Ischemic stroke is one of Canada’s leading causes of morbidity and mortality. Over the last 30 years, we have observed rapid advances concerning how we identify, respond to, and treat patients with acute ischemic stroke. Because this topic is so vast, the focus of this blog post will be:
- To review the latest evidence for endovascular therapy
- To briefly outline how stroke care is currently being delivered in the Ottawa region
- To provide an evidence-based approach to TIA management in the ED
Evidence Review for Endovascular Therapy (EVT) in Acute Ischemic Stroke
Ischemic Stroke Background
- 60-80% of patients with ischemic stroke secondary to a large vessel occlusion will either die or be functionally dependent at 90 days if reperfusion is not achieved1.
- tPA, our previous standard, appears to be much less effective at breaking down large well-established clots such as those found in more proximal blood vessels2.
Early evidence for EVT
In 2015, the first strong evidence for endovascular therapy in the setting of large vessel anterior circulation occlusion came when 5 multicentre RCTs were published in rapid succession in the New England Journal of Medicine:
- MR CLEAN (January 2015)
- ESCAPE (February 2015)
- EXTEND-IA (February 2015)
- REVASCAT (April 2015)
- SWIFT PRIME (June 2015)
For more information on these studies, please see Dr. Simeon Mitchell’s previous Grand Rounds post here.
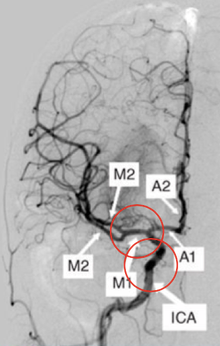
Across most studies, large vessel anterior circulation occlusions refer to those found in the intracranial aspect of the internal carotid artery and the first segment of the middle cerebral artery (M1) as seen in the above schematic.
Commonalities between these five trials include the following:
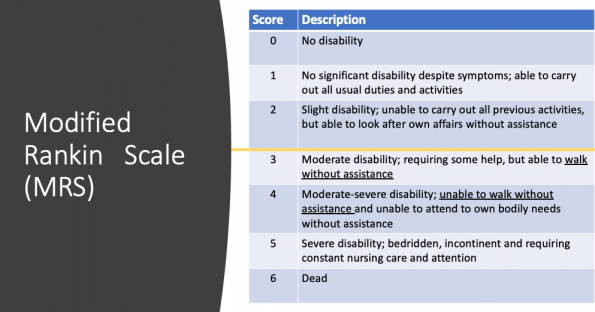
- All assessed functional disability at 90 days using the Modified Rankin Scale (MRS) as a primary outcome (see figure below for a refresher)
- To be included, patients needed to have large strokes with a score of at least 6 on the National Institutes of Health (NIH) Stroke Scale. The median NIH score for patients actually enrolled was much higher across all trials.
- Time of inclusion ranged from 4.5-12 hours, but the median time from symptom onset to randomization was well under 6 hours in all of the trials
- There existed significant heterogeneity in imaging selection across trials from a simple CT/CTA in MR CLEAN to more advanced perfusion imaging in EXTEND-IA and SWIFT PRIME
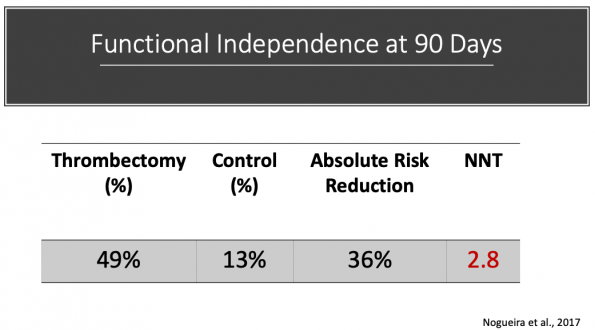
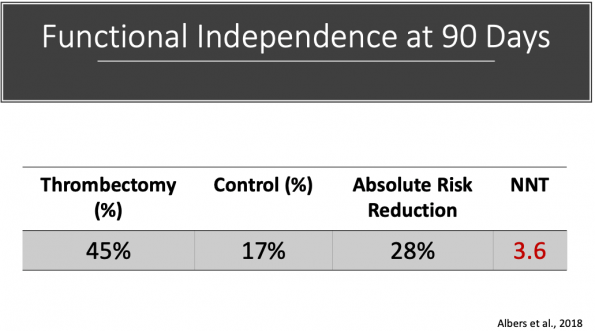
Pooled data from the above trials looking at functional independence at 90 days using a Modified Rankin score of 0-2 demonstrated the following1–6:
- Absolute Risk Reduction (ARR) for EVT vs control = 19.5%
- Number needed to treat (NNT) = 5.1
Therefore, 5 patients with a proximal large vessel occlusion need to be treated with EVT to have one additional functionally independent outcome. Pretty convincing stuff. But again, the majority of patients included in these initial studies were randomized in well under six hours. How effective is EVT in more extended time windows?
