In part 1 of this series, we focused on the physiologic and hemodynamic consequences of aortic stenosis, as well as management in atrial fibrillation and procedural sedation. In this part, we will focus on the management of decompensated aortic stenosis with heart failure, and the approach to PoCUS.
To remind you of the case we began with; Mrs. Smith is a 72F who has a hx of smoking. Her family physician prescribed her a puffer last year when she developed mild dyspnea. She presents to your ED with fatigue and breathlessness on exertion. Labs are normal, CXR is normal. Her lungs sound decreased throughout. The ED is loud, but you think you heard a murmur. You treat her for a COPD exacerbation (the same as on her last 2 visits), and you send her home. But what if you’ve missed the diagnosis of Severe Aortic Stenosis? The consequences of this could be fatal.
Decompensated Aortic Stenosis – Heart Failure and Cardiogenic Shock
Decompensated aortic stenosis will usually present in the form of acute decompensated heart failure and/or cardiogenic shock. Hospital mortality from cardiogenic shock in the setting of severe aortic stenosis has been previously estimated at 70% (11).
Despite this, our knowledge on the acute management of HF and CS in the context of AS is limited due to a paucity of evidence. No current guidelines on this topic exist, and a clear-cut strategy has yet to be defined. Therefore, the recommendations I present to you here are based primarily on expert opinion in combination with small case series and retrospective analyses. I think we can all agree that encountering a decompensated aortic stenosis patient in the ED would be intimidating, to say the least. Today I’m hoping you walk away with a reasonable, safe approach. Much of the below information was informed by a 2021 analytic review, written by a cardiac intensivist at the Mayo Clinic (10). It is one of the most up-to-date guides for the management of the various structural heart disease emergencies.
One important thing to note – there are classic teachings in the management of AS that may not be standing up to the test of time. Inotropes, diuretics, vasodilators and PPV all remain controversial, but may have a role to play depending on the clinical presentation in front of you. We’ll come back to these.
So, when faced with the patient with decompensated aortic stenosis:
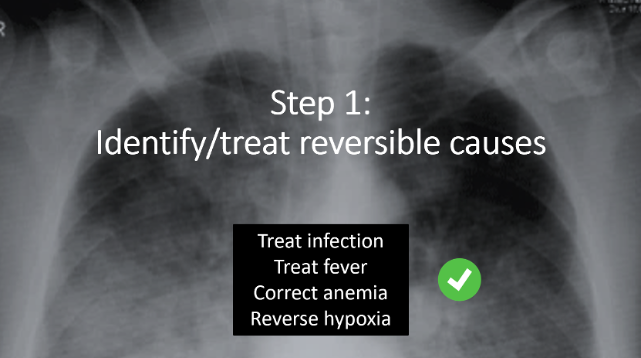
- Step 1: Identify and treat reversible causes. One of the most crucial considerations is whether the patient is sick from their severe AS (ie, this is just progression of their valvular disease), or sick with their AS (as in, there is a superimposed illness that is tipping their AS over the edge). Identification of a reversible cause in a patient who is sick WITH AS will be key to the resuscitation – remember to treat the common things that can add O2 demand to the vulnerable left ventricle. So: treat infection, treat fever, correct anemia, reverse hypoxia.
- Step 2: Optimize preload. Remember, maintaining preload is the GOLDEN RULE of Aortic Stenosis. To do this safely, perform a volume assessment. Is the patient wet, or is the patient dry? If there is no obvious pulmonary edema, start with a 250-500 cc IVF bolus and repeat as needed. Of course, tipping the patient over into pulmonary edema will not be helpful – so careful reassessment of volume status between boluses will be required.
