Gallstone diseases are common and can lead to various intraabdominal emergencies. Gallstones are found in approximately 6% of men and 9% of women in the United States [1]. Epidemiological studies in Scandinavia reported even higher prevalence, finding 13-18% of men and 15-25% of women had gallstones [2]. Common risk factors for developing gallstones include [3]:
- Female
- Obesity
- Rapid /cyclic weight loss
- Gallstones in a first-degree relative
- Hormone replacement therapy (estrogen)
- Ethnicity: Native American, Scandinavian
Of those diagnosed with asymptomatic gallstones, it is estimated that 1-4% will develop gallstone-related complications per year [4]. Common complications include biliary colic, cholecystitis, choledocholithiasis, pancreatitis, and cholangitis.
As such, it is essential to be able to diagnose and treat such conditions in the emergency department (ED). This review will summarize the newest literature in the diagnosis and management of the following conditions in the ED:
(1) Acute cholecystitis:
- History and physical
- Diagnostic criteria
- Imaging and the role of Point of care ultrasound POCUS
- Management
(2) Choledocholithiasis:
- Diagnostic criteria
- Role and efficacy of Serology
- Imaging modalities
- Management
(3) Cholangitis:
- Diagnostic criteria
- Management
Acute Cholecystitis
History and Physical
Acute cholecystitis is widely regarded as an acute inflammation of the gallbladder usually caused by gallstones obstructing the cystic duct. As with any medical condition, the diagnosis of cholecystitis requires an accurate history and physical:
- History
- RUQ or epigastric pain
- Constant and severe pain with radiation to the back or right shoulder
- Pain usually occurs postprandial
- May have had previously shorter episodes occurring after meals
- Fever, nausea, or vomiting
- Physical exam
- RUQ tenderness
- Positive Murphy sign
- Peritoneal signs
Unfortunately, the independent diagnostic value of some of these findings has been put into question by JAMA [5] which published the following findings:
- RUQ pain, + LR: 1.5
- RUQ tenderness, + LR: 1.6
- Fever, + LR: 1.5
- Emesis, + LR: 1.5
- Murphy sign, + LR: 2.8
In that same publication, no single liver enzyme or LFT had a significant LR, sensitivity (Sn), or specificity (Sp) for acute cholecystitis. Surprisingly, leukocytosis and fever only had a LR of 1.6. To put these into perspective, the HiNTS exam has a + LR of 17.2
Diagnostic Criteria
Given these findings and the uncertainty facing many emergency physicians and general surgeons, the Tokyo guidelines were created in 2007 (revised in 2013 and most recently in 2018). This is a consensus statement created by an international committee of surgeons, internists, and emergency physicians primarily from Japan but included expertise from Taiwan, USA, Korea, Netherlands, UK, India, Italy, South Africa, Singapore, Greece, France, Argentina, and Hong Kong [6].
- No significant changes in diagnostic criteria from the TG13 guidelines
- Not many validation studies have been conducted on the diagnostic criteria. They are reviewed below:
- At the time, TG13 diagnostic criteria were self-validated in a small retrospective study, that found they were 91.2% Sn and 96.9% Sp. [7]
- Limitations: retrospective, small N, only done in Japan, more males than females
- A separate validation study was done in Australia [8]
- Sn 83.1%, Sp, 37.1%.
- Over-diagnosed 62.5%. The overall accuracy was 60.3%
- Limitations: Retrospective, few patients, only used ultrasound for imaging with lower SN/SP for ultrasound compared to other studies
- Large collaborative Japan-Taiwan epidemiological study of over 5000 patients [9]
- Local signs of inflammation found in 89.1% of patients
- Systemic signs of inflammation found in 85.9% of patients
- Imaging findings of acute cholecystitis found in 86.4%
- Diagnostic criteria with definite diagnosis in 74.5%
- At the time, TG13 diagnostic criteria were self-validated in a small retrospective study, that found they were 91.2% Sn and 96.9% Sp. [7]
Imaging
Due to its low cost, ease of access, and little to no harm, ultrasound is the first line imaging for suspected gallstones and acute cholecystitis. In a recent systematic review and meta-analysis comparing different imaging modalities, the following Sn and Sp were found [10]:
- Ultrasound (26 studies): Sn: 81%, Sp: 83%
- MRI (3 studies): Sn: 85%, Sp: 81%
- HIDA (cholescintigraphy): Sn: 96%, Sp: 90%
This review only included one study looking at the effectiveness of CT which was done in 1979, and it found a Sn 94% and Sp of 59%. Unfortunately, when looking at more recent literature, CT appears to have a Sn of 39% and Sp of 93% [11]. This is consistent with the message sent by gastroenterologists and surgeons alike suggesting that CT is a poor test to look for biliary disease.