Extended Time Window Studies
DAWN7
- Multicenter RCT of 206 patients from 27 centers in the US, Canada (Toronto Western Hospital), Australia, and Europe
- Patients were randomized between 6 and 24 hours since last known well to receive either mechanical thrombectomy or standard medical care.
- Median time to randomization was 12.2 hours
- Inclusion
- Functionally independent patient
- Proximal occlusion of the ICA or M1
- NIH score of 10 or greater
- Small infarct core on diffusion-weighted MRI or CT perfusion imaging using RAPID software
- Co-primary outcome: functional independence at 90 days (MRS 0-2)
- Results:
- All meaningful subgroups favored thrombectomy
- Mortality benefit NOT demonstrated
- Trial ultimately stopped early for efficacy
In summary, DAWN selected patients with significant clinical deficits (reflected in the high NIH cutoff) that were out of keeping with the small volume of ischemic core identified on advanced neuroimaging. This allowed them to enroll patients with a large volume of at risk, but a potentially salvageable brain. The results speak for themselves.
DIFFUSE-38
- Multicentre RCT of 192 patients from 26 centers in the United States
- Patients randomized between 6 and 16 hours since last known well to receive either mechanical thrombectomy or standard medical management.
- Median time to randomization was 10.8 hours
- Primary outcome: ordinal score on the MRS at 90 days
- Secondary outcome: functional independence at 90 days (MRS 0-2)
- Trial stopped early after an interim analysis prompted by the publication of DAWN results
- Inclusion:
- Functionally independent patient
- NIH score of 6 or greater
- Proximal ICA or M1 occlusion
- Small ischemic core (<70cc), large penumbral volume (>15cc), and a mismatch ratio of 1.8 or greater on diffusion-weighted MRI (25%) or CT perfusion (75%) using the RAPID processing software
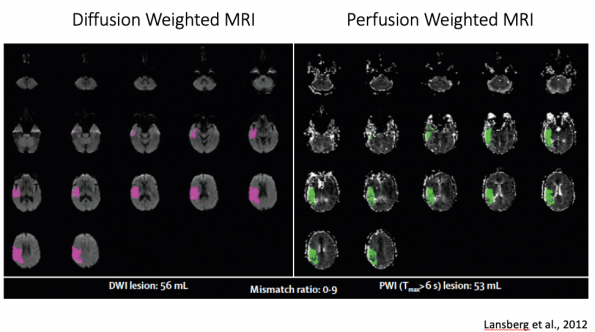
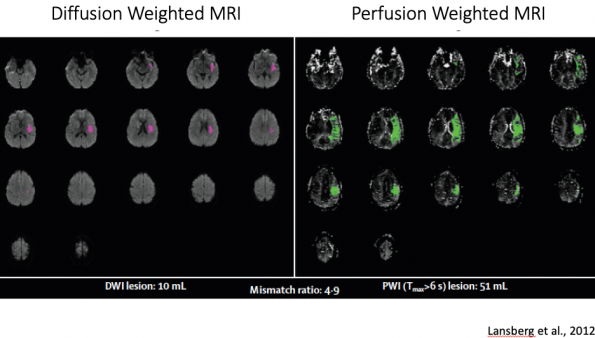
Example of a poor thrombectomy candidate on perfusion imaging9:
The volume of ischemic core (pink) equals the volume of potentially salvageable tissue or penumbra (green), representing a mismatch ratio approaching 1. There is no salvageable brain here.
