Extra Corporeal Membrane Oxygenation (ECMO) has significantly progressed over the past decades. It has become an invaluable tool in the care of adults with severe cardiac and pulmonary disease refractory to conventional management. The implementation of ECMO has even been brought downstairs to the Emergency Department (ED) for select patient presentations in a few countries worldwide, but currently there is only one ED ECMO program in Canada.
We present this post in a 2-part series. In part 1, review the basics of ECMO, as well as a deep dive into the ECMO literature covering the use in specific presentations. In part 2, we will review the factors involved in implementing an ED ECMO program and what the future holds for ED ECMO at the Ottawa Hospital.
ECMO 101
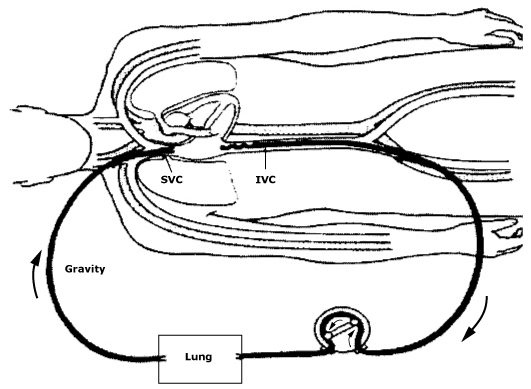
In the simplest form of ECMO, blood is drained from the vascular system, circulated outside the body via a mechanical pump, where it is oxygenated, CO2 removed, and warmed up, before being re-infused back into the bodies circulation.
When ECMO is incorporated into cardiopulmonary resuscitation it is called ECMO-CPR (E-CPR).
ECMO Types
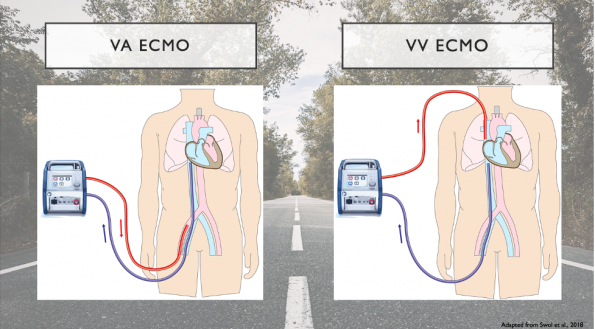
There are two main types of ECMO, which are differentiated by the location of insertion and type of cannulae used:
-
- Veno-arterial ECMO (VA-ECMO)
- Both arterial and venous systems are cannulized.
- Deoxygenated blood is drained from the venous system where it enters the ECMO circulation to be oxygenated, rid of COD and warmed up. The blood is then return to the body’s arterial circulation.
- This type of ECMO provides both respiratory and hemodynamic support.
- Veno-venous ECMO (VV-ECMO)
- Both cannulae are connected to veins/the venous system.
- As shown in the image above, blood is drained from the patients right femoral vein where it is circulated to our ECMO circuit where it is oxygenated, rid of CO2 and rewarmed. The blood is then returned to the patient’s venous circulation via the right internal jugular vein.
- This form of ECMO only provides respiratory support.
- Veno-arterial ECMO (VA-ECMO)
ECMO Indications
These are highly dependent on the organ we are trying to support.
-
- For CARDIAC support, we used VA-ECMO
- The main indication for that relevant to us as emergency physicians is severe cardiogenic shock of diverse etiology, including ACS.
- For RESPIRATORY support, we use VV-ECMO
- For CARDIAC support, we used VA-ECMO
ECMO Contraindications
Like with many other medical therapies, there are absolute and relative contraindications to the use of ECMO. These are summarized in the table below:
| ABSOLUTE | RELATIVE |
|
|
ECMO Complications
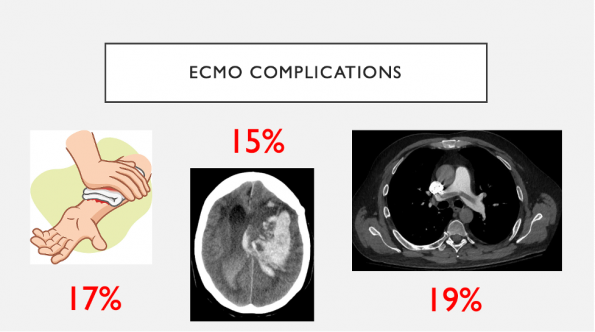
ECMO is not free of complications:
-
- VV- has fewer complications compared to VA-ECMO
- The commonest complication is hemorrhage from the cannulation site which occurs in up to 17% of patients on VV-ECMO and almost double that in VA-ECMO
- There is also a 15% rate of intracerebral hemorrhage
- Blood clots secondary to thrombus formation within the ECMO circuit occurs in 19% of patients
- Other complications include vessel perforation, arterial dissection, distal limb ischemia, and heparin induced thrombocytopenia
Given that ECMO is not a benign intervention, let’s examine the evidence for its use.
