Please refer to Dr. Mark McKinney’s Grand Round Summary from March 2020 (Parts 1 and 2) for a comprehensive literature review on the topic of resuscitating hypothermic patients. In this blog post, I’ll go into a more in-depth review of the staging of hypothermia and how to manage this problem in the field. Furthermore, I’ll summarize the resuscitation of critically ill hypothermic patients taking the most recent guidelines into consideration. Finally, I’ll review the lesser-known topic of non-freezing cold injury.
Physiology of Cold
Prolonged or unprotected exposure to cold environmental temperatures causes accidental (or primary) hypothermia, which is defined as a core body temperature ≤35 °C. Heat is lost through direct contact by way of conduction, through the motion and mixing of molecules by way of convection, and by electromagnetic radiation. Evaporation is a cooling process that expends energy to change liquid into vapour.
Our bodies seek to maintain core temperatures around 37 °C through homeostatic mechanisms. In response to cooling, detected by peripheral and central thermoreceptors, the anterior hypothalamus effects a response to increase and preserve core temperature through two main mechanisms: shivering thermogenesis and peripheral vasoconstriction.
When the cold environment overwhelms these homeostatic mechanisms, pathologic accidental hypothermia sets in. Brain activity declines and cerebral oxygen demand decreases (at approximately 6% per 1 °C drop in core temperature), reflexes slow then become absent, and pupils dilate then become unreactive (at around 28 °C). Cold slows cardiac pacemaker cells leading initially to bradycardia followed by any arrhythmia. The myocardium is very sensitive to ventricular fibrillation (VF) at <28 °C.
Within the blood, clotting factor and platelet activity are reduced. Respiratory rate slows, and cold lungs have poor compliance and elasticity leading to decreased tidal volume. Reduced antidiuretic hormone (ADH) activity leads to volume depletion by way of cold-induced diuresis. Patients experiencing accidental hypothermia are typically hypokalemic due to ion channel dysfunction. Finally, blood glucose is usually high due to catecholamine release, but in malnourished patients may be low.
Staging hypothermia is important for two big reasons: it helps direct initial management, and it enables risk stratification to guide further management. The American Heart Association (AHA), Wilderness Medical Society (WMS), and European Resuscitation Council (ERC) have put forth hypothermia staging systems summarized in the table below:
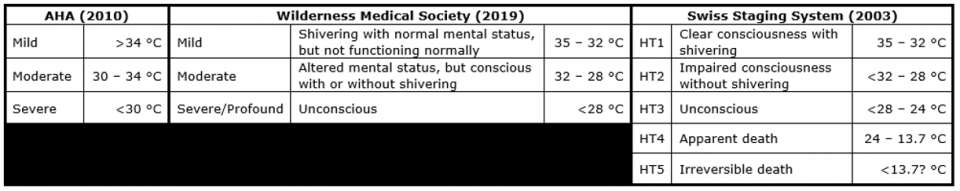
The AHA’s system revolves largely around the response to defibrillation and risk categories are based on core temperature without other clinical parameters. This staging system is not widely accepted and generally not recommended as using the 34°C changes the widely accepted definition of accidental hypothermia and the system is not applicable in situations where core temperature measurement is not feasible such as most prehospital environments.
The WMS hypothermia staging system incorporates clinical findings around mental function with some consideration for shivering. The stages roughly correlate with the listed core temperatures, but there is overlap. A major advantage of the WMS system is that it can be applied in the field in the absence of reliable core temperature measurement.
The Swiss Staging System is the oldest and probably most well-known hypothermia classification scheme. The first three stages are defined by a combination of the presence or absence of shivering and the gradual, then loss of consciousness. The later stages are apparent death, which is presumed to be reversible with treatment, and final irreversible death.