Example of a good thrombectomy candidate on perfusion imaging9:
The volume of potentially salvageable tissue (green) exceeds the volume of ischemic core (pink) by about 5 times. This gives us a mismatch ratio of 5 and would make for a strong EVT candidate. There is a lot of potentially salvageable brain here.
- Results:
- DIFFUSE-3 also showed a mortality benefit with a NNT of 8.3
- Only second thrombectomy trial to do so with ESCAPE being the other
- All meaningful subgroups favored thrombectomy
- Results were again superior to the under 6-hour trials
- DIFFUSE-3 also showed a mortality benefit with a NNT of 8.3
EVT Trial Limitations
- All 7 RCT’s discussed were open-label studies meaning that patients were aware that they received endovascular therapy – the latest, greatest, state of the art treatment
- The inherent subjectivity of Modified Rankin Scale as an outcome measure
- 6/7 trials were stopped early which has the propensity to overestimate treatment effect
- To be included, patients needed to be functionally independent
- This makes sense when you consider the main outcomes for these trials being functional independence, but this likely does not reflect the spectrum of patients we see in the ED
- RAPID processing software used in DAWN, DIFFUSE-3, SWIFT PRIME, and EXTEND-IA to identify ideal thrombectomy candidates is not available in most Canadian centers
- In Ottawa, patients are being selected in a very similar fashion to the ESCAPE trial. CTA is used to identify either proximal ICA or M1 occlusion, the ASPECTS score (calculated on plain CT) is used to identify a small ischemic core, and dynamic CT is used to look for collateral flow (penumbra).
Take-Aways
- Despite the limitations, thrombectomy works! The remarkable consistency of results across trials confirms this and has changed the way we manage our patients with acute ischemic stroke.
- Time is a surrogate for perfusion mismatch. If a mismatch of a small ischemic core and large penumbra volume exists, it does not matter how long a patient is from their “time last seen well,” they are likely to benefit from mechanical thrombectomy. DAWN and DIFFUSE-3 showed this with lower NNT’s than the under 6-hour trials. Where time still does matter is that the percentage of patients that have this perfusion mismatch decreases with time. This is an important consideration when making decisions related to regional stroke bypass guidelines.
Pre-Hospital Stroke Bypass
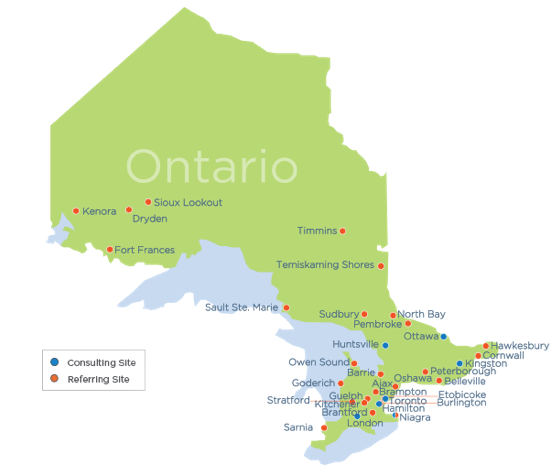
In Ontario, stroke centers are divided into primary stroke centers (outlined in red) and comprehensive stroke centers (outlined in blue).
Primary stroke centers, among other things, need to have 24 hour CT/CTA availability, stroke expertise either on-site or available via Telestroke, and t-PA administration capability. In the Champlain LHIN, the primary stroke centers are as follows:
- Hawkesbury
- Cornwall
- Pembroke
Comprehensive stroke centers have all of the features of a primary stroke center in addition to 24/7 stroke team coverage on-site and EVT capability. The Civic Hospital is the Champlain LHIN’s comprehensive stroke center.
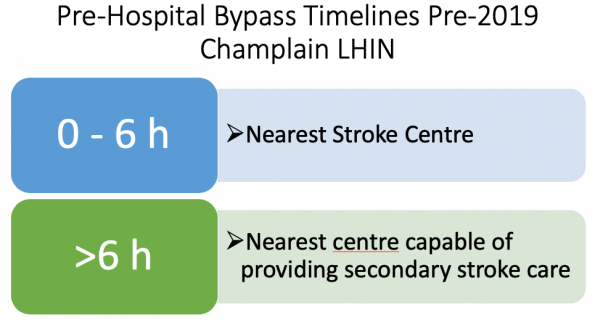
Prior to this year, the pre-hospital stroke bypass timelines were as follows:
For patients within 6 hours of symptom onset, paramedics would perform a quick stroke screen based on the FAST acronym. If the patient screened positive, they would be brought directly to the nearest stroke center, either primary or comprehensive.