Evidence for ECMO Use
In this section, we will be looking at the evidence for the use of ECMO in specific situations: Acute Respiratory Distress Syndrome (ARDS), COVID-19, Hypothermic Arrest and Out-Of-Hospital Cardiac Arrest (OHCA).
ARDS
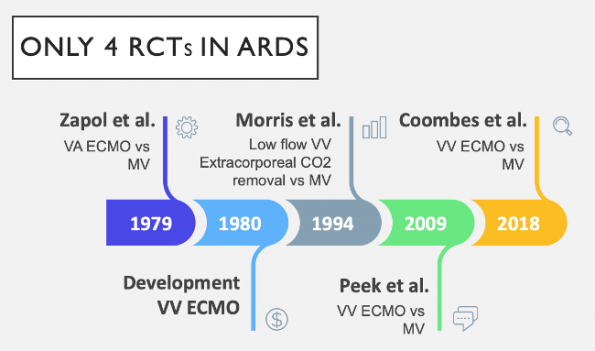
The benefit of ECMO in severe ARDS has long been debated. Because of highly challenging design and conduct issues, there have only been 4 RCTs performed over the past 50 years. To complicate the situation further, there have been advances in the fields ECMO circuits and mechanical ventilation (MV) engineering, as well as ICU care. These factors have led to conflicting overall evidence.
The first two RCTs (Zapol et al., 1979; Morris et al., 1994) did not show any survival benefit for the use of ECMO in ARDS.
-
- Limitations:
- Patients in the ECMO groups experienced large amounts of blood loss per day – on average of 2.7L
- Groups were exposed to high pressure non lung protective ventilation measures.
- Limitations:
CESAR TRIAL (Peek et al., 2009)
-
- UK based multicenter trial that aimed to delineate the safety and clinical efficacy of ECMO with MV compared with MV alone in adult patients with severe ARDS.
- Study Design: 180 patients were taken from 68 centers (population) and randomly assigned in a 1:1 ratio to receive either VV-ECMO and MV (intervention) or MV alone (control).
- Primary Outcome: death or severe disability at 6 months (outcome).
- Patient Allocation: 90 patients were assigned for consideration for ECMO and 90 were assigned to receive conventional management.
- A total of 68 patients received ECMO.
- Results: They found that 63% fo patients allocated to consideration for ECMO treatment compared to 47% of those allocated to conventional management survived to 6 months without disability.
- Conclusion: ECMO vs MV significantly improves survival without severe disability
- Limitations:
- No standardized MV protocol in the control group
- Only 70% of patients in the control group vs 95% in the ECMO group received lung protective ventilation measures
- Only 75% of patients randomized to the ECMO group received ECMO
EOLIA Trial (Coombes et al., 2018)
-
- This was an international multicenter RCT
- Study Design: Adult patients with severe ARDS (population) were randomized to receive VV-ECMO and MV (intervention) or conventional treatment with MV (control)
- Primary outcome: 60-day mortality (outcome)
- This study differed from earlier RCTs in that:
- A standardized MV protocol for the control group was created
- Cross-over to the ECMO group was possible for patients with refractory hypoxemia
- Patient Allocation:
- 121 of 124 patients in the ECMO group received ECMO
- 125 patients randomized to the control group received MV of which 35 crossed over needing rescue ECMO
- Results:
- At 60 days, 35% of patients in the ECMO group compared to 46% in the control group died
- Mortality was reduced by 11% in the ECMO group, but this did not reach statistical significance (relative risk (RR), 0.76; 95% CU, 0.55-1.04; p = 0.09)
- Conclusion: Amongst patients with severe ARDS, 60-day mortality was not significantly lower with ECMO
- Limitations:
- 28% cross-over from the control group to the ECMO group due to refractory hypoxemia
- This potentially results in confound of the primary outcomes resulting in an underappreciation for the benefit of VV-ECMO use, making it hard to draw conclusions regarding the usefulness of ECMO in severe ARDS
As neither the CESAR nor the EOLIA trials were adequately powered to detect a reasonable survival difference between groups, Coombes and colleagues meta-analyzed individual data from these two trials. Their primary outcome was 90-day mortality.
-
- This demonstrated that ECMO had a 12% significant reduction in death without respiratory, cardiovascular, or neurological failure (RR, 0.75; 95% CI, 0.6-0.94; p = 0.013).
Expert Opinion: “All ARDS patients should get a good trial of lung protective ventilation, proning, and inhaled nitric oxide or Flonan before they are truly considered for ECMO” – Dr. Ariel Hendin, MD FRCPC (Emergency and Critical Care Medicine, The Ottawa Hospital, University of Ottawa)
What are we doing for ARDS in Ottawa?
