidence that exists around bite wound management and the lack of robust studies, let’s turn to some recent guidelines and expert opinions for further advice.
Recent Guideline Summaries
- Infectious Diseases Society of America (IDSA) Guidelines for skin and soft tissue infections, 2014
- Give prophylactic antibiotics for high risk patients:
- High risk patients factors:
- Immunocompromised
- Asplenic
- Advanced liver disease
- High risk bite qualities:
- Moderate-severe injuries
- Bites to the hand or face
- Bites that penetrated deeper structures.
- Primary wound closure NOT recommended for wounds, except for wounds to the face and if other wounds required closure just approximate them.
- High risk patients factors:
- National Institute for Health and Care Excellence, Guidelines for bite injuries (2020)
- They separated their recommendations into animal specific bites.
- As long as a cat bite has broken the skin, they recommend considering antibiotics.
- For dog bites they only recommend giving antibiotics if skin is broken and blood drawn
- Similar to the IDSA guidelines, in addition to these factors, for dog bites, they recommend giving antibiotics for high risk bites and high risk patients.
- Give prophylactic antibiotics for high risk patients:
Let’s talk to the experts:
I talked to Dr. Brent Trull, one of the hand surgeons in the department of plastic surgery at TOH, and Dr. Kevin Cheung one of the pediatric plastic surgeons at CHEO to get some insight into what they recommend for bite wounds. Below is a summary of their recommendations.
- Closure of animal bites largely depends on the location and balancing good tissue approximation and scar cosmesis with risk infection.
- For the face, you can generally accept the risks of infection given the functional and cosmetic implications.
- For other areas, such as the hand, it is more of a case-by-case basis.
- Tacking a wound closed to reduce the wound size but leaving enough open for fluid to egress is generally a reasonable approach. For example, close the wound loosely (1 suture every 2-3cm).
- Generally do not recommend doing delayed closure and avoid using glue or running sutures as they would seal a potentially dirty wound too tightly.
- Follow up:
- Plastic surgery generally does not need to see routine animal bites
- Consider referring for large degloving injuries or injuries where there is a concern for deeper structure involvement
- ED consult: If there are concerns on presentation for infection (ex. flexor tenosynovitis)
References
- Aziz, Hassan, et al. “The current concepts in management of animal (dog, cat, snake, scorpion) and human bite wounds.” Journal of Trauma and Acute Care Surgery 78.3 (2015): 641-648.
- Ball, Vincent, and Bradley N. Younggren. “Emergency management of difficult wounds: Part I.” Emergency medicine clinics of North America 25.1 (2007): 101-121.
- Banyard, A. C., and N. Tordo. “Rabies pathogenesis and immunology.” Revue Scientifique et Technique (International Office of Epizootics) 37.2 (2018): 323-330.
- Baxter, Megan, Kerina J. Denny, and Gerben Keijzers. “Antibiotic prescribing in patients who presented to the emergency department with dog bites: a descriptive review of current practice.” Emergency Medicine Australasia 32.4 (2020): 578-585.
- Bhaumik, Soumyadeep, Richard Kirubakaran, and Sirshendu Chaudhuri. “ Primary closure versus delayed or no closure for traumatic wounds due to mammalian bite.” Cochrane Database of Systematic Reviews 12 (2019).
- Chen, Esther, et al. “Primary closure of mammalian bites.” Academic Emergency Medicine 7.2 (2000): 157-161.
- Cheng, Hsu-Tang, Yung-Chang Hsu, and Chao-I. Wu. “Does primary closure for dog bite wounds increase the incidence of wound infection? A meta-analysis of randomized controlled trials.” Journal of Plastic, Reconstructive & Aesthetic Surgery 67.10 (2014): 1448-1450.
- Davis, Benjamin M., Glenn F. Rall, and Matthias J. Schnell. “Everything you always wanted to know about rabies virus (but were afraid to ask).” Annual review of virology 2 (2015): 451-471.
- Dean, D. J., G. M. Baer, and W. R. Thompson. “Studies on the local treatment of rabies-infected wounds.” Bulletin of the World Health Organization 28.4 (1963): 477.
- De Serres, Gaston, et al. “Bats in the bedroom, bats in the belfry: reanalysis of the rationale for rabies postexposure prophylaxis.” Clinical Infectious Diseases 48.11 (2009): 1493-1499.
- Dimcic, Tjasa, Minja Gregoric, and Vesna Breznik. “Rapidly Progressive Infection of Hand After a Cat Bite.” Cureus 12.7 (2020).