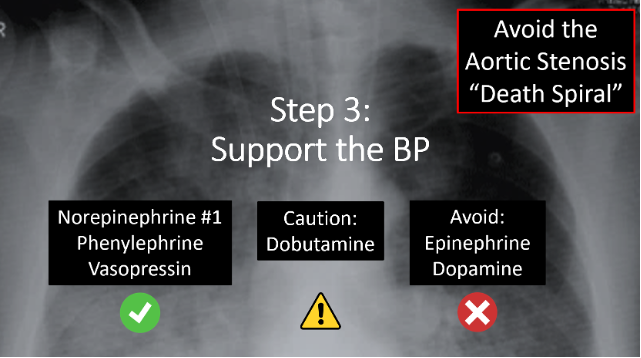
- Step 3: Support the blood pressure. Remember that hypotension in AS causes low coronary perfusion, which worsens myocardial ischemia and dysfunction, further impairing forward flow. This may trigger a dangerous downward hemodynamic spiral, colloquially known as the “Aortic Stenosis Death Spiral”. Recovery from this is challenging, and a small retrospective cohort study published in “Critical Care” by Brunauer in 2014 found that critically ill patients with severe aortic stenosis suffered from terminal cardiovascular collapse at higher blood pressures compared to critically ill patients without severe AS, supporting the idea that hypotension must be avoided in patients with severe AS, as they may become irreparably unstable at a higher BP than the average patient (12).
- In the hypotensive, decompensated aortic stenosis patient, reach first for norepinephrine. Norepinephrine will provide both alpha and beta-1 activation, giving it a theoretical advantage over pure alpha-agents.
- But recall our discussion from earlier around vasopressor choice in AS. Any of these three drugs (norepinephrine, vasopressin, phenylephrine) is reasonable. It can be helpful to consider the patient’s physiology in front of you and use the agent that makes the most sense in the given clinical scenario.
- Note: I recognize it may actually be counter-intuitive here to treat AS with vasopressors, considering the stenotic valve is resulting in LVOT obstruction, and increasing systemic vascular resistance (or squeezing the pipes after the ventricle) only seemingly adds to this afterload. But remember the #1 goal here is to maintain coronary perfusion to avoid the death spiral. With that being said, a good rule of thumb is to aim to use the lowest effective dose, to avoid excess afterload.
- Next, inotropy (specifically dobutamine) may be helpful in severe decompensated AS, but it may also cause harm. The greatest utility is in patients with LV systolic dysfunction (remember not all AS patients have a low EF), and the greatest harm will be in the risk of tachycardia and arrhythmia. The literature suggests the use of a PA catheter to guide this decision – so from an ED perspective, I would recommend early ICU/cardiology involvement prior to initiating inotropes.
- And finally, avoid epinephrine and dopamine due to increased myocardial O2 demand and tachycardia.
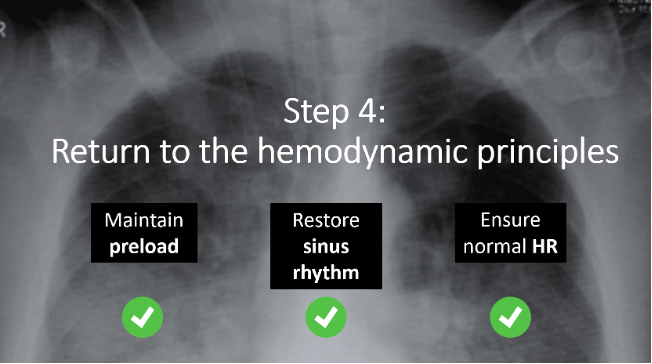
- Step 4: If your patient is still unstable, revisit the hemodynamic principles of Aortic Stenosis we discussed in part one.
- Think back to our earlier discussion on atrial fibrillation. If rapid a.fib is at play, this is the time to consider cardioversion for the patient who is persistently unstable.