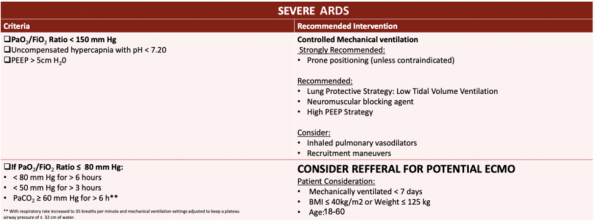
Current Ottawa Heart Institute (OHI) ECMO ARDS inclusion criteria.
Bottom Lines – ECMO in ARDS:
-
- ECMO has a role in ARDS in those with refractory hypoxaemia.
- Consider ECMO early in those failing conventional management.
- ECMO should only be performed in a centre that is well-equipped, has experience, and expertise and only on select patients.
COVID-19
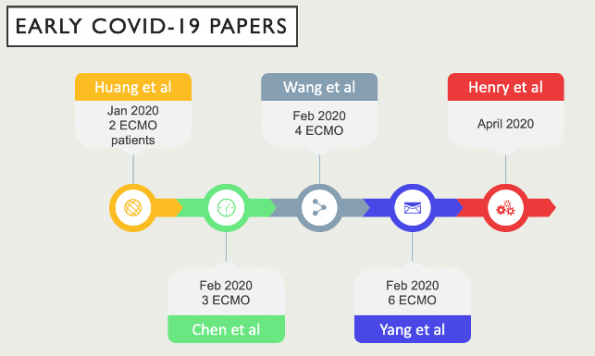
Since January 2020, many have extrapolated the use of ECMO to COVID-19 based upon previous experience with H1N1 and MERS. There have been multiple single center case reports and case series that have looked at the survival benefit of implementing ECMO. However, the overall data is sparse and of very low quality.
-
- Yang and Henry’s studies found higher rates of death amongst ARDS patients secondary to COVID-19 who received ECMO compared to those who only received conventional management.
- Difficult to draw conclusions from these studies as there are many confounding variables.
- Were death rates higher in the ECMO groups as there were innately sicker and were bound to have poorer outcomes and higher mortality rates or did ECMO cause direct harm to COVID patients?
Barbaro et al., 2020
-
- This was the first large, international cohort for ECMO in COVID-19
- Study Design: The study included patients aged 16 or older from 36 countries (population) with a confirmed diagnosis fo COVID-19, who had ECMO support (intervention)
- Primary Outcome: Death at 90 days after ECMO initiation (outcome)
- Results:
- Mortality rate of 37% in the entire COVID-19 cohort supported with ECMO
- A subset of patients with ARDS
- 38% mortality rate within 90 days of ECMO initiation
- No significant difference in survival rates
- Conclusions: Authors would support the use of ECMO in COVID-19
The WHO in May 2020, made the recommendation to ECMO should only be provided to patients with COVID-19 who remain in refractory hypoxemia despite our typical treatments and only in centres that have experience, staff and resources.
What is Ottawa’s Local Practice?
-
- The use of ECMO for COVID-19 is quite limited due to the questionable benefits of ECMO for ARDS in COVID.
- However, in order for it to be considered, a patient needs to have a PF ratio <150, be in a proned position, and remain in refractory hypoxemia or respiratory acidosis
Bottom Lines – ECMO in COVID-19
-
- Most evidence comes from coordinate observational studies
- One large international cohort study supports the use of ECMO in COVID-19
- However, the evidence is inconclusive and caution should be taken when interpreting these results
- At this time we need an RCT
Hypothermic Cardiac Arrest
A core body temperature below 32℃ increases the likelihood of cardiac arrest – a common outcome with accidental hypothermia. VA-EVMO has now become a viable option for patients with severe hypothermia presenting in cardiac arrest:
-
- Allows for rewarming to occur from a central to peripheral direction, thus warming the most vulnerable organs first
- Provides the ability to oxygenate blood externally, thus decreases patient dependence on ventilators and thus ventilator settings
- Allows for continued hemodynamic support after rewarming in patients who are hemodynamically unstable
Austin et al., 2020
-
- Systematic review from 2005 to 2019 that aimed to examine the use of ECMO for accidental hypothermia and its outcomes in adult patients
- They included 19 case reports and series consisting of 47 patients
- Found that 97% of patients that had a cardiac arrest, of which 58% had ventricular fibrillation as their initial rhythm
- 81% of patients survived, of which 19% experienced long-term cognitive impairment
- Survival was found to be significantly associated with three factors
- Potassium on presentation
- Initial body temperature, and
- ECMO rewarming rate
- Survival was found to be significantly associated with three factors
- Conclusion: ECMO is a viable cardiac support option for rewarming patients with accidental hypothermia with acceptable outcomes.