These systems are limited by the individual variability in physiologic responses and clinical signs. The reliability and accuracy of the Swiss system was called into question following the publication of a case series of patients who presented with vital signs despite core temperatures >24 °C and retrospective studies demonstrating a significant discrepancy between stages and measured core temperatures. Furthermore, the inclusion of shivering as a second variable was conflicting, a patient could be staged in one group based on the presence or absence of shivering and in another group based on level of consciousness, and shivering can occur at core temperatures <32 °C. These criticisms led to the ERC recently recommending a Revised Swiss System that uses level of consciousness (based on the AVPU system) as the main staging variable that is outlined in the table below:
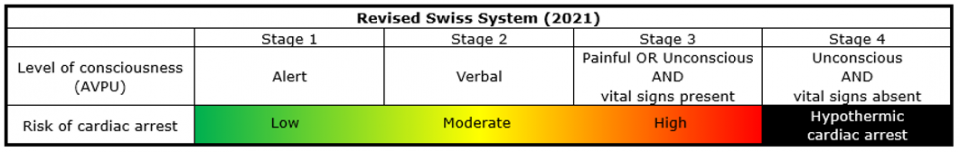
Although level of consciousness correlates linearly with core temperature, the Revised Swiss System stages do not correspond to defined temperature ranges, but rather estimate the risk of cardiac arrest, which is not discretely defined but represented on an overlapping spectrum. This system was introduced to simplify field staging and was endorsed by the WMS earlier this year for the management of avalanche and snow burial victims.
Field assessment and management of accidental hypothermia
After ensuring scene safety, one of the first steps in assessing the hypothermic patient is determining whether they’re suffering from accidental hypothermia or whether their low core temperature is due to a secondary cause. Some secondary causes of hypothermia are listed in the table below, but these lists are not exhaustive and there is overlap:

Patients may be cold-stressed but not meeting the definition of hypothermia. These patients have not had unprotected exposure to the cold for a long enough period to effect any substantial change in core body temperature. These patients may be shivering, but their core temperatures will be >35 °C. Making the distinction between cold-stressed patients and mildly hypothermic patients requires clinical judgment.
They should be otherwise well and perfectly able to take care of themselves. These patients can be managed conservatively by changing their clothes and/or setting to reduce heat loss, generating heat with light exercise, and supplementing their energy stores with high-carbohydrate beverages and/or food.
For hypothermic patients in the out-of-hospital setting, the three pillars of field management are: (1) avoiding causing cardiovascular collapse, (2) preventing and protecting from further heat loss, and (3) rewarming safely.
1. Avoid causing cardiovascular collapse. Hypothermic patients are highly susceptible to cardiovascular instability. Mechanical stimulation of a cold heart during rescue and extrication risks precipitating fatal dysrhythmias. In general, patients should be handled gently and kept horizontal. An important concept to be familiar with is core temperature afterdrop. In hypothermic patients, the relatively vasoconstricted and less massive limbs are colder than the body core and heart. Actions that might increase peripheral blood flow (such as movement or warm water immersion) can lead to a return of cold blood to the heart leading to a further drop in core temperature of up to 5 – 6 °C lowering the threshold for VF.
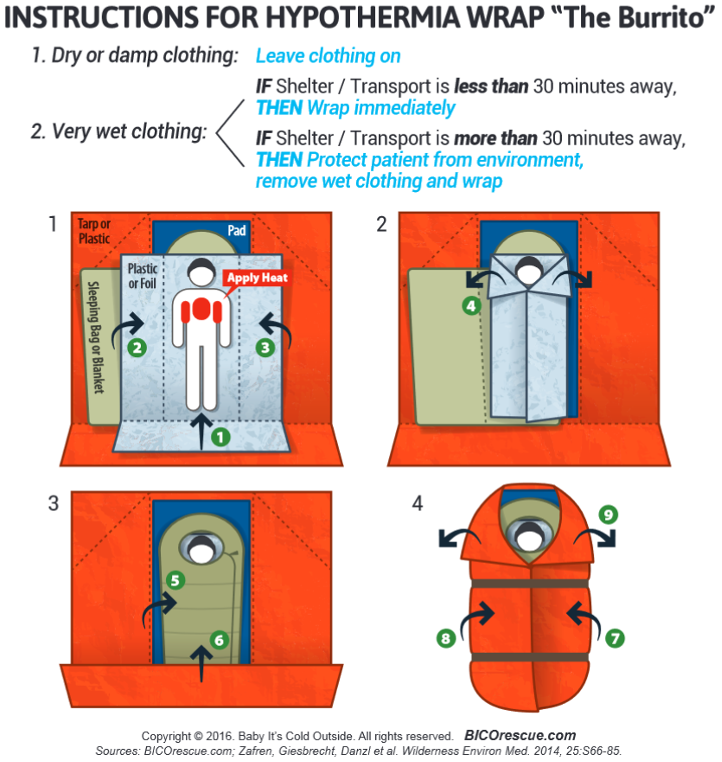
2. Prevent/protect against further heat loss. In the out-of-hospital environment, the most effective and important intervention is protecting from further heat loss. This task can be accomplished with a multilayered, so-called “burrito”, hypothermia wrap (see figure below). The first (most inner) layer in the warp should be a plastic or foil vapour barrier placed directly against the patient that protects against convective and evaporative heat loss while keeping the surrounding layers dry. The next layer is insulation: something should be placed between the patient and the ground, ideally a thick, well-insulated sleeping pad to protect against conductive heat loss. The patient should be wrapped in insulating materials (e.g. sleeping bag, blanket, etc.) making sure you maintain loft. The last outside layer, the tortilla of this burrito, is an external vapour and wind barrier made up of something like a plastic tarp or tent fly that keeps the insulation within dry and protects from further convective heat loss due to wind.
