In Part 1 of our 2-part series on Pneumothorax, Dr. Simon Wells walked us through the varying definitions, epidemiology, diagnostic criteria, and imaging findings of pneumothorax including on point of care ultrasound (POCUS). Click here to review Part 1.
In today’s post, we’ll focus specifically on pneumothorax treatment options for your next shift in the ER. Additionally, we have a summary of the 2020 NEJM publication ‘Conservative versus Interventional Treatment for Spontaneous Pneumothorax’.
Pneumothorax treatment
There are multiple controversies in the subject of pneumothorax management, largely driven by the paucity of high-quality evidence in the area. Problems begin with the lack of international consensus on what constitutes a large pneumothorax, which contributes to substantial geographical variation in practice. One thing both the ACCP and BTS can agree on is that small pneumothoraces can be managed conservatively, however they differ in their approach to large pneumothoraces.25, 26
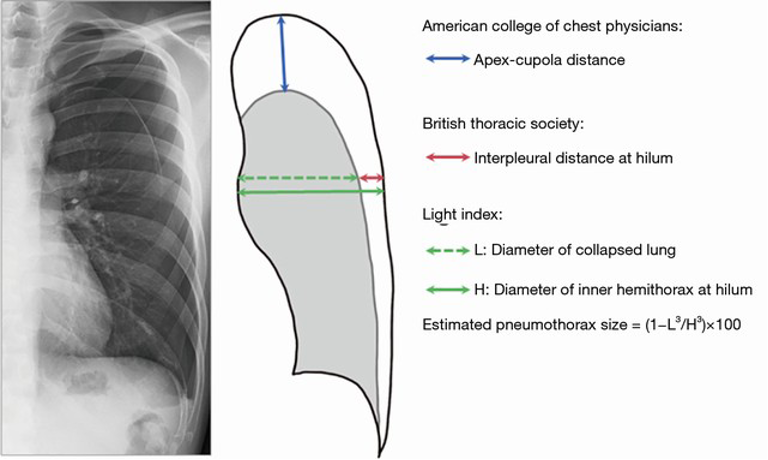
Four key management controversies we’ll highlight below:
- Small bore chest tube (ie. pigtail catheters) vs. large bore chest tube
- Inpatient vs. outpatient (ambulatory) management
- Needle aspiration vs. tube placement
- Conservative treatment vs. interventional treatment
Small bore vs large bore chest tubes
- A study by Benton et al. in 2009 demonstrated similar success rates for 6-12 French tubes vs. 20-32 French tubes in pneumothorax resolution: 39
- Importantly however it noted fewer complications, including infections, in the small bore group.
- A more recent meta-analysis in 2018 of 11 studies has further highlighted this point: 40
- Pigtail catheter – similar initial success rate to large bore tubes (79.8% vs 82.9%), while showing lower complication rate (even in secondary spontaneous pneumothorax), shorter drainage duration and overall decreased hospital length of stay (LOS).
- Pigtail catheters ultimately = smaller incisions, better patient comfort, ambulatory management possible.
- Large bore tubes should be generally reserved for: tension pneumothorax, hemopneumothorax, open pneumothorax, or refractory cases.
Bottom Line: Small bore chest tubes (pigtail) catheters have better patient centred outcomes.
Chest tube complications
- 10-20% of patients with a chest tube will suffer chest-tube related complications.41
- Early complications
- Iatrogenic – ie. improperly placed tubes into adjacent structures
- Positional – tube in pleural cavity but poorly positioned
- Noting once the sterile field is broken, a chest tube cannot be advanced.
- Late Complications
- Infection – cellulitis, empyema, pneumonia
- Give prophylactic antibiotics when chest tube inserted in trauma setting42
- Malfunction – persistent air leak
- Obstruction – occlusion with blood/pus
- Infection – cellulitis, empyema, pneumonia
Inpatient vs ambulatory management
As summarized by a 2012 systematic review (18 studies): 43
- Success with Pigtail + Heimlich Valve (HV): 1060/1235 (85.8%)
- Successful treatment as outpatient: 761/977 (77.9%)
- Similar rates of success for primary spontaneous, secondary spontaneous, & iatrogenic pneumothoraces
- Serious complications were rare
- Long-term outcomes similar to inpatient management
Bottom line: Outpatient ambulatory management is safe and effective.