- Strengths:
- The strengths of this SR was this it only included studies from 2005 onwards, which limits the confounding nature of technological advancements and provides us with the most realistic conclusions.
- Compared to earlier systematic review, this study only looked at adults which reduces data heterogeneity
- Limitations:
- The biggest limitation of this study is its use of case reports and case series which are subject to large amounts of selection and publication bias
The Hope Score
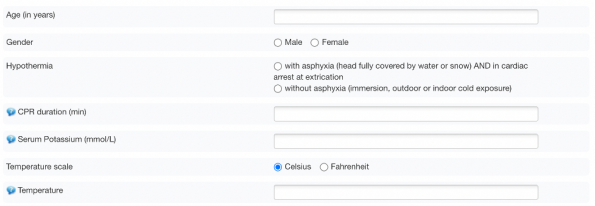
The HOPE score is a prediction score that determines the probability of survival following hypothermic cardiac arrest with ECMO resuscitation
-
- The score ranges from 0 to 100%
- The score uses 6 variables: age, gender, presence of asphyxiation, potassium level, CPR duration and temperature
- The score was derived in a large retrospective study of 237 patients (Pasquier et al., 2018)
- They found that the use of the HOPE score statistically and clinically improved survival probability to hospital discharge compared to dichotomous K levels
- A follow-up validation study was performed which replicated results (Pasquier et al., 2019)

This is the most up-to-date hypothermia protocol at The Ottawa Hospital. It further echoes the recommendation to consider ECMO in cases of severe refractory hypothermia.
Bottom Lines – ECMO in Hypothermic Cardiac Arrest
-
- ECMO should be provided for patients with hypothermic cardiac arrest
- As emergency physicians, we should initiate our initial management including passive and active methods of re-warming while simultaneously talking to CSICU and CV Surgery for ECMO consideration
- The HOPE score has shown to be more predictive of survival than using potassium alone in predicting mortality
Out-Of-Hospital Cardiac Arrest (OHCA)
We know that patients who suffer out-of-hospital cardiac arrest often do not respond to standard CPR and their mortality rates are almost 90%. There is growing interest in research on the use of ECPR in the management of refractory cardiac arrest. Like all other topics of ECMO, the evidence of ECPR is quite controversial.
An initial systematic review performed by ILCOR (Holmberg et al., 2018) found the results from individual studies to be very inconsistent; that all studies lacked a pre-registered protocol and had variable inclusion criteria; risk of bias to be critical; and the body of evidence to be low.
-
- Conclusion: There is inconsistent evidence to either support or refute the use of ECPR for OHCA.
Bougoin et al., 2020
-
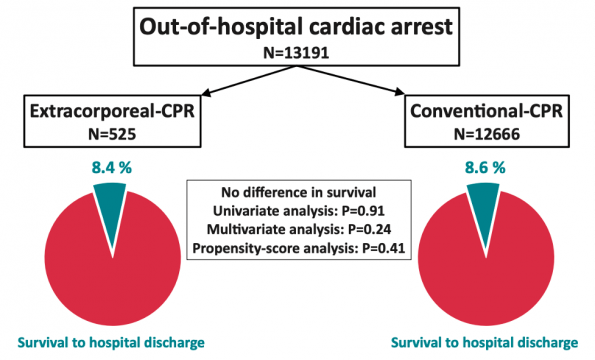
- Study Design: Aimed to compare outcomes of patients with refractory OHCA (population) from a multicenter Paris based registry, from 2011 to 2018, managed with ECPR (intervention) vs conventional CPR (control)
- Primary Outcome: survival to hospital discharge (outcome)
- In this cohort, the decision to perform ECPR was at the discretion of the emergency physician on board an ambulance or in the ED
- There were no clear guidelines in patient selection
- Moreover, cannulation could occur on scene, in the back of an ambulance or in the ED
-
- A total of 525 patients received ECPR and 12,666 got conventional CPR
- Overall hospital survival did not differ significantly between the two groups
- Conclusion: In a population-based registry, there was no difference between ECPR and conventional CPR in survival in refractory OHCA
- Limitations:
- There was no established ECPR protocol for selection of patients
- Essentially, this was done at the discretion of the emergency physician
- Potential amounts of bias as they would choose patients with seemingly better prognostics to undergo ECPR
- Additionally, when comparing the two groups, their baseline demographics, significantly differed
- The ECPR group consisted of more males, were more likely to receive bystander CPR, more likely to receive CPR for more than 30 minutes, and more likely to have an initial shockable rhythm