The problem with this system was that for patients with large vessel occlusions that lived closer to a primary stroke center than a comprehensive stroke center, the ambulance could actually take them further away from an EVT capable hospital. In certain instances, this practice would delay time to definitive management (think of a patient living in Renfrew going to Pembroke rather than the Civic).
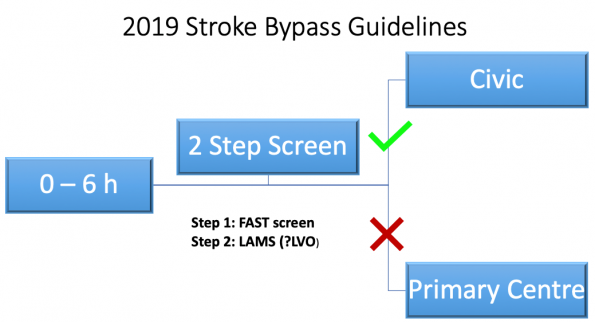
To correct this, new stroke bypass guidelines were developed and implemented for 2019 in the Champlain LHIN.
Timelines have remained unchanged, but paramedics are now performing a two-step screen on scene. The first step involves the same FAST screen as before. The second step involves the use of a more robust screening tool designed to help identify patients most likely to benefit from endovascular therapy. That is, patients most likely to have an anterior circulation large vessel occlusion (LVO). If the patient screens positive on this second step, they go directly to an EVT capable center (the Civic in the Champlain LHIN). If they screen negative on the second step, they go to the nearest stroke center, either primary or comprehensive, as they would have before.
Los Angeles Motor Scale (LAMS)
There are dozens of these screening tools and no robust evidence to declare one superior. The Los Angeles Motor Scale (LAMS) is one of the most popular and is what the paramedics in the Champlain LHIN are currently using.
The LAMS was retrospectively studied in 119 patients known to have an anterior circulation ischemic stroke. Using a cut-off of 4 or greater, this study found an 81% sensitivity and an 89% specificity for identifying patients with LVO.10 The benefit to this screening tool is that it is quick and straightforward to apply. The downside is that it requires the computation of a score and only consists of motor elements. You can imagine a patient presenting with a smaller, more distal stroke affecting the internal capsule that would screen positive despite not having a LVO. Because of this, I suspect the 89% specificity found in this study to be inflated. In fact, more recently, a prospective pre-hospital study published in Stroke found a 65% specificity for a LAMS score of 4 or greater at identifying patients with LVO.11
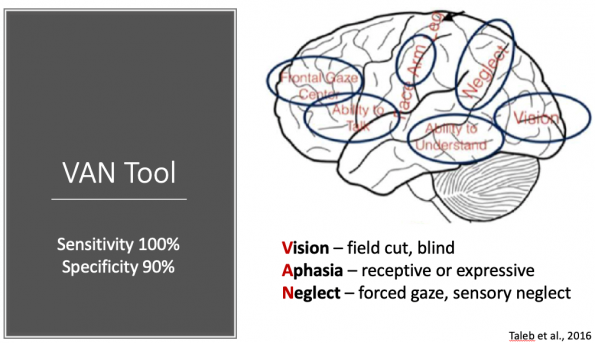
Vision, Aphasia, and Neglect (VAN)
The vision, aphasia, and neglect (VAN) screening tool is another instrument designed to help better identify stroke patients with LVO’s.
Published more recently in 2016, this tool is generating a lot of buzz. It consists of a two-step process that does not require the computation of a score. It works as follows:
- Step 1 – Pronator drift
- Absent – Stop here; Patient is VAN negative
- Present – Continue to step 2
- Step 2 – Vision (Field cut, double vision, blind) or Aphasia (expressive or receptive) or Neglect (forced gaze, unable to feel both sides at same time, ignoring one side)
- No abnormality in any of the above domains – Patient is VAN negative
- Abnormality present in any of the above domains – Patient is VAN positive
This tool appears to be more robust as it is testing function in multiple cortical areas (outlined in the schematic above). As you can imagine, it would take a large stroke to affect two or more of these areas (which is needed to screen positive). This tool was tested prospectively in 62 consecutive code stroke activations and was found to have a 100% sensitivity and a 90% specificity for identifying LVO.12 Obviously, these numbers are too small to draw definitive conclusions, but there are currently multiple validation studies underway, and it is certainly something to keep an eye on as it makes a lot of sense physiologically.