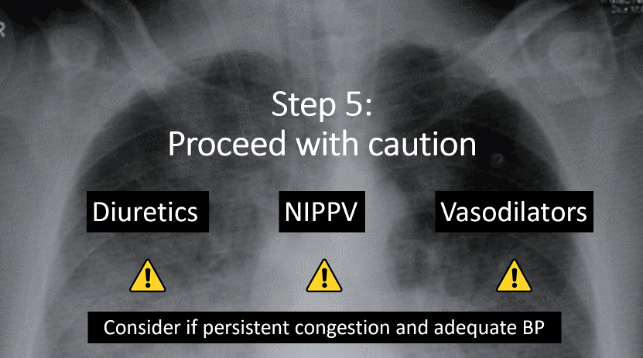
- Step 5: Proceed with caution. For everything else we are about to discuss in the acute management of decompensated AS with heart failure and cardiogenic shock, there is controversy. The safety and utility of the following interventions is still unclear. This is where this disease gets particularly tricky. Recall the beginning of this post, when I alluded to the many subclassifications of severe AS – it is likely within these nuances that the difference in hemodynamic presentation and response to certain therapies lie. Understanding this is beyond the scope of this post. As Emergency Physicians, our job is to understand that some patients with severe AS will tolerate these classic therapies for heart failure, and some will not.
- Though controversial, cautious use of diuretics and PPV may safely improve pulmonary congestion in the AS patient that is not frankly hypotensive. Intubation should be avoided, if possible (10).
- When it comes to vasodilators, however, the standard teaching is that these are dangerous in AS because they can lead to hypotension and low coronary perfusion and they should therefore be universally avoided. However, an attempt to find any evidence to support this theory comes up short. More recently, it has been hypothesized that high systemic vascular resistance has an additive effect on the stenotic valve, which in turn worsens LV afterload, increases myocardial O2 demand, and ultimately further impedes cardiac output.
- A study by Khot et al. was published in the New England Journal of Medicine in 2003 (13). They investigated the use of nitroprusside, a potent vasodilator, in critically ill patients with congestive heart failure, LV systolic dysfunction, and severe aortic stenosis.
- The only exclusion criteria was hypotension, defined as a MAP < 60.
- They found, contrary to classic teaching, in patients with ADHF in severe AS but preserved BP, nitroprusside improved cardiac output.
- Since then, more evidence has been published in support of vasodilator use in severe AS:
- Two studies assessed the hemodynamic response to Nitroprusside infusion in patients with severe AS while undergoing cardiac catheterization (14,15). Hypotensive patients were again excluded. There were no adverse events, and in particular, no episodes of ischemia.
- In 2015, Claveau et al. published a retrospective chart review in two Canadian EDs that found that nitroglycerin use in patients with moderate and severe aortic stenosis was not associated with more clinically relevant hypotension than in patients without AS (16).
- These studies have challenged the classic teaching around avoiding afterload reduction in aortic stenosis. In fact, they support the idea that severe AS with preserved blood pressure may in fact tolerate vasodilatory treatment with improvement in hemodynamic markers.
- A study by Khot et al. was published in the New England Journal of Medicine in 2003 (13). They investigated the use of nitroprusside, a potent vasodilator, in critically ill patients with congestive heart failure, LV systolic dysfunction, and severe aortic stenosis.
- So, to conclude our algorithm on the management of decompensated AS, yes, cautious use of these classic HF therapies can be considered if there is persistent pulmonary congestion, and the patient is not hypotensive.
Definitive Treatment for Symptomatic Aortic Stenosis
So, what’s the next steps for these decompensated patients with severe aortic stenosis? Rapid clinical deterioration may necessitate urgent invasive intervention. This may come in the form of mechanical circulatory support or catheter interventions. Surgical AVR is indicated, but will often not be suitable in these unstable patients.
Above all, the best outcomes for patients with AS presenting with an acute valvular emergency remains in the prompt recognition of the condition with appropriate stabilization and early consultation, and that is our job in the ED.
Point of Care Ultrasound in Aortic Stenosis
Okay, let’s close this post with a discussion on PoCUS. Let’s say an elderly patient comes into your ED in undifferentiated shock. This time, you don’t know that he has severe aortic stenosis – the patient doesn’t even know that he has aortic stenosis.
Although less common than hemodynamic decompensation due to acute coronary syndrome or cardiomyopathy, valvular emergencies should be on your differential in patients with acute-onset heart failure or cardiogenic shock. AS, in particular, should come to mind in patients who are elderly.
In the undifferentiated patient, in addition to our astute physical exam, we are lucky to have another tool in the ED that can point us to a diagnosis of severe AS when little background info is available: point of care echo.