Needle aspiration vs tube placement
- Needle aspiration involves the placement of a catheter into the pleural cavity and manually evacuating air from the cavity using a three-way stopcock and a 60cc syringe. 44
- The British Thoracic Society (BTS) recommends needle aspiration as the initial intervention in large or symptomatic PSP.
- The American College of Chest Physicians (ACCP) does not suggest needle aspiration in any circumstance.
- As per a 2017 Cochrane review on needle aspiration vs. intercostal tube drainage (6 RCT’s , N=435); 45
- Immediate success rate: tube thoracostomy > needle aspiration
- Simple needle aspiration (if successful): shorter hospitalization LOS (mean of 1.66 days)
- One-year, overall review: no difference in adverse events and hospitalization rates.
- Approximately 30% of patients will have a persistent air leak and needle aspiration will not achieve initial resolution in these patients.
Bottom Line: Needle aspiration can be a reasonable first approach, but is not performed at our centre and generally not performed in Canada.
Notes on needle decompression & the aspiration approach
- Traditional Approach – 2nd intercostal space (ICS), midclavicular line
- EM physicians frequently miss the location 46
- 14g angiocath (4.5cm) will fail to reach pleural space in 10-35% 47
- Modern Approach – 4th-5th ICS, anterior axillary line
- Chest wall is thinner
- Vital structures less at risk
- Easier to identify correct anatomical landmarks
Primary vs. secondary pneumothorax treatment
- Both the ACCP and BTS recommend admission for all secondary spontaneous pneumothoraces. 25,26
- The guidelines differ however on their suggested initial treatment strategy for these secondary pneumothoraces.
- Ultimately, these patients are more likely to get a definitive preventative treatment (surgery/pleurodesis).48
- However, evidence exists that they can be managed similar to primary spontaneous pneumothoraces – ambulatory treatment with a small-bore chest tube.
- Ultimately we need more studies to evaluate if patients benefit in the delineation of primary vs secondary pneumothorax when deciding on treatment options.
Oxygenation
- Assuming no air leakage, pneumothorax will gradually resolve as air is reabsorbed into pleural capillaries, with an estimated rate of 2.2% per day. 49
- For patients on 100% oxygen, the partial pressure of all the gases in the capillaries falls (due to displacement of nitrogen by O2), and the rate of reabsorption increased x 4 .50
- Reasonable to give 100% O2 for any patient admitted under observation.
Conservative vs interventional management
- Conservative management is largely ignored in the current available guidelines.
- It is only recommended in small and relatively asymptomatic primary spontaneous pneumothoraces.
- However, it has long been established that conservative management can be effective in even large primary & secondary pneumothoraces.
- Intervention became more widely practiced in 1970-80’s, largely due to advances in equipment, and became the standard.
- There were never any trials comparing intervention to conservative management in large PSP, until recently when a large high-quality RCT looking at this issue was published in thee NEJM.51
Study Review – Conservative versus Interventional Treatment for Spontaneous Pneumothorax (2020)51
Design
- Multi-center, prospective, randomized, open-label, non-inferiority trial
- 39 hospitals in Australia and New Zealand
- Patients 14-50 years old: average age 26, over 85% male, N=316
- Unilateral PSP of 32% or more on CXR according to the Collins method
- Mean PSP size: 65%
- Comparing conservative management to small bore chest tube (12 fr)
- <9% difference chosen as non-inferiority cut-off, with an anticipated 99% success rate after 8 weeks in the intervention group
Exclusion Criteria
- Not a secondary spontaneous pneumothorax
- Previous ipsilateral primary spontaneous pneumothorax
- Coexistent hemothorax
- Bilateral pneumothorax
- “Tension’ pneumothorax” (sBP <90 mmhg, MAP <65 mmHg, shock index ≥1)
- Pregnancy
- Social circumstances (unable to return or unlikely to present for f/u)
- Planned air travel within the following 12 weeks
Intervention
- Small chest tube (≤12 French), attached to an underwater seal, without suction
- CXR was obtained one hour later
- Lung re-expanded + drain not bubbling = drain closed with 3-way stopcock
- Repeat CXR 4 hours later: patient stable, PSP not recurred – drain out and discharged
- Different than usual practice in Canada, in that the patients had no Heimlich valve or chest tube at discharge.