With respect to future directions, tools such as these have the potential to help us in the ED for patients presenting in more extended time windows by allowing us to better select when to activate a stroke code. If a patient screens negative at 12+ hours from symptom onset, they are unlikely to have a LVO and therefore unlikely to be an EVT candidate. From a resource utilization perspective, activating a stroke code in such cases may not be the best use of ED resources.
Take-Aways
- In the Champlain LHIN, we are currently using a two-step stroke screen in the pre-hospital setting to better select and more appropriately direct the patients most likely to have large vessel occlusions
- At present, there is no strong evidence to guide the selection of one screening tool over another. Expect significant future research in this area
- The use of a screening tool could help us in the ED by standardizing the way we activate our stroke codes in the 6-24 hour time window
An Evidence-Based Approach to TIA Management in the ED
Background
- Patients who suffer from a TIA are at a markedly elevated risk of experiencing a subsequent stroke
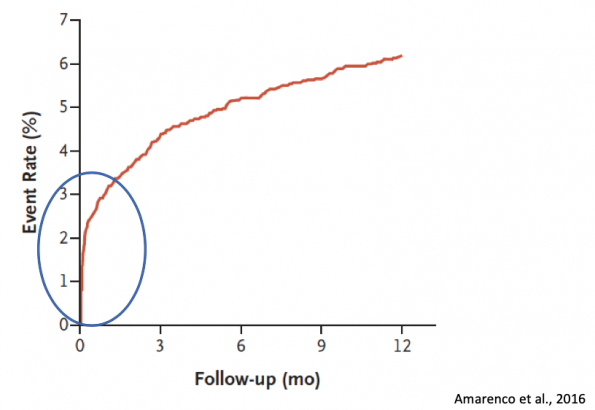
- Although previous studies have quoted the 90-day risk to be anywhere from 8-12%13, improved work-up, management, and access to follow-up in recent years has lowered this number to a more reasonable but still significant 5%14
- What remains true is that even with these better therapies, the risk of stroke remains highest in the immediate days following TIA
- This means that what we do in the emergency department matters!
Management of Atrial Fibrillation and TIA
- Straightforward – start an anticoagulant in the ED with very few exceptions
- If the patient is already on an anticoagulant, ensure compliance
- If the patient is already on an antiplatelet agent, this can potentially be held unless it is for a clear indication (post PCI).
- Use your judgement here
Evidence Gap
- For patients without A-Fib presenting with a TIA, the management has historically been less clear
- Starting the patient on Aspirin is the established standard, but…
- What if the patient is already on Aspirin?
- What about dual antiplatelet therapy?
CHANCE Trial15
- Multicenter, double-blind RCT in China that enrolled over 5000 patients
- Compared Aspirin monotherapy with a placebo to Aspirin-Plavix dual therapy
- The dual therapy group only received dual therapy for the first 21 days followed by Plavix monotherapy from day 22-90.
- The control group received ASA and a placebo for the entire 90 days.