3. Rewarm in a slow, controlled manner. Field rewarming is not highly effective but should be initiated within the limits of prehospital care to supplement shivering, which alone can raise core body temperature by 3 – 4 °C/h. Active external rewarming can be provided by applying warm water bottles or large electric/chemical heating pads to the axillar, chest, then back in that order. Segmentally inspect the skin, so exposure and heat loss are minimized, every 20 – 30 min to check for early signs of burns.
Patients with hemodynamic instability or core temperatures less than 28 °C should be transported/transferred to an extracorporeal life support (ECLS)-capable center for further management.

Rewarming moderate-severely hypothermic patients in the emergency department
It is crucial to obtain an accurate core temperature measurement. Core temperature guides management and allows more accurate prognostication. In brief, the gold standard means to core temperature measurement in hypothermic patients is an esophageal probe. The patient’s airway must be protected and the probe should be advanced approximately 24 cm below the larynx into the lower third of the esophagus. IR tympanic thermometers should never be used in hypothermic patients. The rectum is not well perfused in hypothermia and changes in rectal temperature tend to lag behind the core by as much as an hour. Bladder temperature better approximates core and is an acceptable option for patients who are not intubated but is still prone to lag.
Rewarming techniques are broadly divided into passive and active rewarming. Passive rewarming includes things like drying the patient and covering them with a blanket; it depends on the patient’s functioning thermoregulatory responses and metabolic reserves. Active rewarming is adding energy into the system and is usually divided into external and internal. These are the techniques that should be employed in the ED. Active external rewarming includes forced air-warming systems and controlled water-filled blankets. These systems rewarm at approximately 0.5 – 1 °C/h. The control unit for temperature-controlled water-filled blankets should be programmed to target 36 °C. Some sources list warm intravenous fluid as external rewarming whereas others list is as internal. In any case, warmed IV fluid is not particularly effective as a rewarming modality and should never be used as the sole method. It’s value really comes from volume resuscitation. The fluid of choice is normal saline ideally warmed to 40 – 42 °C given as a bolus to improve hemodynamics.
For hemodynamically unstable patients, active internal rewarming techniques are available, but should only be used if ECLS is unavailable. Gastric lavage is not particularly effective and has an unacceptably high risk-to-benefit ratio. Bladder lavage is very slow and unreliable. Neither of these techniques are recommended. Thoracic and peritoneal lavage are more effective but again, the risks are generally not with the benefit and they should only be reserved for settings where ECLS is not available. Finally, hemodialysis or continuous renal replacement therapy have relatively high rewarming rates, but their use is limited in hemodynamically unstable patients and again should only be considered if ECLS is not an option.