- There was no established ECPR protocol for selection of patients
- These results would likely not be applicable to a Canadian setting as the system of care in France is quite different
- They have a prehospital emergency physician on board the ambulance, 911 to scene times are often longer in Europe
- Rates of bystander CPR in France are about 59% compared to our 40% here in Ontario
- A total of 525 patients received ECPR and 12,666 got conventional CPR
ECMO initiation in the Louvre
Bartos et al., 2020
-
- Aimed to examine the effect of resuscitation duration on survival in patients undergoing ECPR for OHCA compared to conventional CPR
- Applied a strict inclusion criteria that was developed and currently being used by the Yannopoulous group in Minnesota
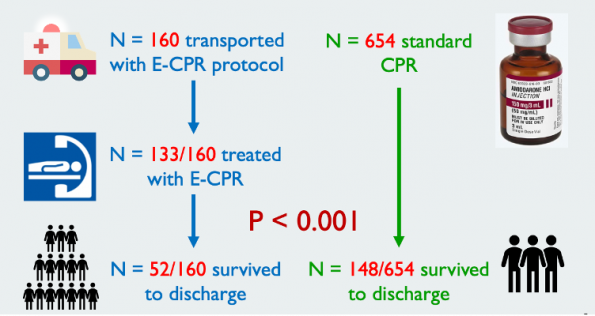
- The intervention group comprised of 160 adults with refractory OHCA with a shocked rhythm treated with the University of Minnesota ECPR protocol (described in Yannopoulous et al., 2016)
- This group was compared with 654 patients taken from the Amiodarone arm of the Amiodarone, Lidocaine or Placebo study
- Primary Outcome: Good neurological outcome at the time of discharge
-
- Results: 33% of those transported for ECPR compared to 23% from the ALPS cohort survived with favorable neurologic outcome (p<0.001)
- Conclusion: ECPR significantly improved hospital survival and more importantly with functional neurological outcome
- Strengths:
- Strict inclusion criteria for patient selection of patients
- Protocolized use of ECPR for adults with refractory OHCA
- Limitations:
- Single-center observational study
Then this led to the first and only RCT in OHCA which was recently published in the Lancet by Yannopoulous and colleagues.
Yannopoulous, et al., 2020

This was a phase 2, single center, open label, pragmatic RCT that aimed to compare survival to hospital discharge comparing ED standard ACLS resuscitation versus ECMO facilitated resuscitation. Of note, this study employed Bayesian statistics in that they used previous data to develop a probability of hospital discharge.
-
- Included patients:
- Adults aged 18-75 with out of hospital cardiac arrest;
- An initial shockable rhythm;
- No ROSC with three shocks;
- Able to fit in the Automated CPR compressor, and;
- An estimated transfer time to the ED shorter than 30 minutes.
- Included patients:
-
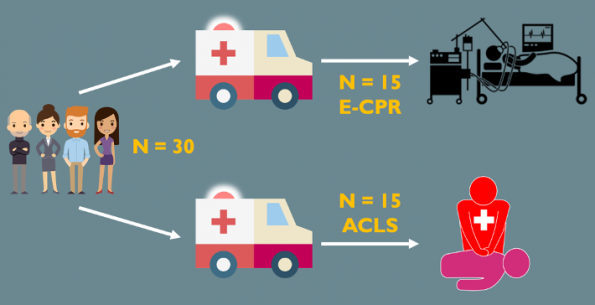
- Study Design: 30 patients were randomized to one of the two standards of care:
- 15 patients in the ECMO group were transferred directly to the Cath lab and underwent resuscitation with an interventional cardiologist.
- Of note, this paper was not clear as to how many interventionalists were involved or whether this was the sole working of Dr. Yannopoulous himself.
- 15 patients in the conventional CPR group were transferred to the ED and resuscitated by an emergency physician.
- 15 patients in the ECMO group were transferred directly to the Cath lab and underwent resuscitation with an interventional cardiologist.
- Primary outcome: Survival to Hospital Discharge. Secondary endpoints were survival and functionally favorable status at hospital discharge and at 3 and 6 months.
- Results:
- With respect to their primary outcome, they found that survival to hospital discharge was seen in 6 out of 14 patients in the ECMO group, versus 1 out of 15 patients in the standard ACLS arm. This was a risk difference of 36% that showed a posterior probability of ECMO superiority.
- With respect to their secondary outcomes, survival to 3 and 6 months was 43% for the ECPR group and 0% for the conventional ACLS group. All survivors had good functional assessment scores at 6 months.
- Conclusion: Survival for patients with refractory OHCA can be significantly increased by early implementation of ECMO
- Strengths:
- Strong methodology
- The Minnesota group has arguably the most well organized, experienced and funded ECMO system in the world.