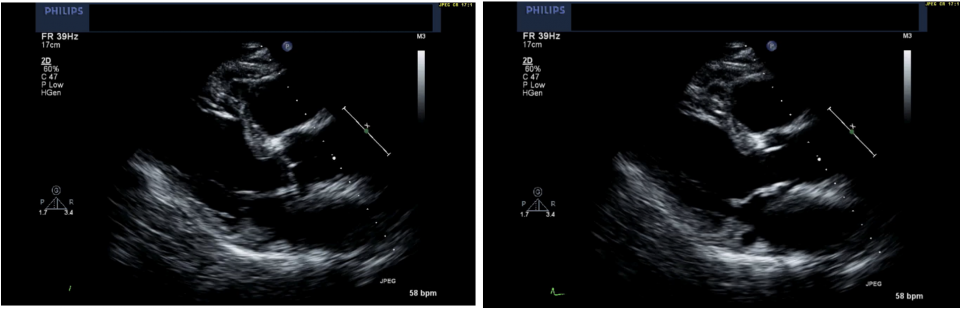
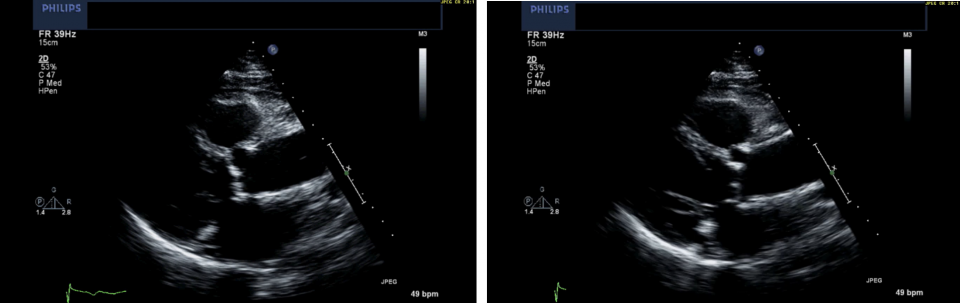
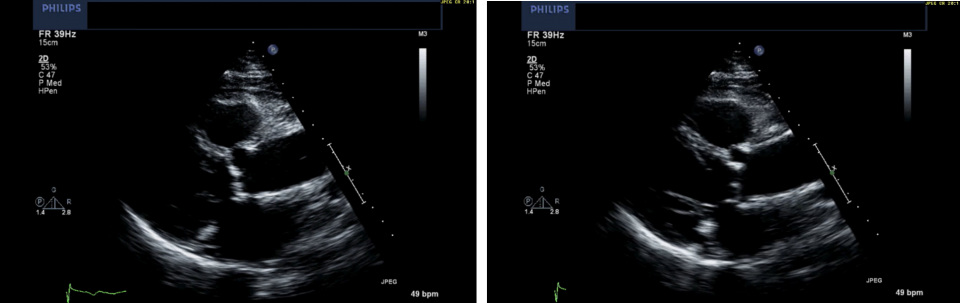
If you’re obtaining parasternal long axis (PSLA) and short axis (PSSA) views in most of your sick patients already, I’m here today to tell you that screening for severe AS is a relatively easy thing to incorporate into your practice. Especially in the long axis view, you’re already doing the work. You just need to train your eyes to pay attention to the centre of your image. Take a look below. All of the images used here were published in a 2015 article by Alzahrani et al., and you can refer back to them for the full videos (17).
In the PSLA view, bring your attention to the aortic valve in the centre of the screen. In the two images below, we see a normal aortic valve with thin cusps that look like two hands in prayer when the valve closes. There is no heavy calcification on this valve, and the cusps are opening well.
In comparison, the two images below depict non-severe AS. At the centre of your screen, you see bright, bulky calcification of the aortic valve, with notably reduced opening of the bottom cusp, while the top cusp is still demonstrating reasonable movement.
Severe AS is captured in the images below. At the centre of your screen, you see that bright, bulky calcification of the aortic valve, now with markedly reduced opening of both cusps. In addition, the LV wall appears thickened, and the left atrium is dilated.
