Review of the formative literature of our current tPA time window in Acute Ischemic Stroke
After numerous case series and smaller prospective studies suggesting benefit for thrombolytic therapy (tPA), the early 90s ushered in the era of thrombolytic RCTs for acute ischemic stroke. Below, you will find a comprehensive review of these trials, and their impact on the development of the current tPA time window for the treatment of acute ischemic stroke. A second part blog will follow, to address the controversies surrounding the use of tPA.
For an updated review of current ED management of Acute Stroke, you can visit our Grand Rounds Summary from 2019.
ECASS-1
This was the first tPA trial. It was a double blinded RCT of patients with acute ischemic stroke presenting to hospital within 6 hours of symptom onset.
Data analysis utilized both intention-to-treat and modified intention to treat strategies. Justification for the modified analysis included a high rate of protocol violation (17.4%) which would disproportionately affect the tPA arm.
With respect to functional independence, they found a clinically relevant but not statistically significant improvement in those who received tPA (OR 1.15 [95% CI 0.98-1.35], P = 0.38). With respect to their modified analysis, the improvement in functional independence became statistically significant: (OR 1.29 [95% CI 1.09-1.54], P = 0.035).
A signal of harm was present. In the ITT analysis, there was an absolute increase in mortality of 5.2%, P = 0.08. In the modified analysis, this figure was more marginal at 2.9%, P = 0.36. [1]
NINDS
NINDS is one of the better-known stroke trials. This RCT examined outcomes in patients who received tPA within 3 hours of symptoms onset. They found a statistically significant 13% absolute difference in minimal or no disability favouring tPA. Rates of symptomatic intracranial hemorrhage were elevated in tPA recipients at 6%, P < 0.001. Mortality rates however were similar, and if anything favoured tPA (4% absolute improvement, P = 0.3).
This trial was the impetus for the initial 3-hour time window. [2]
ECASS-II
This RCT focused on examining patients presenting between 0-3h and 3-6h. The study dichotomized patients to excellent (mRS 0-1) or poor (mRS 2-6) outcomes. It is worth nothing that this dichotomization amongst stroke patients is optimistic.
They found a non-significant 3.7% (P = 0.277) benefit for tPA recipients. Results were similar and did not reach statistical significant when broken down by time period. Of note however, only 20% of their patients were in the 0-3h time period, and so the study was inadequately powered to truly examine this time period.
When the data was re-examined with dichotomization around good (mRS 0-2) versus poor (mRS 3-6) outcomes, a statistically significant benefit was found for tPA (8.3% benefit, P = 0.024).
No difference was seen between groups regarding mortality (0.2% difference, P = 0.816). [3]
ATLANTIS B
ATLANTIS B was a North American study investigating treatment of patients between 3-5 hours after symptoms onset.Primary outcome was excellent neurologic outcomes (NIHSS 0-1 at 90 days). No difference was seen (2%, P = 0.65) between groups. Moreover, an ordinal analysis didn’t highlight any benefit either.
A 5.4% (p < 0.001) absolute increase in symptomatic intracranial hemorrhage was seen in the tPA arm. Concerningly, there was also a trend towards increased mortality in those who received tPA (4% absolute increase, P = 0.08).
This trial ultimately was stopped due to futility, however, many rightfully argue that based on the data presented, had the trial not been stopped when it was, it likely would have been stopped eventually due to concerns of harm.
This was the FIRST negative tPA trial. [4]
ATLANTIS A
The sibling to the ATLANTIS B study, ATLANTIS A evaluated tPA administration across multiple time windows between 0-6h, with 35% of its participants in the 5-6h time period.
Their primary outcome was a comparison between proportions of patients who had a 4-point or greater improvement in NIHSS at 30 days, as compared to at the time of presentation.
They found a 15% absolute difference (P = 0.05) favouring the placebo arm.
Moreover, the study showed statistically significant absolute increases in symptomatic ICH (11.3%) and mortality (15.5%) for tPA participants.
This staggering profile of harm was largely driven by the 5-6h cohort, which had mortality and symptomatic ICH rates of 31.9% and 18.2%, respectively. It is worth noting that patients with CT findings of more advanced strokes who had been excluded in previous trials were included in this one. Additionally, by chance, stroke severity was greater in the 5-6h cohort. Thus, it is difficult to know if the profile of harm for tPA seen in this study was due to thrombolysis of completed infarctions, an effect of more severe strokes in the tPA arm, or a product of chance. [5]
ECASS-III
The ECASS-III trial re-examined the idea of expanding the tPA window, following the negative ATLANTIS trials. This study looked exclusively at strokes between 3 and 4.5 hours, with a primary outcome focused on excellent neurological outcomes.
They found a 7.2% (P = 0.04) absolute benefit for excellent neurological outcomes. This benefit was seen across severities of disability on ordinal analysis (P = 0.02).