- Its ECMO system has an extremely well developed prehospital and ECMO program with very experienced and highly trained staff who have performed ECMO on multiple patients in the past who are thus proficient with cannulations
- Moreover, their results further reinforce the importance of better patient selection and a need for a protocolled use of ECPR in out of hospital cardiac arrest
- Limitations:
- Small sample size of 30 patients
- This was also a single center study open label study
- Additionally, I do question the external validity of this study due to the way in which their program is set-up. I do think these results would be applicable to a Canadian center right now. And it would be interesting to see what a multicenter RCT would show.
- Study Design: 30 patients were randomized to one of the two standards of care:
Hsu et al., 2021
The Extracorporeal Cardiopulmonary Resuscitation for Refractory Out-Of-Hospital Cardiac Arrest (EROCA) pilot RCT aimed to assess the feasibly of hitting critical time periods from OHCA to ED arrival and from ED arrival to initiation of ECMO.
-
- They found that out of all patients randomized to the ECPR group, only 42% had a 911 call to ED arrival time less than or equal to 30 minutes.
- In those who were eligible for ECPR, only 60% had a door to ECPR initiation of less than or equal to 30 minutes
- The authors concluded that the EROCA trial did not meet predefined feasibility outcomes for selecting OHCA patients for expedited transport and initiation of ECPR in the ED
- What the EROCA trial taught us was that it is hard to replicate the findings from the ARREST trial as these centers lack the strong developed protocol that is well established in Minnesota
- The results also call into question the generalizability of the results from the ARREST trial
Expert Opinion: “The findings of the ARREST trial are highly compelling and echo with identical numbers other prominent papers [CHEER trial] that have been published in the western world with very well developed ECMO systems and protocols…an RCT on ECPR in OHCA is needed…until then, I would advocate for the use of ECPR on the right patient” – Dr. George Mastoras, MD FRCPC (Emergency Medicine, University Health Network, University of Toronto)
Expert Opinion: “Many places will try and replicate the findings from the ARREST trial but will all fail as there are numerous limiting factors such as having a well-developed EMS system and ECMO protocols and funding…” – Dr. Sheldon Cheskes, MD CCFP-EM (Emergency Medicine, Sunnybrook Health Sciences Center, University of Toronto)
What is Our Local Practice?
-
- In the process of a Tiered roll out for ECPR (more on this below)
- OHI is not currently offering ECPR in cases of OHCA
Bottom Lines: ECMO in OHCA
-
- ECPR can be considered in OHCA when flow can be initiated within 60 minutes from cardiac arrest
- Should only be performed in wisely selected patients in a center with an established ECPR program, protocol, experience and expertise
Part 2: Implementation of an ED ECMO Program
In this section, we will discuss the factors that are needed to implement an ED ECMO program and what happens when a code ECPR is called at St. Paul’s hospital in Vancouver which is currently the only ED ECMO center in Canada.
Expert Opinion:There are many necessary components to a successful ED ECMO program. There needs to be a very well-developed pre-hospital system with strict inclusion and exclusion criteria that paramedics can apply. There also needs to be a number of staff who able to provide ECMO. ECMO provides also need proficiency and expertise. – Dr. Brian Grunau, MD MHSc (Emergency Medicine, St. Paul’s Hospital, University of British Columbia)
Development of a Pre-Hospital System
-
- The design of this system depends on what prehospital system we currently have in place
- Most high performing systems, such as our system in Ottawa use a stay and play paradigm vs a scoop and run paradigm
- If we want to add on ECPR into our system, we need to implement a scoop and run strategy
- This then necessitates the development of protocols that outline strict inclusion and exclusion crieria
- We then need to train paramedics to identify cases that meet inclusion for ECPR so that we are not altering care for patients who are not going to get treatment with ECPR
- We then need to implement strategies for rapid transport of these patients
- We know in literature that scoop and run paradigms have poorer resuscitation quality, poorer CPR fundamentals which then lends to poorer overall outcomes. So we need to ensure good cardiac resuscitation with a focus on fundamentals during extrication and transport
- Role fo simulations
What happens when a “Code ECPR” is called at St. Paul’s Hospital?
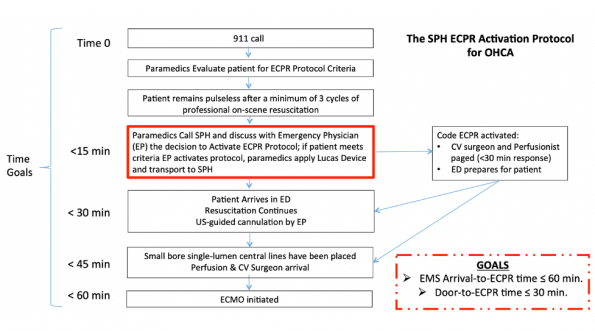
Above is a timeline of the events that occur when a 911 call is received to ECMO initiation at St. Paul’s Hospital in Vancouver. There are two overall time goals:
-
- 911 call to ECPR time of 60 minutes;
- Door to ECPR time of 30 minutes.