- Plavix load was 300 mg
- ASA load was variable and dependent on physician discretion
- Exclusions:
- Any history of spontaneous intracerebral hemorrhage
- Patients with isolate sensory, visual, or vertigo symptoms were excluded to reduce the number of TIA mimics
- Primary outcome: new stroke (ischemic or hemorrhagic) at 90 days
- Results:
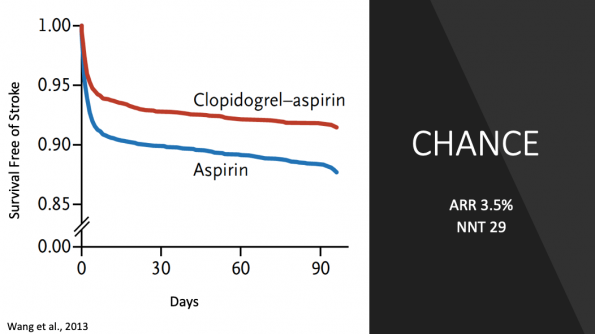
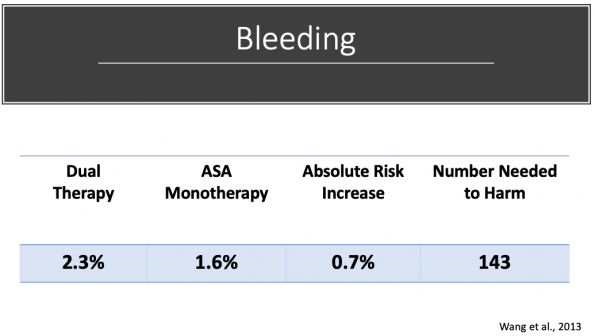
- Percentage of ischemic stroke events at 90 days in the dual group was 7.9% vs 11.3% in the ASA monotherapy group
- Relative risk reduction = 31%
- Absolute risk reduction = 3.5%
- Number needed to treat = 29
- Adverse events:
-
- Difference driven completely by non- major bleeding (bleeding leading to a transfusion but not leading to hemodynamic compromise)
- No difference in ICH, fatal bleeding, or bleeding leading to hemodynamic instability
- Criticism
- Chinese only population with higher overall stroke rate
- Polymorphism affecting Plavix metabolism more common in the Chinese population
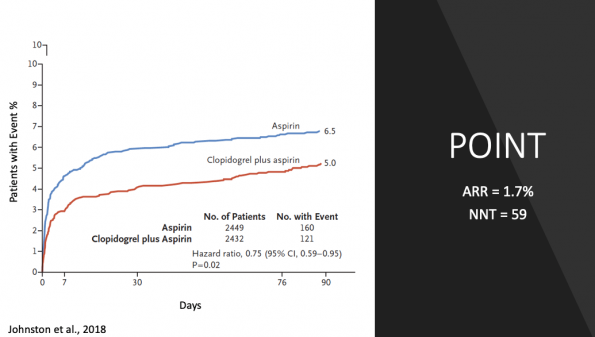
POINT16
- Randomized, double-blind, placebo-controlled study of nearly 5000 patients from 10 different countries including the US and Canada
- Patients were randomized within 12 hours following their index visit for TIA or minor stroke to receive either ASA + placebo for 90 days or ASA-Plavix dual therapy for 90 days.
- Loading dose of Plavix was 600 mg (double that of CHANCE)
- ASA load was again left to physician discretion
- Like CHANCE, patients with isolated sensory, visual, or dizziness symptoms were excluded to limit mimics
- Primary outcome: composite of ischemic stroke, myocardial infarction, or death from ischemic vascular causes at 90 days
- Secondary outcome: ischemic stroke
- Results:
- Percentage of ischemic stroke events at 90 days in the dual group was 4.6% 6.3% for the ASA monotherapy group (note significantly lower event rates than CHANCE)
- Relative risk reduction = 26%
- Absolute risk reduction = 1.7%
- Number needed to treat = 59
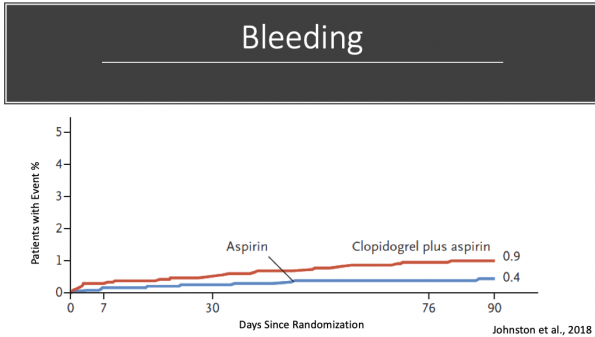
- Adverse Events:
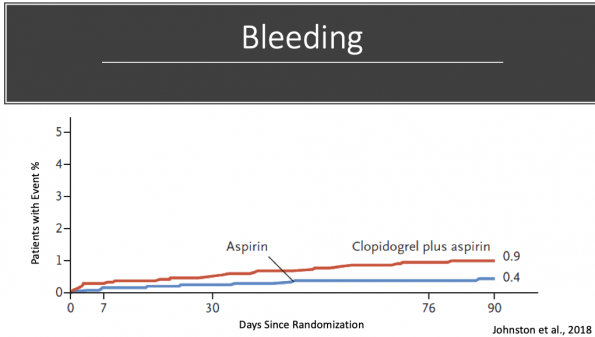
- Chart above reflects major bleeding, defined as:
- Symptomatic intracranial hemorrhage
- Intraocular bleeding with vision loss
- Transfusion of 2 or more units of red cells or whole blood equivalents (less important)
- Hospitalization or prolongation of current hospitalization (less important)
- Death due to hemorrhage
- Major bleed rates were 0.9% in the dual group 0.4% in ASA monotherapy group
- Absolute risk increase 0.5%
- Number needed to harm (NNH) = 200
- Difference in bleed rates were driven completely by non-intracranial major hemorrhage (GI bleeds)
- No difference in death, hemorrhagic stroke or symptomatic ICH
- Criticism
- Despite NNT of 59 to prevent one ischemic stroke and NNH of 200 to cause one GI bleed, the trial was stopped early due to crossing its pre-specified boundary for harm.