Point of Care Ultrasound (POCUS)
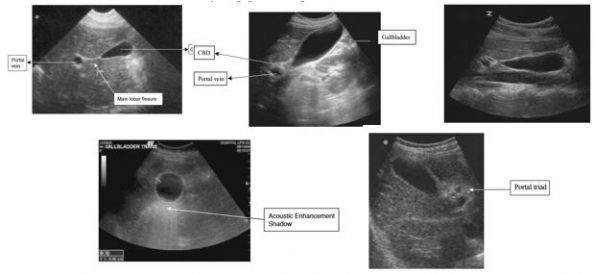
Over the last decade, POCUS has become a mainstay in many EDs worldwide. It is commonly used to look for free fluid in the abdomen, AAAs, IUPs in first-trimester pregnancy, pericardial effusions, hydronephrosis, cardiac function, and cholecystitis. Have a look at our Ultrasound Archives for more information on POCUS use in the ED. Before looking at the literature for identification of acute cholecystitis in the ED with POCUS, here is a basic review for the identification of the gallbladder:
- Use a curvilinear probe
- The patient can be supine but left lateral-decubitus often yields a better view
- Start with the probe oriented longitudinally under the right costal margin and sweep along the costal margin until an image of the gallbladder is obtained
- Asking the patient to take and hold a deep breath can often help with visualization
- Look for the exclamation point sign identifying the portal vein near the gallbladder
- Obtain a true longitudinal view by rotating probe on its axis
- Scan in multiple longitudinal and transverse planes fanning through the entire gallbladder
- HINT: Using the color setting may help differentiate the gallbladder from portal vein/IVC
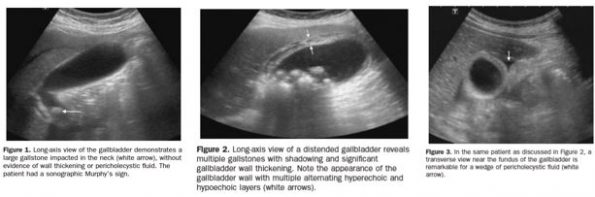
- Cholelithiasis
- Gallbladder wall thickening (>3 mm)
- Pericholecystic fluid
- Sonographic Murphy sign
- A prospective study released in the Annals of Emergency Medicine looking at the diagnostic accuracy of ED-performed POCUS and radiology-performed ultrasound for detection of acute cholecystitis as determined by surgical pathology found [13]:
- ED POCUS: Sn: 87%, Sp: 82%
- Radiology Ultrasound: Sn: 83%, Sp: 86%
- It concluded that a negative ED POCUS makes it unlikely that the patient has acute cholecystitis
- Limitations: Small sample size, physicians not blinded, experienced physicians/residents in ultrasound
- A similar study conducted in Tehran assessing the diagnostic accuracy of ED bedside ultrasound for acute cholecystitis found excellent agreement between POCUS and radiology performed ultrasound. Furthermore, they calculated bedside ultrasound as having [14]:
- Sn: 89.58%, Sp: 96.59% for detection of acute cholecystitis
- Limitations: Highly trained ED physicians/senior residents, small sample size
Despite this evidence, the current debate revolves around the utility of only using POCUS as the confirmatory imaging modality. Currently, in many centers including ours, the standard of care is to obtain a comprehensive radiology-performed ultrasound prior to general surgery referral despite having a positive POCUS. The primary reason appears to be a mix of medicolegal issues, consultant comfort with ED-performed ultrasound, and an already established standard of care and culture of practice.
Thus, studies are looking at the efficacy of going straight from POCUS to general surgery without obtaining a formal ultrasound. A recent study looked at just this topic after reaching an agreement between ED POCUS physicians and general surgeons. Overall, they found a significant reduction in ED wait time by exclusively performing POCUS prior to disposition. 29% of all patients who had RUQ scans were referred to general surgery. Of the 29 % who were referred, 43 % got cholecystectomy [15].