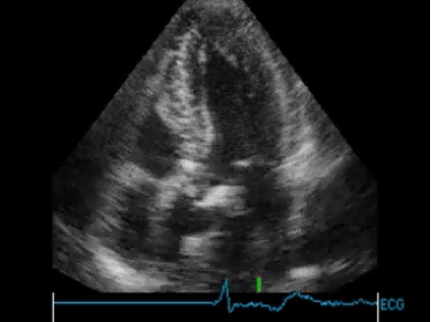
To see the aortic valve in your PSSA view, a minor adjustment is required. Drop your probe angle (sweeping anteriorly) until the circular valve comes into view. Check out a great video on obtaining this view here.
Finally, you can also obtain an apical 5-chamber view to assess the aortic valve – severe AS is shown below.
Now, is it feasible for ED docs to use PoCUS to detect severe AS? A small body of evidence suggests yes, it is. A retrospective case control records review of 60 patients who had a complete echocardiogram found that standard Echo views can be interpreted by Emergency Physicians with appropriate training to identify severe AS with good specificity (17).
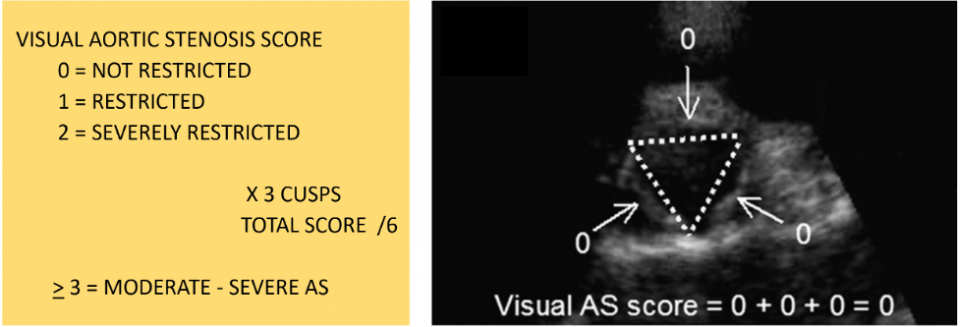
Since then, a team of Cardiologists in Japan has published a series of trials looking at the accuracy of PoCUS in the detection of clinically-relevant AS (18–20). They created a “Visual AS Score”, summarized below.
In their third study from 2021, real-life patients were included if they presented to the ED with symptoms possibly attributed to a cardiac cause. Emergency Physicians performed standard focused cardiac ultrasound, but they also calculated a Visual AS Score for each patient. All patients went on to have comprehensive echos by expert sonographers. They found that emergency physicians and expert sonographers had excellent inter-rater agreement, and a score of 3 or higher on the Visual AS Score performed very well in screening for moderate-severe AS.
Summary
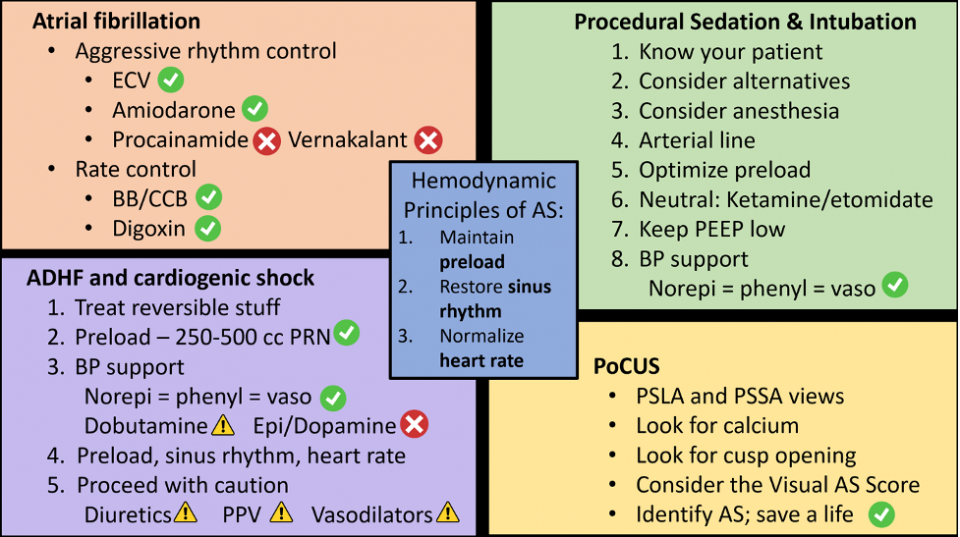
So, here we talked about the three hemodynamic principles of aortic stenosis (optimize preload, restore sinus rhythm, and normalize the heart rate), how to care for ED patients with atrial fibrillation in aortic stenosis, a safe approach to procedural sedation and intubation in aortic stenosis, a reasonable approach to the AS patient presenting with heart failure and shock, and the use of PoCUS in the detection of severe aortic stenosis. I hope you can use these skills to identify and stabilize this dangerous condition, and ultimately, save a life.