Time 0 here is when the 911 call comes in as it provides the best estimate of the time of cardiac arrest. Paramedics will then see if the patient meets specific inclusion criteria (shown below), while continuing on scene resuscitation.

If paramedics deem that the patient meets inclusion criteria, they then call into St. Paul’s to discuss the case with the Emergency Physician (EP).
If the EP is in agreement that the patient meets criteria for ECPR, a code ECPR is called:
-
- This code calls in two teams – a cannulation team and a resuscitation team;
- These two teams work independently of one another;
- The sole purpose of the cannulation team is to gain access and start the ECMO circuit;
- The resuscitation team’s main priority is continuing ACLS care;
- These two teams present to the resuscitation bay and set up prior to the arrival of the patient.

On patient arrival, the patient is then rapidly screened again for additional ECPR inclusion.
-
- While screening is taking place, compressions are continued via the LUCAS device, the cannulation team starts to gain vascular access and the resuscitation team continues typical ACLS level care.
- Once these criteria are confirmed the patient is then set up to the circuit.
Currently, at St. Paul’s hospital, their median 911 to ECMO initiation time is about 60-65 minutes and their door to ECMO initiation time is 18 minutes. These times are not bad but are not comparable to the times shown from the Minnesota group.
Expert Opinion: “In order for an ECPR program to be successful like the Yannopoulous program in Minnesota, the program has to be extremely mature and developed in all realms from prehospital care to ED and Cath lab care to ICU care…Only once these are established and we have our reps in critical care experience can we be proficient at ECPR and navigate issues as they arise…once these fundamental aspects are solid, we may be able to replicate the results from the ARREST trial and that ECPR may be the future of cardiac arrest care…” – Dr. George Mastoras, MD FRCPC (Emergency Medicine, University Health Network, University of Toronto)
Part 4: Future Directions at TOH
This brings us to the last section of this blog post. Here, we are going to take a look at what ED ECMO might look like at TOH.
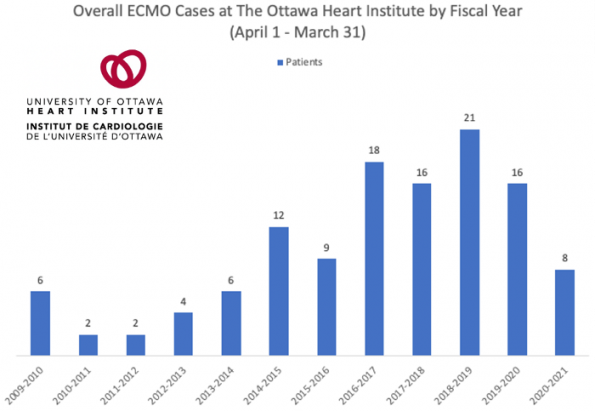
Over the last decade, there has been approximately 114 cases of ECMO. The breakdown of the underlying disease process in these cases is unclear. However, there has been a gradual increase in the number of cases yearly. Even then in Ottawa we currently do not have the ECMO volume compared to other major centers in Canada.
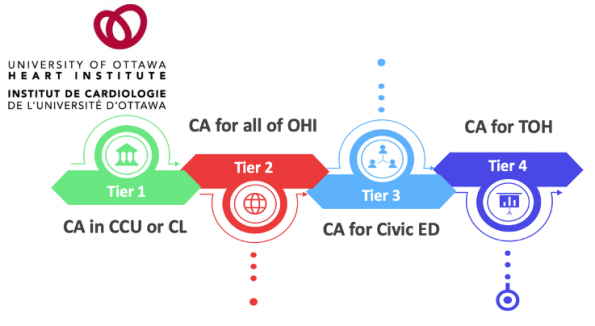
In Ottawa, we are working towards developing this maturity and experience. The Ottawa Heart Institute (OHI) led by Dr. Brock Wilson, currently has a tiered roll out system for ECPR.
-
- Tier 1: Perform ECPR for IHCA that occurs only in the CCU or Cath lab at the OHI.
- Tier 2: Provide ECPR for all IHCA in OHI. This is where we are at now.
- Tier 3: Expand ECPR for cardiac arrests that occur in all of the OHI and in the Civic ED.
- Tier 4: Include ECPR for cardiac arrests to both the civic and general campuses.
Having a roll out like this, will allow us to increase our experience with ECPR, establish expertise and to establish a solid ECPR protocol.
Moreover, there is discussion between a member of the ED Resuscitation Committee about turning the Civic ED into the Cardiac arrest center. Should this occur, we would potentially take all cardiac arrests including those with non-shockable rhythms. To develop this would require a protocol for an EMS bypass to the Civic ED. Once this happens our volumes would increase, and this may give us the necessary exposure we need.