- Dire, Daniel J. “Cat bite wounds: risk factors for infection.” Annals of emergency medicine 20.9 (1991): 973-979.
- Elenbaas, Robert M., W. Kendall McNabney, and William A. Robinson. “Evaluation of prophylactic oxacillin in cat bite wounds.” Annals of emergency medicine 13.3 (1984): 155-157.
- Fernandez, Ritin, et al. “Water for wound cleansing.” Cochrane database of systematic reviews 9 (2002).
- Fooks, Anthony R., et al. “Rabies (Primer).” Nature Reviews: Disease Primers 3.1 (2017).
- Griego, Robert D., et al. “Dog, cat, and human bites: a review.” Journal of the American Academy of Dermatology 33.6 (1995): 1019-1029.
- Hampson, Katie, et al. “Estimating the global burden of endemic canine rabies.” PLoS neglected tropical diseases 9.4 (2015): e0003709.
- Henry, Francis P., Elizabeth M. Purcell, and Patricia A. Eadie. “The human bite injury: a clinical audit and discussion regarding the management of this alcohol fuelled phenomenon.” Emergency Medicine Journal 24.7 (2007): 455-458.
- Hemachudha, Thiravat, et al. “Human rabies: neuropathogenesis, diagnosis, and management.” The Lancet Neurology 12.5 (2013): 498-513.
- Henton, J., and A. Jain. “Cochrane corner: antibiotic prophylaxis for mammalian bites (intervention review).” Journal of Hand Surgery (European Volume) 37.8 (2012): 804-806.
- Iqbal, Tanzeem. “Towards evidence based emergency medicine: best BETs from the Manchester Royal Infirmary. BET 1. Antibiotics in cat bites.” Emergency medicine journal: EMJ 25.10 (2008): 686-687.
- Jaindl, Manuela, et al. “The management of bite wounds in children—a retrospective analysis at a level I trauma centre.” Injury 43.12 (2012): 2117-2121.
- Kaplan, M. M., et al. “Studies on the local treatment of wounds for the prevention of rabies.” Bulletin of the World Health Organization 26.6 (1962): 765.
- Keates, Liisamaria. “Rabies vaccines: WHO position paper—recommendations.” Vaccine (2010).
- Kennedy, Stephen A., Laura E. Stoll, and Alexander S. Lauder. “Human and other mammalian bite injuries of the hand: evaluation and management.” JAAOS-Journal of the American Academy of Orthopaedic Surgeons 23.1 (2015): 47-57.
- Khan, Muhammad N., and Abul H. Naqvi. “Antiseptics, iodine, povidone iodine and traumatic wound cleansing.” Journal of tissue viability 16.4 (2006): 6-10.
- Maimaris, C., and D. N. Quinton. “Dog-bite lacerations: a controlled trial of primary wound closure.” Emergency Medicine Journal 5.3 (1988): 156-161.
- Medeiros, Iara Marques, and Humberto Saconato. “Antibiotic prophylaxis for mammalian bites.” Cochrane Database of Systematic Reviews 2 (2001).
- Mitnovetski, Sergei, and Frank Kimble. “Cat bites of the hand.” ANZ journal of surgery 74.10 (2004): 859-862.
- Morgan, Marina, and John Palmer. “Dog bites.” Bmj 334.7590 (2007): 413-417.
- National Advisory Committee on Immunization. “Recommendations regarding the management of bat exposures to prevent human rabies.” Canada Communicable Disease Report 35.ACS-7 (2009): 1-28.
- National Institute for Health and Care Excellence. “Human and animal bites: antimicrobial prescribing guideline evidence review.” NICE. (2020): 1-55.
- National Institute for Health and Care Excellence. “Human and animal bites: antimicrobial prescribing guideline.” NICE. (2020): 1-27.
- Ong, Yee Siang, and L. Scott Levin. “Hand infections.” Plastic and Reconstructive Surgery 124.4 (2009): 225e-233e.
- Oehler, Richard L., et al. “Bite-related and septic syndromes caused by cats and dogs.” The Lancet infectious diseases 9.7 (2009): 439-447.
- Ontario Agency for Health Protection and Promotion (Public Health Ontario). Management of patients with suspected rabies exposure: guidance for health care providers working with your local public health unit. Toronto, ON: Queen’s Printer for Ontario; 2017.
- Ontario Ministry of Health. Management of potential rabies exposures guideline, 2020.
- Paschos, Nikolaos K., et al. “Primary closure versus non-closure of dog bite wounds. A randomised controlled trial.” Injury 45.1 (2014): 237-240.