References
- Otto CM, Nishimura RA, Bonow RO, Carabello BA, Erwin JP, Gentile F, et al. 2020 ACC/AHA Guideline for the Management of Patients With Valvular Heart Disease: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2021 Feb 2;143(5):e72–227.
- Vahanian A, Beyersdorf F, Praz F, Milojevic M, Baldus S, Bauersachs J, et al. 2021 ESC/EACTS Guidelines for the management of valvular heart disease: Developed by the Task Force for the management of valvular heart disease of the European Society of Cardiology (ESC) and the European Association for Cardio-Thoracic Surgery (EACTS). Eur Heart J. 2022 Feb 14;43(7):561–632.
- Stortecky S, Buellesfeld L, Wenaweser P, Heg D, Pilgrim T, Khattab AA, et al. Atrial Fibrillation and Aortic Stenosis. Circ Cardiovasc Interv. 2013 Feb;6(1):77–84.
- Kubala M, de Chillou C, Bohbot Y, Lancellotti P, Enriquez-Sarano M, Tribouilloy C. Arrhythmias in Patients With Valvular Heart Disease: Gaps in Knowledge and the Way Forward. Front Cardiovasc Med [Internet]. 2022 [cited 2023 Nov 10];9. Available from: https://www.frontiersin.org/articles/10.3389/fcvm.2022.792559
- Stiell IG, de Wit K, Scheuermeyer FX, Vadeboncoeur A, Angaran P, Eagles D, et al. 2021 CAEP Acute Atrial Fibrillation/Flutter Best Practices Checklist. Can J Emerg Med. 2021 Sep 1;23(5):604–10.
- Andrade JG, Aguilar M, Atzema C, Bell A, Cairns JA, Cheung CC, et al. The 2020 Canadian Cardiovascular Society/Canadian Heart Rhythm Society Comprehensive Guidelines for the Management of Atrial Fibrillation. Can J Cardiol. 2020 Dec;36(12):1847–948.
- Darby AE, DiMarco JP. Management of Atrial Fibrillation in Patients With Structural Heart Disease. Circulation. 2012 Feb 21;125(7):945–57.
- Pascale King AM. Atrial Fibrillation/Flutter Best Practice Checklist [Internet]. EMOttawa Blog. 2022 [cited 2023 Nov 10]. Available from: https://emottawablog.com/2022/04/atrial-fibrillation-flutter-best-practice-checklist/
- van Diepen S, Katz JN, Albert NM, Henry TD, Jacobs AK, Kapur NK, et al. Contemporary Management of Cardiogenic Shock: A Scientific Statement From the American Heart Association. Circulation. 2017 Oct 17;136(16):e232–68.
- Jentzer JC, Ternus B, Eleid M, Rihal C. Structural Heart Disease Emergencies. J Intensive Care Med. 2021 Sep 1;36(9):975–88.
- Buchwald AB, Meyer T, Scholz K, Unterberg C, Schorn B. Efficacy of balloon valvuloplasty in patients with critical aortic stenosis and cardiogenic shock—the role of shock duration. Clin Cardiol. 2001;24(3):214–8.