- In a way, this technique more similarly mimics needle aspiration protocols.
- No resolution after initial treatment, or if primary pneumothorax recurred under observation – stopcock opened, underwater seal drain resumed, patient admitted.
Comparison (conservative therapy group)
- Patients observed for minimum of 4 hours before repeat CXR.
- If did not need supplemental O2 and were walking comfortably – Discharged home with analgesia and written instructions.
- Interventions were done under five conditions:
- Clinically significant symptoms persisted despite adequate analgesia
- Chest pain or dyspnea prevented mobilization
- Patient was unwilling to continue with conservative treatment
- Patient’s condition became unstable (SBP<90, Shock index >1, RR >30, Spo2 <90% on RA)
- Repeat CXR showed an enlarging pneumothorax along with physiological instability
Primary Outcome
- Full lung re-expansion on CXR, determined by treating MD, within 8 weeks
- Multiple secondary outcomes were also analyzed (see below)
Results
- Radiologic resolution at 8 weeks: intervention 98.5% vs. conservative group 94.4%
- Absolute difference 4.1% (95% CI -8.6-0.5%)
- P value for non-inferiority = 0.02
- Conservative therapy non-inferior to invasive therapy
- Numbers were very similar in the per-protocol analysis
| Secondary Outcomes | Intervention (N=154) | Conservative (N=162) | RR/HR (95% CI) |
| Days to CXR resolution (median) | 16 | 30 | HR 0.49 (0.39-0.63) |
| Complete symptom resolution at 8 weeks | 93.4% | 94.6% | RR 1.1 (-4.4-6.7) |
| Days to symptom resolution (median) | 15.5 | 14 | HR 1.11 (0.88-1.40) |
| PSP recurrence within 12 months | 16.8% | 8.8% | RR 1.90 (1.03-3.52) |
| Serious adverse events | 12.3% | 3.7% | RR 3.3 (1.37-8.1) |
| Inpatient days (mean) | 6.1 | 1.6 | RR 2.8 (1.8-3.6) |
| Days off work (mean) | 10.9 | 6.0 | RR 2.0 (1.0-3.0) |
| Chest tube for >72hrs | 51% | 9.3% | RR 41.7 (32.6-50.8) |
| Need for intervention | 94.2% | 15.4% | RR 6.1 (4.24-8.77) |
| Number of CXR total | 10.9 | 6.4 | RR 1.7 (1.6-1.8) |
Criticisms and study issues
- Lost patients: outcome data at 8 weeks for only 256/316 (272/316 at 9 weeks)
- Primary analysis treated data obtained after 8 weeks as missing
- Crossover: 25 patients in the conservative group underwent intervention
- 10 in the intervention arm declined treatment
- Selection: took 6 years to recruit 316 patients, over 2600 patients screened
- Only applies to a small subset of pneumothorax patients
- Sensitivity analysis – if accounting for all lost data as treatment failures:
- Primary Outcome: Intervention 93.5% vs. Conservative 82.5%
- Risk Difference -11.0% = outside of non-inferiority margin of -9%
Conclusions
- The authors concluded that this study “provides modest, but statistically fragile, evidence that conservative management was non-inferior to interventional management for radiographic resolution within 8 weeks…”
- It is reasonable to offer conservative management in a stable, mildly symptomatic Patient with a large first-time PSP, as long as you can ensure close follow-up.
- Conservative management = better patient centred outcomes.
- No RCT exists that demonstrates that chest tubes are better than conservative care.
- This trial addresses a critical question and lays the foundation for a possible shift in management of larger PSP’s.
- Ultimately this may leave us with a difficult choice as to what treatment is best-suited for the patient we are seeing. My suggestion would be to consider conservative management as an option, and to discuss this option with patients when laying out potential treatments. Depending on your institution, this management option may need to be cleared by thoracic surgery.
Pneumothorax treatment take-home points
- Small-bore chest tubes/pigtails should be used instead of large-bore chest tubes.
- Ambulatory management is safe and effective for most cases.
- Needle aspiration is a potential option, but not typically done in Canada.
- Conservative management is non-inferior to interventional management in one RCT and had better patient centred outcomes.
- Discuss conservative management as an option with select patients with large, primary spontaneous pneumothoraces.
References



This is an absolutely fantastic review. Thanks for putting all of it together.