Management
In the Tokyo guidelines once a patient receives a diagnosis of acute cholecystitis, the next step would be to risk stratify them based on severity [6].
Since their release, numerous studies have validated this severity grading. Those with higher severity were found to have increased length of stay and higher 30-day mortality. Management of these patients in the ED requires:
- Early identification
- Resuscitative measures for critically ill, hemodynamically compromised patients
- Early antimicrobial therapy as per local antibiogram
- NOTE: Anaerobic therapy is indicated only if a biliary-enteric anastomosis is present/suspected
- Early referral to the appropriate service (often general surgery)
Bonus: Acalculous cholecystitis
- Almost exclusively seen in patients with low flow states (i.e. Critically ill)
- Often ICU patients with sepsis and without a clear source of jaundice.
- Patients often have numerous risk factors, most commonly an immunocompromised state.
Summary
- The Tokyo guideline is a helpful outline for diagnosing acute cholecystitis
- The diagnostic impression of an experienced physician is estimated to have a +LR of 25-30 [5]
- There is a growing role of POCUS in the ED
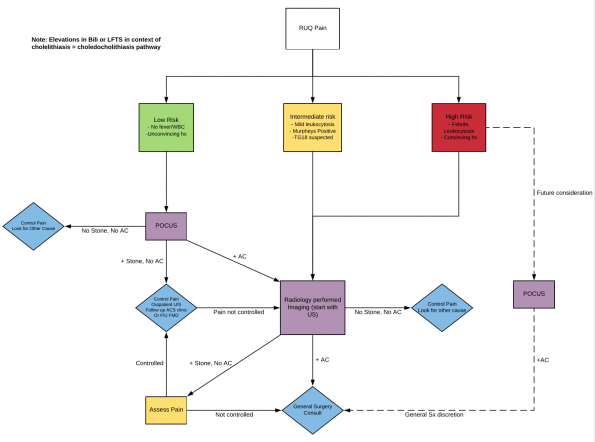
- Suggested pathway for incorporating POCUS into the diagnosis of acute cholecystitis in the ED:
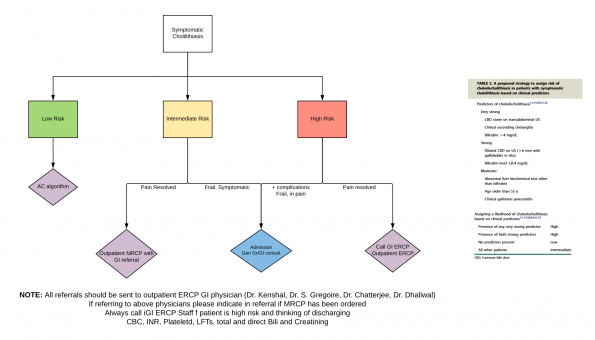
Choledocholithiasis
Approximately 10-20% of individuals having cholecystectomy will be found to have choledocholithiasis. Identifying these patients is often a difficult but essential task given the possibly deadly complications (i.e. pancreatitis, cholangitis). As above, these patients will often present with RUQ pain suggestive of biliary disease.
Diagnostic criteria
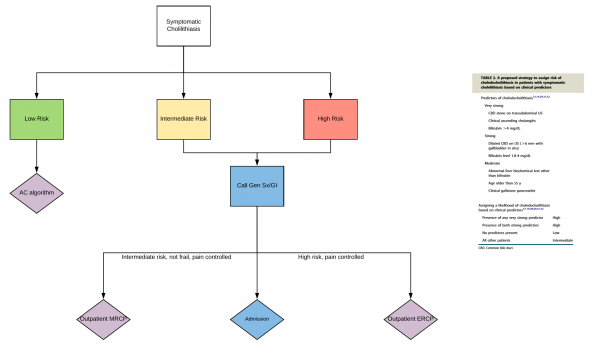
The most common diagnostic tool for identifying choledocholithiasis comes from the American society for gastrointestinal endoscopy. As shown below, this tool attempts to risk stratify patients based on patient characteristics, serologies, and imaging findings [16]:
According to these guidelines a high-risk patient has >50% likelihood, intermediate risk patients have between 10-50% likelihood, and low-risk patients have <10% likelihood of developing choledocholithiasis. Based on this the patient will undergo various investigations/procedures. Low-risk patients may go directly to laparoscopic cholecystectomy. Intermediate risk patients may go on to have endoscopic ultrasound vs. MRCP prior to ERCP. High-risk patients will often have ERCP.