- Pearce, John MS. “Louis Pasteur and rabies: a brief note.” Journal of Neurology, Neurosurgery & Psychiatry 73.1 (2002): 82-82.
- Perron, Andrew D., Mark D. Miller, and William J. Brady. “Orthopedic pitfalls in the ED: fight bite.” The American journal of emergency medicine 20.2 (2002): 114-117.
- Pfortmueller, Carmen A., et al. “Dog bite injuries: primary and secondary emergency department presentations—a retrospective cohort study.” The Scientific World Journal 2013 (2013).
- Quinn, James V., et al. “Randomized controlled trial of prophylactic antibiotics for dog bites with refined cost model.” Western journal of emergency medicine 11.5 (2010): 435.
- Rothe, Karin, Michael Tsokos, and Werner Handrick. “Animal and human bite wounds.” Deutsches Ärzteblatt International 112.25 (2015): 433.
- Rui-feng, Chen, et al. “Emergency treatment on facial laceration of dog bite wounds with immediate primary closure: a prospective randomized trial study.” BMC emergency medicine. Vol. 13. BioMed Central, 2013.
- Rupprecht, Charles E., et al. “Evidence for a 4-dose vaccine schedule for human rabies post-exposure prophylaxis in previously non-vaccinated individuals.” Vaccine 27.51 (2009): 7141-7148.
- Sartelli, Massimo, et al. “2018 WSES/SIS-E consensus conference: recommendations for the management of skin and soft-tissue infections.” World Journal of Emergency Surgery 13.1 (2018): 1-24.
- Schär, Lukas, et al. “Bite injuries to the hand and forearm: analysis of hospital stay, treatment and costs.” Journal of hand surgery (European volume) 48.1 (2023): 41-45.
- Seegmueller, Jessica, et al. “Cat and dog bite injuries of the hand: early versus late treatment.” Archives of orthopaedic and trauma surgery 140 (2020): 981-985.
- Shewring, D. J., et al. “The management of clenched fist ‘fight bite’injuries of the hand.” Journal of hand surgery (European volume) 40.8 (2015): 819-824.
- Singer, Adam J., and Alexander B. Dagum. “Current management of acute cutaneous wounds.” New England Journal of Medicine 359.10 (2008): 1037-1046.
- Singer, Adam J., Judd E. Hollander, and James V. Quinn. “Evaluation and management of traumatic lacerations.” New England Journal of Medicine 337.16 (1997): 1142-1148.
- Smith, P. F., A. M. Meadowcroft, and D. B. May. “Treating mammalian bite wounds.” Journal of clinical pharmacy and therapeutics 25.2 (2000): 85-99.
- Stevens, Dennis L., et al. “Practice guidelines for the diagnosis and management of skin and soft tissue infections: 2014 update by the Infectious Diseases Society of America.” Clinical infectious diseases 59.2 (2014): e10-e52.
- Suwansrinon, Kanitta, et al. “Is injecting a finger with rabies immunoglobulin dangerous?.” The American journal of tropical medicine and hygiene 75.2 (2006): 363-364.
- Talan, David A., et al. “Bacteriologic analysis of infected dog and cat bites.” New England Journal of Medicine 340.2 (1999): 85-92.
- Tabaka, Meg E., et al. “Predictors of infection from dog bite wounds: which patients may benefit from prophylactic antibiotics?.” Emergency Medicine Journal 32.11 (2015): 860-863.
- Talan, David A., et al. “Clinical presentation and bacteriologic analysis of infected human bites in patients presenting to emergency departments.” Clinical infectious diseases 37.11 (2003): 1481-1489.
- Tarantola, Arnaud. “Four thousand years of concepts relating to rabies in animals and humans, its prevention and its cure.” Tropical medicine and infectious disease 2.2 (2017): 5.
- Vermeulen, H., S. J. Westerbos, and D. T. Ubbink. “Benefit and harm of iodine in wound care: a systematic review.” Journal of Hospital Infection 76.3 (2010): 191-199.
- Warshawsky, Bryna, and Shalini Desai. “Exposure to bats: updated recommendations.” CMAJ 182.1 (2010): 60-60.
- World Health Organization. “Rabies vaccines: WHO position paper, April 2018–Recommendations.” Vaccine 36.37 (2018): 5500-5503.
- World Health Organization. WHO expert consultation on rabies: third report. Vol. 1012. World Health Organization, 2018.
- World Health Organization. “Zero by 30: The global strategic plan to end human deaths from dog-mediated rabies by 2030.” (2018).



Good recap, thanks. Saw my first dog bite of the year yesterday .