Additionally, it came in the context of a more palatable harm profile, with symptomatic intracranial hemorrhage rates of 2.4% (P = 0.008) as compared to the ~6-10% in previous trials. Mortality was no different between arms (0.7% difference, P = 0.68). [6]
IST-3
The IST-3 trial looked to put the question of tPA for acute ischemic stroke administered within 6 hours to rest. It was a pragmatic, international, multi-center RCT with a larger sample size.
The primary outcome of this study was the number of patients who were alive and independent (Oxford Handicap Score 0-2) at 6 months. The trial found a modest trend towards benefit without statistical significant (OR 1.13 [95% CI 0.95 – 1.36], P = 0.181) for tPA.
An ordinal analysis added as a secondary outcome in a protocol amendment did identify a statistically significant benefit for tPA however (OR 1.17 [95% CI 1.03 – 1.33], P = 0.016).
Rates of symptomatic intracranial hemorrhage were consistent with what had been seen before for those who received tPA (6%, p < 0.0001), and mortality was no different between arms (0% difference, P = 0.672). [7]
Summary
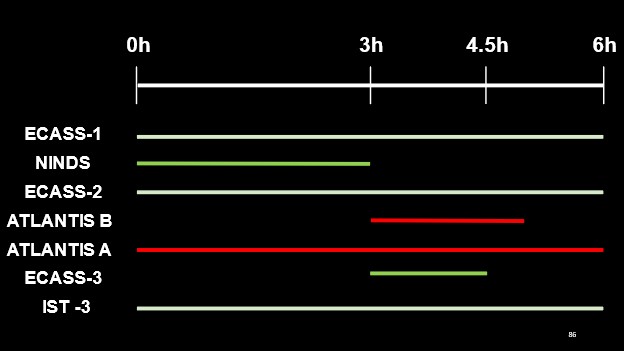 To summarize the findings of these trials as it pertains to thrombolytic therapy (tPA), this figure highlights the time windows evaluated by each trial and their respective findings (white lines indicating equivocal results, red for negative outcomes with tPA administration and green indicating a favourable outcome with tPA administration).
To summarize the findings of these trials as it pertains to thrombolytic therapy (tPA), this figure highlights the time windows evaluated by each trial and their respective findings (white lines indicating equivocal results, red for negative outcomes with tPA administration and green indicating a favourable outcome with tPA administration).
References
[1] Hacke W, Kaste M, Fieschi C, Toni D, Lesaffre E, Von Kummer R, et al. Intravenous Thrombolysis With Recombinant Tissue Plasminogen Activator for Acute Hemispheric Stroke: The European Cooperative Acute Stroke Study (ECASS). JAMA J Am Med Assoc 1995;274:1017–25. https://doi.org/10.1001/jama.1995.03530130023023.
[2] Group TNI of ND and S rt-PSS. Tissue Plasminogen Activator for Acute Ischemic Stroke. N Engl J Med 1995;333:1581–8. https://doi.org/10.1056/NEJM199512143332401.
[3] Hacke W, Kaste M, Fieschi C, Von Kummer R, Davalos A, Meier D, et al. Randomised double-blind placebo-controlled trial of thrombolytic therapy with intravenous alteplase in acute ischaemic stroke (ECASS II). Lancet 1998;352:1245–51. https://doi.org/10.1016/S0140-6736(98)08020-9.
[4] Clark WM, Wissman S, Albers GW, Jhamandas JH, Madden KP, Hamilton S. Recombinant tissue-type plasminogen activator (Alteplase) for ischemic stroke 3 to 5 hours after symptom onset the ATLANTIS study: A randomized controlled trial. J Am Med Assoc 1999;282:2019–26. https://doi.org/10.1001/jama.282.21.2019.
[5] Clark WM, Albers GW, Madden KP, Hamilton S. The rtPA (Alteplase) 0- to 6-hour acute stroke trial, part A (A0276g): Results of a double-blind, placebo-controlled, multicenter study. Stroke 2000;31:811–6. https://doi.org/10.1161/01.STR.31.4.811.
[6] Hacke W, Kaste M, Bluhmki E, Brozman M, Dávalos A, Guidetti D, et al. Thrombolysis with Alteplase 3 to 4.5 Hours after Acute Ischemic Stroke. N Engl J Med 2008;359:1317–29. https://doi.org/10.1056/nejmoa0804656.
[7] Sandercock P, Wardlaw JM, Lindley RI, Dennis M, Cohen G, Murray G, et al. The benefits and harms of intravenous thrombolysis with recombinant tissue plasminogen activator within 6 h of acute ischaemic stroke (the third international stroke trial [IST-3]): A randomised controlled trial. Lancet 2012;379:2352–63. https://doi.org/10.1016/S0140-6736(12)60768-5.




Trackbacks/Pingbacks