Unfortunately, in a recent study [17] looking at the accuracy of common algorithms for predicting CBD stones which included the ASGE consensus, the results were moderate at best.
- Patients categorized as high risk for CBD according to ASGE: Sn: 71%, Sp: 50%
- Patients found to be at moderate/intermediate risk: Sn: 98%, Sp: 4%
This suggests that the ASGE guidelines may help in ruling out the presence of CBD stones in certain populations but do not accurately predict the presence of a stone.
What about liver function tests? Can they help predict Choledocholithiasis?
One of the most challenging aspects of dealing with biliary disease can be interpreting various LFTs and attempting to infer their diagnostic meaning especially when it comes to choledocholithiasis. Fortunately, there have been numerous studies looking to determine the exact accuracy of biochemical markers for CBD stones.
- In one retrospective study [18] looking at 1002 patients who underwent laparoscopic cholecystectomy, the values of 5 biochemical parameters that were measured preoperatively (GGT, ALP, ALT, AST, and total bilirubin) to assess their role in predicting CBD stones.
- The study listed the following as abnormal values: GGT >60 U/L, ALP >100 U/L, ALT >40 U/L, AST >45 U/L, Total Bili >27 mmol/L. (Note: these are similar to the values provided by the Medical Council of Canada)
Results:
- At least one abnormal elevation among the 5 parameters had a SN of 87.5%
- Total Bili had the highest specificity at 87.5%, PPV of 27.4, and LR at 3.9
- GGT had the best sensitivity at 84.1 and a NPV of 97.9
Summary:
- Total bilirubin is the best test to detect CBD stones
- If all 5 biochemical markers are normal, then it is highly unlikely that the patient has a CBD stone
- If the GGT is normal, there is a 97.9% chance that the patient does not have a CBD stone
Another more recent retrospective study [19] analyzed the LFTs of 1059 patients who visited the ER with gallbladder related symptoms. They were subdivided into 3 groups:
- acute cholecystitis without CBD stone (AC – CBD),
- acute cholecystitis with CBD stone (AC + CBD), and
- chronic cholecystitis without CBD stone (CC – CBD).
Acute cholecystitis was diagnosed based on TG13 and CBD stones were detected by ERCP or surgery.
Results:
- LFTs were greatest in AC+CBD on admission
- GGT was the most reliable predictor with a cutoff value of 224 yielding Sn: 80.6, Sp: 75.3
- All LFTs had NPV >90%
- LFTs in the CBD group did not improve until stone removed
What imaging modality best detects CBD stones?
There are numerous imaging modalities available to detect CBD stones. In the ED the most readily available are ultrasound and CT. As stated above, the consensus is that CT is a poor test to look at the biliary tree. Below is a summary of other available modalities and their accuracy in detecting CBD stones.
- A Cochrane review [20] comparing ultrasound to LFTs for diagnosis of CBD stones found:
- Ultrasound had a Sn: 0.73 and a Sp: 0.91
- Bilirubin >22.23 mmol/L: Sn: 0.84 and a Sp: 0.91
- Bilirubin > 2x normal limit: Sn: 0.42, Sp: 0.97
- Of note, the authors note these studies were of poor methodological quality and results of ultrasound varied considerably
- Another study looking at serology and ultrasound for diagnosis of choledocholithiasis [21] found that ultrasound had a Sn of 55% and Sp of 75% in detecting dilated CBD
- A meta-analysis (27 studies, 2673 patients) looking at the performance of endoscopic ultrasound in detecting choledocholithiasis [22] found it had a Sn: 94% and a Sp: 95%
- Another systematic review looking at a total of 13 studies [23] found that MRCP had a Sn of 93% and a Sp of 94%
Summary
- Normal LFTs make it highly unlikely that a CBD stone is present
- Total bilirubin appears to be the most specific biomarker for detecting CBD stones
- Ultrasound is not a great test for CBD stones, but it is easily performed and better than CT
- ASGE risk stratification is not perfect, but a reasonable place to start
- Below are two proposed pathways for the management of patients with suspected choledocholithiasis based on ASGE stratification
- NOTE: This is institution dependent. Please discuss with your hospital for local practices. We highly recommend speaking to GI before sending any patient home who is at intermediate or high risk for having CBD stones. There is a role for talking to local experts before sending patients home who are stable and pain-free to arrange prompt GI follow up in 1-2 days for investigations.