- Brunauer A, Koköfer A, Bataar O, Gradwohl-Matis I, Dankl D, Dünser MW. The arterial blood pressure associated with terminal cardiovascular collapse in critically ill patients: a retrospective cohort study. Crit Care. 2014;18(6):719.
- Khot UN, Novaro GM, Popović ZB, Mills RM, Thomas JD, Tuzcu EM, et al. Nitroprusside in Critically Ill Patients with Left Ventricular Dysfunction and Aortic Stenosis. N Engl J Med. 2003 May 1;348(18):1756–63.
- Eleid MF, Nishimura RA, Sorajja P, Borlaug BA. Systemic Hypertension in Low-Gradient Severe Aortic Stenosis With Preserved Ejection Fraction. Circulation. 2013 Sep 17;128(12):1349–53.
- Lloyd JW, Nishimura RA, Borlaug BA, Eleid MF. Hemodynamic Response to Nitroprusside in Patients With Low-Gradient Severe Aortic Stenosis and Preserved Ejection Fraction. J Am Coll Cardiol. 2017 Sep 12;70(11):1339–48.
- Claveau D, Piha-Gossack A, Friedland SN, Afilalo J, Rudski L. Complications Associated With Nitrate Use in Patients Presenting With Acute Pulmonary Edema and Concomitant Moderate or Severe Aortic Stenosis. Ann Emerg Med. 2015 Oct 1;66(4):355-362.e1.
- Alzahrani H, Woo MY, Johnson C, Pageau P, Millington S, Thiruganasambandamoorthy V. Can severe aortic stenosis be identified by emergency physicians when interpreting a simplified two-view echocardiogram obtained by trained echocardiographers? Crit Ultrasound J. 2015 Apr 18;7:5.
- Abe Y, Ito M, Tanaka C, Ito K, Naruko T, Itoh A, et al. A Novel and Simple Method Using Pocket-Sized Echocardiography to Screen for Aortic Stenosis. J Am Soc Echocardiogr. 2013 Jun;26(6):589–96.
- Furukawa A, Abe Y, Ito M, Tanaka C, Ito K, Komatsu R, et al. Prediction of aortic stenosis-related events in patients with systolic ejection murmur using pocket-sized echocardiography. J Cardiol. 2017 Jan;69(1):189–94.
- Furukawa A, Abe Y, Morizane A, Miyaji T, Hosogi S, Ito H. Simple echocardiographic scoring in screening aortic stenosis with focused cardiac ultrasonography in the emergency department. J Cardiol. 2021 Jun;77(6):613–9.



This is a fantastic continuation of the discussion on aortic stenosis! I appreciate the in-depth analysis and case studies you’ve shared. It really helps to understand the complexities of the condition and the importance of timely intervention. Looking forward to Part 3!
This post was incredibly informative! I appreciated the deep dive into the mechanisms of aortic stenosis and the latest management strategies. Such a crucial condition that often gets overlooked. Looking forward to more insights in the future!
This article wonderfully highlights the complexities of aortic stenosis. The detailed explanations and clinical insights are incredibly helpful for those of us trying to understand the condition better. I appreciate the emphasis on treatment options and the importance of early intervention. Looking forward to more posts on this topic!
Thank you for the insightful continuation on aortic stenosis! I appreciate the detailed explanations and case studies you shared. It’s eye-opening to understand the implications of this condition more clearly. Looking forward to more posts on related topics!
This post on aortic stenosis took a deep dive into the complexities of the condition. I appreciated the clear explanations and the emphasis on the importance of early diagnosis and treatment. It’s fascinating how advancements in medical technology can change the management of this disease. Looking forward to the next installment!
This post provides such a clear insight into aortic stenosis and its challenges. I appreciate how you broke down the complexities of the disease and its treatment options. Looking forward to the next part!
Very informative post—the step-by-step breakdown of managing aortic stenosis, especially in critical situations like cardiogenic shock, is really helpful. I like how you structured it into clear stages (identify causes, optimize preload, support BP), which makes complex clinical decisions easier to follow. The inclusion of ultrasound examples also adds strong practical value. It might be even better to include quick summary tables or decision flowcharts for faster reference in urgent settings.