Essentially, for hemodynamically unstable patients with severe primary (accidental) hypothermia, all roads lead to ECLS. Venoarterial extracorporeal membrane oxygenation (VA-ECMO) is the ECLS modality of choice for hypothermic patients with peripheral cannulation being the preferred approach. It has lower heparinization requirements and can provide hemodynamic and respiratory support as patients recover. ECMO can achieve rewarming rates as high as 15 °C/h, but the optimal rate in hypothermia is unknown and most organizations suggest slower rates of 1 – 2 °C/h. The indications for ECLS in primary hypothermia are listed below:

ECLS provides a significant survival benefit compared to other rewarming methods and is the gold standard. ECMO is continued until the patient is in a hemodynamically stable rhythm with adequate native perfusion and oxygenation and they’ve warmed to at least 32 °C.
Regarding other aspects of resuscitating hypothermic patients who have vital signs, there is limited data. Airway management should proceed as in normothermic patients. The risk of laryngoscopy or intubation triggering arrhythmia is outweighed by the need for intervention. Bear in mind that at low core temperatures, particularly <30 °C, the effects of sedative and paralytic drugs are prolonged, so doses should be reduced, and intervals extended. There is limited evidence around the use of vasoactive and antidysrhythmic agents with some concerns around decreased responsiveness and drug accumulation in tissue. No guidelines provide recommendations for or against their use.
Advanced resuscitation in hypothermic cardiac arrest
As mentioned earlier, for every degree drop in core temperature, the brain’s oxygen utilization drops 6%. This effect dramatically improved the brain’s tolerance for low- or no-flow states. As a result, patients suffering from primary, accidental hypothermia have surprisingly good outcomes. The lowest temperature any human has survived with good neurologic recovery was 11.8 °C. The longest documented no-flow time was 70 min without CPR and the longest total resuscitation was almost 9 hours. For the right patient, there is potential to make a big difference and what is survivable is very different from most other more common causes of cardiac arrest.
In suspected primary hypothermic arrest, detecting pulses is difficult, patients can tolerate relatively long no/low flow states, and mechanically stimulating a cold heart can risk converting a weakly perfusing pseudo-PEA like rhythm into a non-perfusing rhythm. Guidelines recommend a prolonged pulse check for 45 – 60 s prior to initiating CPR. Contraindications to CPR are obvious signs of fatal injury (e.g. decapitation, open head injury with loss of brain matter, truncal transection, incineration, or a chest wall that is so stiff that compressions are not possible) – traditional indicators of death such as fixed and dilated pupils, rigor mortis, and dependent lividity are unreliable.
When patients are in ventricular dysrhythmias at core temperatures less than 30 °C, defibrillation is unlikely to be successful and if successful, there is a high likelihood that the rhythm reverts to VF or VT. In general, the number of defibrillation attempts should be limited, but there is some variation among guidelines. The AHA and WMS recommend one shock, then deferring further shocks until the patient is rewarmed a few degrees or to greater than 30 °C. The ERC recommends limiting defibrillation to 3 shocks if less than 30 °C. The efficacy of vasoactive drugs is questionable and there is concern for tissue accumulation – there is reasonable consensus to hold them until warmed above 30 °C, then doubling the interval between doses until normothermia. All the guidelines emphasize that core rewarming should be the priority treatment.
Ultimately, patients in hypothermic cardiac arrest will likely need VA-ECMO. The question as to whether a patient might benefit from ECMO comes down to whether protective brain cooling occurred before critical brain hypoxia? If the answer to that question is yes, then the ECMO may be beneficial. Carefully consider whether the history is suggestive of accidental hypothermia or if might there be some other cause of hemodynamic instability (e.g. major trauma, drowning, intoxication, or witnessed arrest). If the patient has not improved after being rewarmed to around 30 – 32 °C, strongly consider alternative causes.
Recall that in accidental hypothermia, serum potassium tends to be low. Elevated potassium indicates irreversible cell death. A potassium above 12 mmol/L is generally regarded as not survivable. The HOPE score incorporates age, sex, presence or absence of asphyxia, CPR duration, serum potassium, and core temperature into a probability of survival for hypothermic cardiac arrest patients undergoing ECLS rewarming. This score is endorsed by the ERC and WMS. A cutoff probability of 10% was used to decide which hypothermic patients in cardiac arrest would benefit from ECLS in an external validation study.