- High rate of medication non-compliance (similar between groups)
Evidence Summary
- Both CHANCE and POINT showed decreased ischemic stroke rates with dual antiplatelet therapy at 90 days with an NNT between 29 and 59
- There was no difference in either trial for fatal or intracranial bleeding
- In CHANCE, there was no difference in major bleeding – period
- In POINT, the difference in bleeding was driven by non-fatal GI bleeds
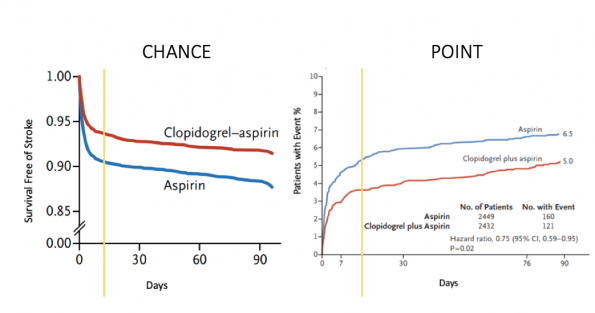
If we look at these two graphs again, we can see that the effect of dual antiplatelet therapy seems to be realized early and then levels off after that with the two slopes equaling each other.
Compare this with the bleeding graph from POINT. Notice the relatively linear slope of the dual antiplatelet graph, suggesting that bleed rates are low early, and continue to increase steadily with time.
Putting it together
- The risk of stroke following TIA is highest in the first few days after the index visit
- The greatest effect of dual antiplatelet therapy following TIA is realized early, certainly within the first three weeks but perhaps sooner
- The risk of bleeding increases linearly with time
Canadian Stroke Best Practices 2018 Recommendation
- Patients with non-cardioembolic very high-risk TIA should be treated with dual antiplatelet therapy for 21 days.
A Word on ‘Risk’
A patient’s risk depends on two factors:
- How likely is this to be a true TIA
- There is nothing that inherently makes one TIA ‘riskier’ than another. What is meant by the term ‘risk’ is how likely is it that the patient in front of you is truly experiencing a TIA and not a mimic? If you think the most likely diagnosis is TIA, the patient is high risk. If other diagnoses are equally likely (migraine, seizure, psychogenic), then the patient’s risk is lower.
- How soon did the patient present
- The highest risk of stroke following TIA is realized If the patient is presenting two weeks after their TIA, they have already made it through the highest risk period.
My Approach
- High-Risk TIA
- Plavix 300 mg load in the ED and a script for 75 mg daily for 20 subsequent days
- ASA 160 mg load in the ED and a script for 81 mg daily indefinitely
- If they are already on Aspirin, add Plavix
- If they are already on Plavix, add Aspirin
- If they are already on an agent, there is no need to reload that agent
- Low-Risk TIA
- ASA alone is reasonable
- Don’t send them to the stroke clinic on nothing!!