Overall, it will be years until we obtain the necessary volume, exposure, and development of strong prehospital and ECMO protocols for ECPR. We should not expect to see ECPR program landing in an Ontario ED anytime soon.
Expert Opinion: Both Dr. Christian Vaillancourt, an emergency physician and cardiac arrest researcher in Ottawa and Dr. Sheldon Cheskes, believes that it may be higher yield to focus on the fundamentals in improving cardiac arrest survival compared to developing an ED ECMO program. These would include improving rates of bystander CPR, improving rates of bystander AED use, and measuring CPR quality. Both are adamant that all these measures would improve survival far greater than any ECMO program would, assuming it would first be successful. Additionally, the cost of an ECMO program would far exceed community education measures.
Overall Take-Home Messages
-
- ECMO has a role in ARDS in those with refractory hypoxemia and should be considered early on in those patients who are failing conventional management.
- ECMO for COVID-19 is quite limited due to the questionable benefits and inconclusive evidence of ECMO for ARDS in COVID. It should be considered on a case-by-case basis.
- ECMO should be provided for patients with hypothermic cardiac arrest and as EPs on the ground floor, we should initiate our therapies and get early involvement of our colleagues in CSICU and CV surgery.
- ECPR can be considered in OHCA when flow can be initiated within 60 minutes from cardiac arrest.
- For all cases of ECMO, consideration should be placed only for wisely selected patients in a center with an established ECMO program, protocol, experience and expertise
- ED ECMO may be the future of cardiac arrest care, however, we should not expect it in our EDs anytime soon. In the meantime, while we gain critical care experience in ECMO, we should focus on improving fundamentals of cardiac arrest care.
References
- Austin et al., 2020. Outcomes of Extracorporeal Life Support Use in accidental Hypothermia: A Systematic Review. Ann Thoracic Surgery; 110:1926-32
- Barbaro et al., 2020. Extracorporeal membrane oxygenation support in COVID-19: an international cohort study of the extracorporeal life support organization registry. Lancet.
- Bartos et al., 2020. Improve survival with extracorporeal cardiopulmonary resuscitation despite progressive metabolic derangement associated with prolonged resuscitation. Circulation.
- Bougouin et al., 2020. Extra corporeal cardiopulmonary resuscitation in out-of-hospital cardiac arrest: a registry study. European Heart Journal.
- Coombes et al., 2018. Extracorporeal Membrane Oxygenation for Severe Acute Respiratory Distress Syndrome. NEJM
- Coombes et al., 2020. ECMO for Severe ARDS: Systematic Review and Individual Patient Data Meta-analysis. Intensive Care Med.
- Henry., 2020. COVID-10, ECMO, and lymphopenia: a word of caution. Lancet.
- Holmberg et al., 2018. Extracorporeal cardiopulmonary resuscitation for a cardiac arrest: a systematic review. Resuscitation.
- Hsu et al., In press. Extracorporeal cardiopulmonary resuscitation for refractory out-of-hospital cardiac arrest: results of a randomized feasibility trial of expedited out-of-hospital transport. Annals of Emergency Medicine.
- Makdisi and Wang., 2015. Extracorporeal Membrane Oxygenation (ECMO) Review of a Lifesaving Technology. J Thoracic Disl; 7(7):E166-176
- Pasquier et al., 2018. Hypothermia outcome prediction after extra orporeal life-support for hypothermic cardiac arrest patient: The HOPE score. Resuscitation.
- Pasquier et al., 2019. Hypothermia outcome prediction after extracorporeal life-support for hypothermia cardiac arrest patients: An external validation of the HOPE score. Resuscitation.
- Peek et al., 2009. Efficiency and economic assessment of conventional ventilatory support versus extracorporeal membrane oxygenation for severe adult respiratory failure (CESAR): a multicentre randomized controlled trial. Lancet.
- Squires at al., 2016. Contemporary extracorporeal membrane oxygenation therapy in adults: Fundamental principles and systematic review of the evidence. J Thoracic and Cardiovascular Surgery; 152(1):20-31
- Stub et al., 2015. Refractory cardiac arrest treated with mechanical CPR, hypothermia, ECMO and early reperfusion (the CHEER trial). Resuscitation.
- Swol et al., 2018. Extracorporeal Life Support in the Emergency Department: A Narrative Review for the Emergency Physician. Resuscitation
- Yang et al., 2020. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study. Lancet
- Yannopoulous et al., 2020. Advanced reperfusion strategies for patients with out-of-hospital cardiac arrest and refractory ventricular fibrillation (ARREST): phase 2, single centre, open-label, randomized controlled trial. Lancet.
- World Health Organization (WHO). Interim guidance May 27, 2020. Clinical management of COVID-19.
- Zapol et al., 1979. Extracorporeal Membrane Oxygenation in Severe Acute Respiratory Failure. JAMA