Staging and management of non-freezing cold injury
Non-freezing cold injury (NFCI; formally known as trench foot or immersion foot) is a clinical diagnosis. There must be an exposure to wet cold for at least several hours at temperatures near freezing or an exposure for days at higher temperatures up to 15°C. Patients typically describe decreased sensation lasting at least 30 min and pain on rewarming. Although classically a disease of war, in more modern, urban settings, NFCI affects people through occupational exposure (e.g. dock workers), people experiencing homelessness, recreation or sport participants, and attendees to large, multi-day music festivals. The biggest risk factors are immobility, wet footwear, and a history of vasculopathy.
The pathophysiology of non-freezing cold injury is poorly understood – it’s hypothesized to be due to some combination of microcirculatory dysfunction and peripheral nerve injury. The tissue has not been frozen (unlike frostbite). There are four stages of NFCI characterized in the table below:

The first step in treating NFCI, after addressing hypothermia if present, is removing the wet clothing, elevating the limb, and gently patting it dry. The next step depends on the stage:
- If patients are in stages 1 or 2, the treatment is slow rewarming of the affected limb at room temperature. Do not immerse the limbs in warm water as that can worsen symptoms.
- If patients present in stage 3, the limb should be cooled using a fan, preferably in a cool room. If cooling does not achieve adequate pain control, then a trial of amitriptyline (50 – 100 mg PO qHS) can be considered.
Tissue necrosis and infection are rare – there’s no need for prophylactic antibiotics.
If osteomyelitis is suspected, obtain imaging to confirm.
All patients should receive tetanus prophylaxis if not up to date. Patients affected by NFCI may suffer long term complications relating to peripheral neurovascular injury with abnormal sympathetic tone and hyperalgesia. For patients suffering long-term consequences of NFCI or for patients suffering intractable pain, a discussion with Chiropody or Orthopedic Surgery is warranted. The key to prevention is wearing warm, dry clothing and air-drying at-risk limbs for at least 8 hours per day.
References
American Heart Association. Guidelines for cardiopulmonary resuscitation and emergency cardiovascular care. Part 10.4: Hypothermia. Circulation. 2005 Dec 13;112(24). doi: 10.1161/CIRCULATIONAHA.105.166566
Bristow I. Don’t forget trench foot. The Podiatric Dermatology Blog. November 19, 2019. https://www.foot.expert/post/2019/11/10/trench.
Bush JS, Lofgran T, Watson S. Trench Foot. [Updated 2023 Aug 8]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK482364/
Dow J et al. Wilderness Medical Society Clinical Practice Guidelines for the Out-of-Hospital Evaluation and Treatment of Accidental Hypothermia: 2019 Update. Wilderness Environ Med. 2019 Dec;30(4S):S47-S69. doi: 10.1016/j.wem.2019.10.002
Mroczek T, Gladki M, Skalski J. Successful resuscitation from accidental hypothermia of 11.8°C: where is the lower bound for human beings? Eur J Cardiothorac Surg. 2020 Nov 1;58(5):1091-1092. doi: 10.1093/ejcts/ezaa159
Musi ME, Sheets A, Zafren K, Brugger H, Paal P, Hölzl N, Pasquier M. Clinical staging of accidental hypothermia: The Revised Swiss System: Recommendation of the International Commission for Mountain Emergency Medicine (ICAR MedCom). Resuscitation. 2021 May;162:182-187. doi: 10.1016/j.resuscitation.2021.02.038
Paal P et al. Accidental hypothermia-an update: The content of this review is endorsed by the International Commission for Mountain Emergency Medicine (ICAR MEDCOM). Scand J Trauma Resusc Emerg Med. 2016 Sep 15;24(1):111. doi: 10.1186/s13049-016-0303-7
Paal P, et al. Accidental Hypothermia: 2021 Update. Int J Environ Res Public Health. 2022 Jan 3;19(1):501. doi: 10.3390/ijerph19010501
Zafren K, Danzl D. Hypothermia, frostbite, and nonfreezing cold injuries. In: Rosen’s Emergency Medicine: Concepts and Clinical Practice. 10th ed. Elsevier; 2023:1750-1770.
Zafren K. Nonfreezing Cold Injury (Trench Foot). Int J Environ Res Public Health. 2021 Oct 6;18(19):10482. doi: 10.3390/ijerph181910482