- If your suspicion for TIA is high enough to send them to stroke clinic, it is indefensible not to at least start Aspirin
- TIA with A-Fib
- Start a DOAC in the ED with very few exceptions
- If already on an antiplatelet, it can be reasonable to hold, but it depends on the indication
References
- Goyal M, Demchuk AM, Menon BK, et al. Randomized assessment of rapid endovascular treatment of ischemic stroke. N Engl J Med. 2015;372(11):1019-1030. doi:10.1056/NEJMoa1414905
- Goyal M, Menon BK, van Zwam WH, et al. Endovascular thrombectomy after large-vessel ischaemic stroke: a meta-analysis of individual patient data from five randomised trials. Lancet. 2016;387(10029):1723-1731. doi:10.1016/s0140-6736(16)00163-x
- Saver JL, Goyal M, Bonafe A, et al. Stent-Retriever Thrombectomy after Intravenous t-PA vs. t-PA Alone in Stroke. N Engl J Med. 2015;372(24):2285-2295. doi:10.1056/NEJMoa1415061
- Jovin TG, Chamorro A, Cobo E, et al. (REVASCAT) Thrombectomy within 8 Hours after Symptom Onset in Ischemic Stroke. N Engl J Med. 2015;372(24):150417035025009. doi:10.1056/NEJMoa1503780
- Campbell BCV, Mitchell PJ, Kleinig TJ, et al. Endovascular Therapy for Ischemic Stroke with Perfusion-Imaging Selection. N Engl J Med. 2015;372(11):1009-1018. doi:10.1056/NEJMoa1414792
- Berkhemer OA, Fransen PSS, Beumer D, et al. A Randomized Trial of Intraarterial Treatment for Acute Ischemic Stroke. N Engl J Med. 2015;372(1):11-20. doi:10.1056/NEJMoa1411587
- Nogueira RG, Jadhav AP, Haussen DC, et al. Thrombectomy 6 to 24 Hours after Stroke with a Mismatch between Deficit and Infarct. N Engl J Med. 2017;378(1):11-21. doi:10.1056/nejmoa1706442
- Albers GW, Marks MP, Kemp S, et al. Thrombectomy for Stroke at 6 to 16 Hours with Selection by Perfusion Imaging. N Engl J Med. 2018;378(8):708-718. doi:10.1056/NEJMoa1713973
- Lansberg MG, Straka M, Kemp S, et al. MRI profile and response to endovascular reperfusion after stroke (DEFUSE 2): A prospective cohort study. Lancet Neurol. 2012;11(10):860-867. doi:10.1016/S1474-4422(12)70203-X
- Nazliel B, Starkman S, Liebeskind DS, et al. A Brief Prehospital Stroke Severity Scale Identifies Ischemic Stroke Patients Harboring Persisting Large Arterial Occlusions. Stroke. 2008;39(8):2264-2267. doi:10.1161/STROKEAHA.107.508127
- Noorian AR, Sanossian N, Shkirkova K, et al. Los Angeles Motor Scale to Identify Large Vessel Occlusion. Stroke. 2018;49(3):565-572. doi:10.1161/STROKEAHA.117.019228
- Teleb MS, Hage A Ver, Carter J, Jayaraman M V., McTaggart RA. Stroke vision, aphasia, neglect (VAN) assessment-a novel emergent large vessel occlusion screening tool: pilot study and comparison with current clinical severity indices. J Neurointerv Surg. 2017;9(2):122-126. doi:10.1136/neurintsurg-2015-012131
- Perry JJ, Sharma M, Sivilotti MLA, et al. A prospective cohort study of patients with transient ischemic attack to identify high-risk clinical characteristics. Stroke. 2014;45(1):92-100. doi:10.1161/STROKEAHA.113.003085
- Amarenco P, Lavallée PC, Labreuche J, et al. One-Year Risk of Stroke after Transient Ischemic Attack or Minor Stroke. N Engl J Med. 2016;374(16):1533-1542. doi:10.1056/NEJMoa1412981
- Wang Y, Wang Y, Zhao X, et al. Clopidogrel with Aspirin in Acute Minor Stroke or Transient Ischemic Attack for the CHANCE Investigators*. N Engl J Med. 2013:11-19. doi:10.1056/NEJMoa1215340
- Johnston SC, Easton JD, Farrant M, et al. Clopidogrel and Aspirin in Acute Ischemic Stroke and High-Risk TIA. N Engl J Med. 2018;379(3):215-225. doi:10.1056/NEJMoa1800410