Cholangitis
Cholangitis is inflammation of biliary tree most commonly caused by bacterial infection due to biliary stasis or obstruction. The most common cause of cholangitis is gallstones, but other causes include primary sclerosing cholangitis, benign biliary stenosis, and malignant disease to name a few.
Diagnosis
In the literature, Charcot’s triad (RUQ pain, fever, and jaundice) is often cited as an identifying feature of cholangitis. Although this has a very high specificity, recent studies suggest that this is only 26% sensitive [24]. In such a life-threatening disease missing even a few cases can have fatal outcomes. As such, the Tokyo guidelines have proposed diagnostic criteria to improve sensitivity (~90%) for detecting acute cholangitis. Of note, these are unchanged from TG13 diagnostic criteria. They are outlined below [24]:
Imaging
As stated above, the diagnostic ability of ultrasound is limited in assessing the common bile duct. Given that patients with cholangitis will often present to the ED with some form of hemodynamic instability it is the opinion of gastroenterology and general surgery at our center to obtain a CT scan first to rule out other intra-abdominal causes.
Management
- Early empiric antibiotics and fluids
- Low threshold for suspicion
- Early call to gastroenterology and general surgery as treatment will require both (endoscopic, percutaneous, surgical)
References
- Everhart, J., Khare, M., Hill, M., & Maurer, K. (1999). Prevalence and ethnic differences in gallbladder disease in the United States. Gastroenterology, 117(3), 632-639. doi: 10.1016/s0016-5085(99)70456-7
- Halldestam, I., Enell, E., Kullman, E., & Borch, K. (2004). Development of symptoms and complications in individuals with asymptomatic gallstones. British Journal Of Surgery, 91(6), 734-738. doi: 10.1002/bjs.4547
- Bellows, C., Berger, D., & Crass, R. (2005). Management of Gallstones. American Family Physician, 72(4), 637-642.
- Sanders, G., & Kingsnorth, A. (2007). Gallstones. BMJ, 335(7614), 295-299. doi: 1136/bmj.39267.452257.ad
- Trowbridge, R., Rutkowski, N., & Shojania, K. (2003). Does This Patient Have Acute Cholecystitis? JAMA, 289(1). doi: 10.1001/jama.289.1.80
- Yokoe, M., Hata, J., Takada, T., Strasberg, S., Asbun, H., & Wakabayashi, G. et al. (2018). Tokyo Guidelines 2018: diagnostic criteria and severity grading of acute cholecystitis (with videos). Journal Of Hepato-Biliary-Pancreatic Sciences, 25(1), 41-54. doi: 10.1002/jhbp.515
- Yokoe, M., Takada, T., Strasberg, S., Solomkin, J., Mayumi, T., & Gomi, H. et al. (2012). New diagnostic criteria and severity assessment of acute cholecystitis in revised Tokyo guidelines. Journal Of Hepato-Biliary-Pancreatic Sciences, 19(5), 578-585. doi: 10.1007/s00534-012-0548-0
- Naidu, K., Beenen, E., Gananadha, S., & Mosse, C. (2016). The Yield of Fever, Inflammatory Markers and Ultrasound in the Diagnosis of Acute Cholecystitis: A Validation of the 2013 Tokyo Guidelines. World Journal Of Surgery, 40(12), 2892-2897. doi: 10.1007/s00268-016-3660-5
- Yokoe, M., Takada, T., Hwang, T., Endo, I., Akazawa, K., & Miura, F. et al. (2017). Descriptive review of acute cholecystitis: Japan-Taiwan collaborative epidemiological study. Journal Of Hepato-Biliary-Pancreatic Sciences, 24(6), 319-328. doi: 10.1002/jhbp.450
- Kiewiet, J., Leeuwenburgh, M., Bipat, S., Bossuyt, P., Stoker, J., & Boermeester, M. (2012). A Systematic Review and Meta-Analysis of Diagnostic Performance of Imaging in Acute Cholecystitis. Radiology, 264(3), 708-720. doi: 10.1148/radiol.12111561
- Shakespear, J., Shaaban, A., & Rezvani, M. (2010). CT Findings of Acute Cholecystitis and Its Complications. American Journal Of Roentgenology, 194(6), 1523-1529. doi: 10.2214/ajr.09.3640
- Noble, V., Nelson, B., & Sutingco, N. (2007). Manual of Emergency and Critical Care Ultrasound(pp. 136-151). Cambridge University Press.
- Summers, S., Scruggs, W., Menchine, M., Lahham, S., Anderson, C., & Amr, O. et al. (2010). A Prospective Evaluation of Emergency Department Bedside Ultrasonography for the Detection of Acute Cholecystitis. Annals Of Emergency Medicine, 56(2), 114-122. doi: 10.1016/j.annemergmed.2010.01.014
- Shekrachi, B., Rafsanjani, S., Fomani, N., & Chahardoli, M. (2018). Emergency Department Bedside Ultrasonography for Diagnosis of Acute Cholecystitis; a Diagnostic Accuracy Study. Emerg (Tehran), 6(1).
- Hilsden, R., Leeper, R., Koichopolos, J., Vandelinde, J., Parry, N., Thompson, D., & Myslik, F. (2018). Point-of-care biliary ultrasound in the emergency department (BUSED): implications for surgical referral and emergency department wait times. Trauma Surgery & Acute Care Open, 3(1), e000164. doi: 10.1136/tsaco-2018-000164
- Maple, J., Ben-Menachem, T., Anderson, M., Appalaneni, V., Banerjee, S., & Cash, B. et al. (2010). The role of endoscopy in the evaluation of suspected choledocholithiasis. Gastrointestinal Endoscopy, 71(1), 1-9. doi: 10.1016/j.gie.2009.09.041
- Ebrahim, M., Sorensen, L., Jorgensen, L., & Kalaitzakis, E. (2018). Current clinical algorithms for predicting common bile duct stones have only moderate accuracy. Digestive Endoscopy, 30(4), 477-484. doi: 10.1111/den.12994
- Yang, M., Chen, T., Wang, S., Tsai, Y., Su, C., & Wu, C. et al. (2007). Biochemical predictors for absence of common bile duct stones in patients undergoing laparoscopic cholecystectomy. Surgical Endoscopy, 22(7), 1620-1624. doi: 10.1007/s00464-007-9665-2
- Ahn, K., Yoon, Y., Han, H., & Cho, J. (2016). Use of Liver Function Tests as First-line Diagnostic Tools for Predicting Common Bile Duct Stones in Acute Cholecystitis Patients. World Journal Of Surgery, 40(8), 1925-1931. doi: 10.1007/s00268-016-3517-y
- Gurusamy, K., Giljaca, V., Takwoingi, Y., Higgie, D., Poropat, G., Štimac, D., & Davidson, B. (2015). Ultrasound versus liver function tests for diagnosis of common bile duct stones. Cochrane Database Of Systematic Reviews. doi: 10.1002/14651858.cd011548
- Isherwood, J., Garcea, G., Williams, R., Metcalfe, M., & Dennison, A. (2014). Serology and ultrasound for diagnosis of choledocholithiasis. The Annals Of The Royal College Of Surgeons Of England, 96(3), 224-228. doi: 10.1308/003588414×13814021678033
- Tse, F., Liu, L., Barkun, A., Armstrong, D., & Moayyedi, P. (2008). EUS: a meta-analysis of test performance in suspected choledocholithiasis. Gastrointestinal Endoscopy, 67(2), 235-244. doi: 10.1016/j.gie.2007.09.047
- Kaltenthaler, E., Bravo Vergel, Y., Chilcott, J., Thomas, S., Blakeborough, T., Walters, S., & Bouchier, H. (2004). A systematic review and economic evaluation of magnetic resonance cholangiopancreatography compared with diagnostic endoscopic retrograde cholangiopancreatography. Health Technology Assessment, 8(10). doi: 10.3310/hta8100
- Kiriyama, S., Kozaka, K., Takada, T., Strasberg, S., Pitt, H., & Gabata, T. et al. (2018). Tokyo Guidelines 2018: diagnostic criteria and severity grading of acute cholangitis (with videos). Journal Of Hepato-Biliary-Pancreatic Sciences, 25(1), 17-30. doi: 10.1002/jhbp.512